Advanced Green Synthesis of Cimetidine: A Scalable Solution for Pharmaceutical Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high purity with environmental sustainability, a challenge effectively addressed by the innovative production method detailed in patent CN113234018B. This proprietary technology outlines a sophisticated five-step synthetic route for cimetidine, a critical H2 receptor antagonist used globally for treating digestive ulcers, which fundamentally restructures the traditional supply chain dynamics for this active pharmaceutical ingredient. By shifting away from hazardous reagents and low-yield processes, this method offers a compelling value proposition for stakeholders focused on cost reduction in pharmaceutical intermediates manufacturing and regulatory compliance. The core innovation lies in the strategic selection of 2-chloroethanol as a starting material, coupled with a unique oxidation sequence that bypasses the generation of noxious sulfur-containing byproducts common in legacy syntheses. For R&D directors and procurement managers alike, understanding the mechanistic advantages of this route is essential for securing a reliable cimetidine supplier capable of meeting stringent quality standards without compromising on delivery timelines or ecological responsibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the large-scale production of cimetidine has been plagued by significant technical and environmental hurdles that inflate operational costs and complicate waste management protocols. As illustrated in the reaction schemes of prior art, traditional routes often rely on the condensation of phenol derivatives or the use of cyanogen chloride, a substance known for its severe lachrymatory properties and high toxicity, posing substantial risks to worker safety and requiring specialized containment infrastructure. Furthermore, existing industrial processes frequently exhibit a disappointing overall yield of approximately 46%, necessitating larger batch sizes to meet demand and thereby increasing the burden on downstream purification systems. Perhaps most critically, these conventional methods generate methyl mercaptan as a unavoidable byproduct, a compound characterized by its intensely offensive odor and potential for environmental contamination, which forces manufacturers to invest heavily in scrubbing technologies and odor control measures to maintain community relations and regulatory standing.
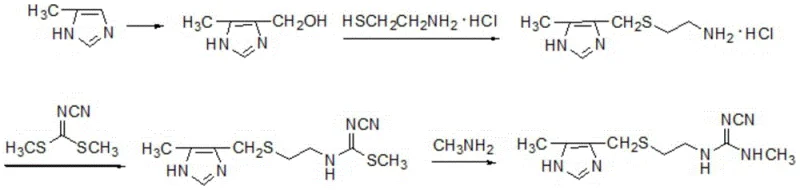
The Novel Approach
In stark contrast to these legacy challenges, the novel approach disclosed in the patent introduces a streamlined, eco-friendly pathway that leverages abundant and inexpensive feedstocks to achieve superior process efficiency. The new methodology initiates with the nucleophilic substitution of 2-chloroethanol using thiocyanate salts, a reaction facilitated by catalytic amounts of iodide to ensure high conversion rates under mild reflux conditions. This strategic pivot eliminates the need for dangerous cyanogen chloride and completely avoids the formation of methyl mercaptan, thereby resolving the persistent odor issues that have long burdened cimetidine production facilities. The subsequent steps involve a carefully controlled oxidation using peroxyacetic acid and a series of condensation reactions that build the guanidine moiety with high fidelity, ultimately delivering a total yield exceeding 63%, which represents a dramatic improvement in material throughput and resource utilization for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Peroxyacetic Acid Oxidation and Guanidine Formation
The chemical elegance of this synthesis is best understood through the lens of its key transformation steps, particularly the oxidation of the thiourea intermediate and the subsequent construction of the cyanoguanidine group. In the third step of the sequence, the intermediate (II), which contains a thioamide functionality, undergoes oxidation with peroxyacetic acid to form intermediate (III), a sulfonic acid derivative that serves as a highly activated electrophile. This oxidation must be conducted with rigorous temperature control, typically maintaining the reaction mixture between 8°C and 20°C during the addition of the oxidant, to prevent over-oxidation or decomposition of the sensitive nitrogen-sulfur framework. The resulting sulfonic acid species is uniquely positioned to react with cyanamide in the fourth step, facilitating a nucleophilic attack that displaces the sulfonate group and installs the critical nitrile functionality required for the final drug structure, showcasing a clever use of leaving group ability to drive the reaction forward without requiring harsh coupling agents.
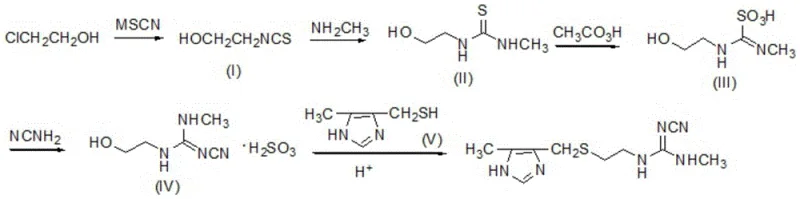
Beyond the primary bond-forming events, the impurity profile of the final cimetidine product is meticulously managed through the specific choice of solvents and recrystallization protocols embedded within the process design. The use of ethanol as a primary solvent for multiple steps, including the aminolysis and the final condensation with imidazole thiol, ensures that polar byproducts and inorganic salts remain soluble or can be easily filtered off during the workup phases. For instance, the final purification involves adjusting the pH to alkaline conditions followed by hot filtration with activated carbon, a dual-action step that removes colored impurities and residual metal ions while promoting the crystallization of the target molecule in its thermodynamically stable form. This attention to detail in the downstream processing guarantees that the resulting high-purity cimetidine meets the rigorous specifications demanded by global pharmacopeias, minimizing the risk of batch rejection and ensuring consistent therapeutic efficacy for the end-user.
How to Synthesize Cimetidine Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to maximize yield and minimize side reactions. The process begins with the preparation of the isothiocyanate intermediate, followed by sequential functionalization to build the side chain before the final cyclization-like attachment to the imidazole ring. Operators must pay close attention to the stoichiometry of the peroxyacetic acid and the temperature gradients during the exothermic oxidation phase to ensure safety and reproducibility. The detailed standardized synthetic steps, including specific molar ratios, reaction times, and isolation techniques for each of the five intermediates, are outlined in the comprehensive guide below to assist technical teams in replicating this high-efficiency protocol.
- React 2-chloroethanol with thiocyanate (MSCN) in the presence of an iodide catalyst to form intermediate (I), 2-hydroxyethyl isothiocyanate.
- Condense intermediate (I) with methylamine in ethanol to produce the thiourea derivative, intermediate (II).
- Oxidize intermediate (II) using peroxyacetic acid at controlled low temperatures to generate the activated sulfonic acid intermediate (III).
- React intermediate (III) with cyanamide to form the guanidine precursor, intermediate (IV).
- Perform the final condensation between intermediate (IV) and imidazole thiol (V) under acidic reflux conditions to yield high-purity cimetidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented production method translates directly into tangible operational improvements and risk mitigation strategies that enhance the overall resilience of the pharmaceutical supply network. By replacing scarce or hazardous raw materials like cyanogen chloride with commodity chemicals such as 2-chloroethanol and ammonium thiocyanate, manufacturers can secure a more stable supply base that is less susceptible to market volatility or regulatory restrictions on toxic substances. This shift not only simplifies the logistics of raw material acquisition but also drastically reduces the costs associated with specialized storage and handling of dangerous goods, allowing for a more streamlined and cost-effective inventory management system that supports continuous production schedules without interruption.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the elimination of expensive reagents and the significant increase in overall process yield, which reduces the cost of goods sold per kilogram of active ingredient. By avoiding the generation of methyl mercaptan, facilities save substantially on waste treatment and odor abatement technologies, while the use of common solvents like water and ethanol lowers the expenditure on specialized organic solvents and their recovery systems. Furthermore, the high conversion rates in each step minimize the volume of mother liquor requiring recycling or disposal, leading to a leaner manufacturing process that maximizes asset utilization and delivers substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production is not bottlenecked by the supply constraints often associated with niche fine chemical intermediates. Since the starting materials are produced on a massive industrial scale for other applications, the risk of shortage is negligible, providing a reliable cimetidine supplier with the confidence to commit to long-term contracts and just-in-time delivery models. This stability is crucial for downstream pharmaceutical companies that require uninterrupted access to high-quality intermediates to maintain their own finished dosage form production lines and meet patient demand globally.
- Scalability and Environmental Compliance: From an environmental perspective, the absence of malodorous byproducts and toxic reagents simplifies the permitting process for new production lines and reduces the regulatory burden on existing facilities. The process is inherently safer and cleaner, making it easier to scale from pilot plant quantities to multi-ton commercial production without encountering the exponential increase in waste management complexity seen in older technologies. This alignment with green chemistry principles not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the corporate social responsibility profile of the brand, appealing to increasingly eco-conscious stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced cimetidine synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, offering clarity on how this method outperforms traditional approaches in terms of yield, safety, and environmental impact. Understanding these nuances is vital for technical decision-makers evaluating the feasibility of adopting this route for their own manufacturing portfolios or sourcing strategies.
Q: How does this new cimetidine production method improve upon conventional synthesis routes?
A: Conventional routes often suffer from low overall yields (around 46%) and generate hazardous byproducts like methyl mercaptan, which causes severe odor and environmental pollution. This patented method utilizes readily available raw materials like 2-chloroethanol and avoids toxic reagents such as cyanogen chloride, achieving a significantly higher total yield of over 63% while eliminating malodorous emissions.
Q: What are the critical reaction conditions for the oxidation step in this process?
A: The oxidation of intermediate (II) to intermediate (III) requires precise temperature control to ensure safety and selectivity. The process involves dropwise addition of peroxyacetic acid while maintaining the reaction system temperature below 20°C, typically between 8-10°C initially, followed by stirring at room temperature to complete the conversion efficiently.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It employs common solvents like water and ethanol, uses inexpensive catalysts like potassium iodide, and simplifies purification steps through crystallization and filtration, making it highly cost-effective and environmentally compliant for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cimetidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is paramount for the future competitiveness of the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN113234018B are fully realized in practical, large-volume manufacturing environments. We are committed to delivering high-purity cimetidine that adheres to stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch against international standards.
We invite global partners to engage with our technical procurement team to discuss how this innovative production method can optimize your supply chain and reduce your overall manufacturing footprint. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the specific economic advantages tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your pharmaceutical operations.
