Advanced Green Synthesis of Cimetidine: Technical Breakthroughs for Commercial Scale-Up
Advanced Green Synthesis of Cimetidine: Technical Breakthroughs for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways that balance high efficiency with stringent environmental standards. Patent CN114394939A introduces a transformative three-step synthetic methodology for producing cimetidine, a critical histamine H2 receptor antagonist widely used in treating peptic ulcers. This innovative approach addresses long-standing challenges in API intermediate production by eliminating hazardous by-products and simplifying operational complexity. By leveraging readily available raw materials and mild reaction conditions, this technology offers a compelling value proposition for a reliable cimetidine intermediate supplier aiming to optimize their production portfolio. The process achieves a total yield exceeding 90% and purity levels above 99.50%, demonstrating exceptional potential for industrial application.
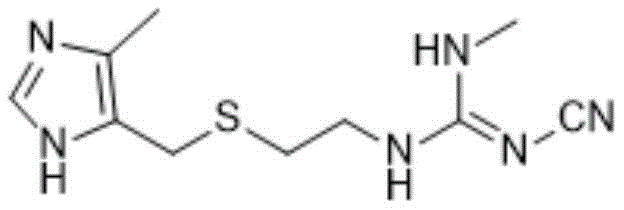
Furthermore, the strategic design of this synthesis route minimizes the generation of toxic waste, specifically avoiding the production of methyl mercaptan which plagues older methodologies. This aligns perfectly with modern green chemistry principles, reducing the burden on waste treatment facilities and lowering overall operational risks. For procurement managers and supply chain heads, understanding the nuances of this patent is crucial for securing a stable supply of high-quality active pharmaceutical ingredients. The following analysis delves into the technical specifics, comparing this novel approach against conventional limitations to highlight its commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cimetidine has been fraught with significant environmental and operational hurdles that impact cost and scalability. Traditional routes, such as the one disclosed in CN 101838241, rely on the condensation of 4-hydroxymethyl-5-methylimidazole hydrochloride with cystamine hydrochloride, followed by reaction with dimethyl cyanamide sulfonate. A critical drawback of this legacy process is the generation of methyl mercaptan, a volatile compound with an intensely offensive odor that poses severe safety and environmental hazards. The handling of such malodorous by-products requires specialized containment systems and extensive scrubbing technologies, driving up capital expenditure and operational costs. Additionally, these multi-step sequences often suffer from prolonged production cycles and moderate yields, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN114394939A presents a streamlined alternative that circumvents these issues through a clever rearrangement of synthetic logic. The new route initiates with the methylamination of 2-cyanoimino-1,3-thiazolidine to form a key guanidine intermediate, bypassing the need for toxic sulfonating agents entirely. This fundamental shift in chemistry eliminates the formation of methyl mercaptan, thereby drastically simplifying the environmental compliance landscape for manufacturers. The process operates under mild conditions, typically between 25°C and 60°C, which reduces energy consumption and enhances safety profiles. By integrating these improvements, the novel approach facilitates cost reduction in pharmaceutical manufacturing while ensuring a consistent supply of high-quality material suitable for global regulatory standards.
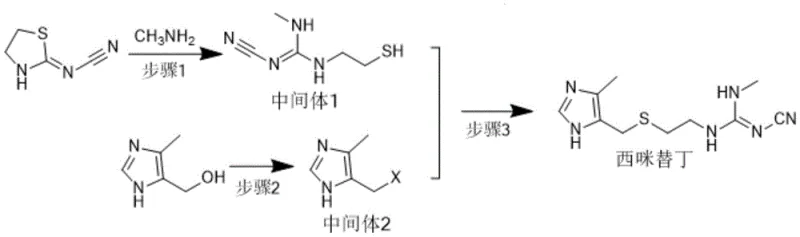
Mechanistic Insights into Methylamination and Coupling Reactions
The core of this synthetic breakthrough lies in the precise control of nucleophilic substitution and ring-opening reactions. The first step involves the reaction of 2-cyanoimino-1,3-thiazolidine with methylamine, where the amine acts as a nucleophile to open the thiazolidine ring. This methylamination is highly sensitive to temperature and molar ratios; experimental data indicates that maintaining a temperature range of 40°C to the solvent boiling point, with a molar ratio of substrate to methylamine between 1:1.5 and 1:3, optimizes the conversion to N-cyano-N'-(2-mercaptoethyl)-N''-methylguanidine. Deviations from these parameters can lead to incomplete reactions or the formation of side products, underscoring the importance of rigorous process control in commercial scale-up of complex pharmaceutical intermediates.
Subsequent steps focus on the activation of the imidazole moiety and its coupling with the guanidine intermediate. The halogenation of 4-hydroxymethyl-5-methylimidazole hydrochloride using thionyl chloride converts the hydroxyl group into a reactive chloromethyl group, creating a potent electrophile. This intermediate then undergoes a nucleophilic attack by the thiol group of the guanidine derivative under alkaline conditions. The choice of base, whether inorganic like sodium hydroxide or organic like triethylamine, plays a pivotal role in neutralizing the generated acid and driving the equilibrium forward. As illustrated in the reaction scheme below, this final coupling step is exothermic and requires careful thermal management to prevent degradation, ensuring the final product meets the stringent purity specifications required for API production.
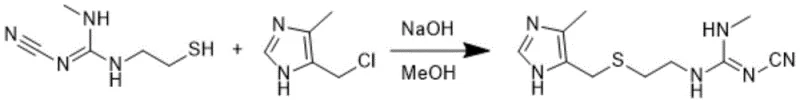
How to Synthesize Cimetidine Efficiently
Implementing this synthesis route requires a disciplined approach to reaction parameters and workup procedures to maximize efficiency. The process is divided into three distinct stages: the preparation of the guanidine intermediate, the halogenation of the imidazole precursor, and the final condensation. Each stage demands specific solvent systems, such as ethanol for the aminolysis and dichloroethane for the chlorination, to ensure optimal solubility and reaction kinetics. Operators must adhere strictly to the recommended stoichiometric ratios and temperature profiles to achieve the reported yields of over 90%. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Perform methylamination of 2-cyanoimino-1,3-thiazolidine with methylamine in organic solvent at 40°C to boiling point to obtain Intermediate 1.
- Conduct halogenation of 4-hydroxymethyl-5-methylimidazole hydrochloride using thionyl chloride at 25-40°C to generate Intermediate 2.
- React Intermediate 1 and Intermediate 2 under alkaline conditions at temperatures below solvent boiling point to finalize cimetidine synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this green synthesis route offers substantial strategic benefits for organizations focused on cost efficiency and supply chain resilience. The elimination of hazardous by-products like methyl mercaptan not only reduces waste disposal costs but also mitigates the risk of production shutdowns due to environmental non-compliance. Furthermore, the use of commercially available and inexpensive starting materials ensures that the cost of goods sold remains competitive, even in fluctuating raw material markets. This stability is crucial for procurement managers negotiating long-term contracts for high-purity cimetidine, as it guarantees a predictable pricing structure and reliable delivery schedules.
- Cost Reduction in Manufacturing: The simplified workflow significantly lowers operational expenditures by removing the need for complex scrubbing systems required to handle toxic sulfur gases. By avoiding expensive reagents like dimethyl cyanamide sulfonate and utilizing common solvents such as methanol and ethanol, the overall material cost is substantially reduced. Additionally, the high yield of each step minimizes raw material waste, directly contributing to improved profit margins and resource efficiency in large-scale production facilities.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway ensures consistent output quality, reducing the likelihood of batch failures that can disrupt supply chains. Since the raw materials are widely sourced and the reaction conditions are mild, manufacturers can easily scale production from pilot batches to multi-ton quantities without significant re-engineering. This flexibility allows suppliers to respond rapidly to market demand surges, providing a secure source of critical API intermediates for downstream drug formulation.
- Scalability and Environmental Compliance: The absence of noxious emissions aligns the process with increasingly strict global environmental regulations, facilitating smoother permitting and inspection processes. The ability to recycle solvents like dichloroethane and thionyl chloride further enhances the sustainability profile of the operation. This eco-friendly approach not only protects the corporate brand reputation but also future-proofs the manufacturing asset against tightening legislative frameworks regarding industrial emissions and waste management.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology, we have compiled answers to common inquiries regarding the process specifics and quality outcomes. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these details is essential for assessing the feasibility of integrating this route into existing manufacturing lines or for qualifying new vendors.
Q: How does this new synthesis method improve environmental safety compared to conventional routes?
A: Unlike traditional methods such as CN 101838241 which generate malodorous and volatile methyl mercaptan by-products, this novel route utilizes 2-cyanoimino-1,3-thiazolidine and avoids toxic sulfur-containing waste, significantly enhancing EHS compliance.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: Optimal yields exceeding 90% are achieved by maintaining the methylamination step at 40°C with a molar ratio of 1:1.5-3, and performing the final coupling at 50°C with a molar ratio of intermediates at 1:1.0-1.5.
Q: What purity levels can be expected from this synthetic pathway?
A: The process consistently delivers cimetidine with a purity greater than 99.50%, meeting stringent pharmacopoeia standards through simple recrystallization without complex purification cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cimetidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering cimetidine and its intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can drive value and efficiency for your organization.
