Advanced Asymmetric Hydrogenation Technology for High-Purity Optically Active Carbonyl Compounds
The pharmaceutical and fine chemical industries constantly seek robust methodologies for generating chiral building blocks with exceptional optical purity. Patent CN101675020A presents a significant technological advancement in the field of asymmetric synthesis, specifically detailing a method for synthesizing optically active carbonyl compounds through the asymmetric hydrogenation of alpha,beta-unsaturated carbonyl compounds. This innovation addresses long-standing challenges in homogeneous catalysis, particularly regarding the stability and reusability of transition metal catalysts. By employing optically active transition metal catalysts that are soluble in the reaction mixture and possess at least one carbon monoxide ligand, the process achieves superior control over stereochemistry. The core breakthrough lies in the precise management of carbon monoxide concentrations during different stages of the catalytic cycle, ensuring that the catalyst remains active and stable throughout prolonged operation. This technology is particularly relevant for the production of high-value intermediates used in flavors, fragrances, and active pharmaceutical ingredients, where enantiomeric excess is a critical quality attribute.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of alpha,beta-unsaturated aldehydes and ketones has been plagued by issues related to catalyst deactivation and inconsistent enantioselectivity. Conventional homogeneous catalytic systems often suffer from insufficient stability of the catalyst complexes or the catalytically active metal species formed in situ. In many prior art methods, such as those described in earlier patents like WO 2006/040096, the pretreatment of the catalyst and the subsequent hydrogenation reaction were difficult to optimize simultaneously regarding carbon monoxide concentration. Typically, catalyst pretreatment required significantly higher concentrations of carbon monoxide than the actual hydrogenation step. When these steps were not strictly separated or controlled, large amounts of carbon monoxide from the pretreatment phase could be entrained into the hydrogenation reactor. This excess carbon monoxide often acted as a catalyst poison, inhibiting the hydrogenation activity and leading to lower yields and reduced catalyst turnover numbers. Furthermore, controlling the carbon monoxide concentration in the reaction mixture during hydrogenation was technically challenging, often resulting in batch-to-batch variability that is unacceptable for commercial manufacturing of high-purity intermediates.
The Novel Approach
The methodology disclosed in CN101675020A overcomes these deficiencies through a distinct three-stage process that decouples catalyst activation from the hydrogenation reaction. First, the catalyst precursor is pretreated with a specific gas mixture containing 20-90 volume percent carbon monoxide and 10-80 volume percent hydrogen at pressures ranging from 5 to 100 bar. Crucially, the second step involves the removal of excess, unbound carbon monoxide from the pretreated catalyst before it is introduced to the hydrogenation environment. This ensures that the catalyst enters the reaction zone in an optimized state without carrying over inhibitory levels of CO. Finally, the asymmetric hydrogenation is conducted in the presence of hydrogen that contains a strictly controlled, low concentration of additional carbon monoxide, specifically between 100 and 1200 ppm. This trace amount of CO is sufficient to stabilize the active catalyst species and prevent decomposition but is low enough to avoid poisoning the catalytic cycle. This precise engineering of the reaction environment allows for the asymmetric hydrogenation to proceed at a substantially constant carbon monoxide concentration, leading to consistent product quality and extended catalyst lifespan.
Mechanistic Insights into CO-Stabilized Asymmetric Hydrogenation
The success of this synthetic route relies heavily on the intricate interplay between the transition metal center, the chiral ligand, and the carbon monoxide promoter. The transition metal catalysts employed are typically soluble complexes of Group VIII metals, with Rhodium (Rh) and Iridium (Ir) being particularly preferred due to their high activity and selectivity profiles. These metals are coordinated with optically active ligands containing at least one phosphorus or arsenic atom, with chiral diphosphines like (R,R)-chiraphos being exemplary candidates. The presence of at least one carbon monoxide ligand in the catalyst structure is fundamental to its stability. During the catalytic cycle, the CO ligand helps maintain the electronic and steric environment necessary for the metal center to facilitate the hydride transfer to the olefinic double bond of the substrate. Without this stabilization, the metal complex might aggregate into inactive clusters or decompose under the thermal and pressure stresses of the reaction. The mechanism involves the coordination of the alpha,beta-unsaturated substrate to the metal center, followed by migratory insertion and reductive elimination to form the saturated chiral product. The careful regulation of CO partial pressure ensures that the coordination sphere of the metal remains intact without blocking the substrate binding sites entirely.
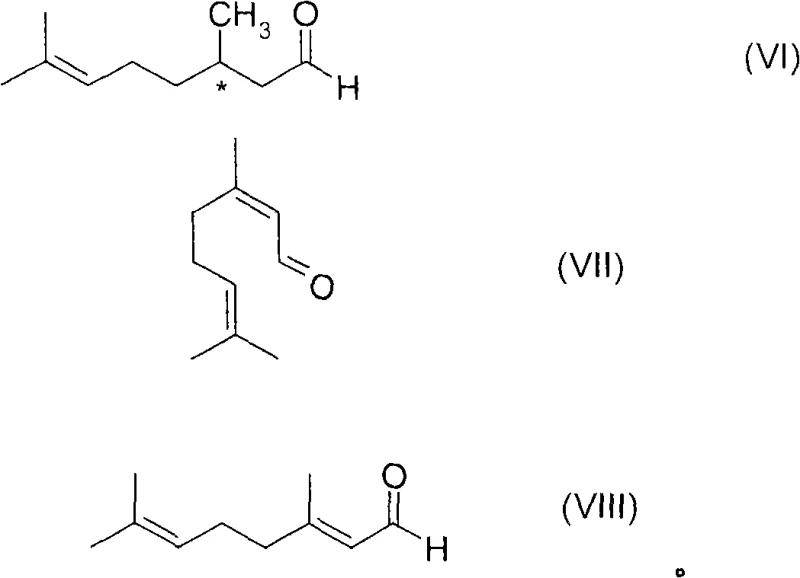
Impurity control is another critical aspect managed by this mechanistic design. In conventional hydrogenation, side reactions such as isomerization of the double bond or over-reduction can occur if the catalyst speciation is not uniform. By maintaining a constant and low level of carbon monoxide (100-1200 ppm) during the reaction, the system suppresses the formation of non-selective catalytic species that might lead to racemic byproducts or structural isomers. The patent highlights that the maximum achievable enantiomeric excess depends on the purity of the starting material, particularly the E/Z isomer ratio of the double bond. However, the robust nature of the CO-stabilized catalyst ensures that even with mixed isomer feeds, the process can yield the desired enantiomer with high fidelity, often achieving enantiomeric excess values of at least 80 percent, and frequently reaching up to 99 percent ee. This level of stereocontrol is essential for downstream applications where the biological activity or olfactory profile is strictly dependent on the absolute configuration of the molecule.
How to Synthesize Optically Active Carbonyl Compounds Efficiently
The synthesis of these high-value chiral intermediates requires precise adherence to the patented protocol to maximize yield and optical purity. The process begins with the selection of appropriate starting materials, such as alpha,beta-unsaturated aldehydes or ketones, which may include complex terpenes like geranial or neral. The detailed operational parameters, including pressure ranges of 5-100 bar for pretreatment and temperatures between 0 and 100 degrees Celsius for hydrogenation, must be strictly monitored. The following guide outlines the standardized approach derived from the patent specifications to ensure reproducible results in a laboratory or pilot plant setting.
- Pretreat the catalyst precursor with a gas mixture containing 20-90% carbon monoxide and 10-80% hydrogen at 5-100 bar pressure.
- Remove excess unbound carbon monoxide from the pretreated catalyst prior to the hydrogenation step.
- Conduct asymmetric hydrogenation in the presence of hydrogen containing a controlled carbon monoxide content of 100-1200 ppm.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the enhanced stability and reusability of the homogeneous catalyst system. In traditional homogeneous catalysis, the high cost of precious metal catalysts like Rhodium and Iridium, combined with chiral ligands, often makes the process economically prohibitive unless the catalyst can be recycled efficiently. By stabilizing the catalyst with controlled carbon monoxide levels, this method significantly extends the operational life of the catalyst, allowing for multiple cycles or continuous operation without frequent replenishment. This directly impacts the cost of goods sold by reducing the consumption of expensive catalytic materials per kilogram of product produced. Furthermore, the ability to run the process continuously using gas circulation reactors improves throughput and reduces downtime associated with batch processing, leading to a more reliable supply of critical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the drastic reduction in catalyst consumption and waste generation. Because the catalyst remains stable and active for extended periods due to the CO stabilization effect, the frequency of catalyst replacement is significantly lowered. Additionally, the process eliminates the need for complex workup procedures often required to remove decomposed metal species from the product stream. The ability to separate the product via distillation while retaining the catalyst in the reaction loop for reuse creates a closed-loop system that minimizes raw material loss. This efficiency gain allows for substantial cost savings in the manufacturing of high-purity optically active compounds, making it competitive against less efficient synthetic routes.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reaction conditions. The method tolerates a degree of variability in feedstock quality, such as mixtures of E/Z isomers, without compromising the final optical purity. This flexibility reduces the dependency on ultra-high purity starting materials, which can be a bottleneck in the supply chain. Moreover, the continuous nature of the preferred embodiment means that production can be scaled up or down more responsively to market demand compared to rigid batch processes. The use of standard industrial gases like hydrogen and carbon monoxide, which are readily available globally, further secures the supply chain against raw material shortages.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology offers a cleaner production profile. The high selectivity of the reaction minimizes the formation of byproducts, thereby reducing the load on waste treatment facilities and lowering the environmental footprint of the manufacturing site. The process is designed to be scalable from laboratory benchtop to industrial tonnage, utilizing standard high-pressure reactors and gas handling equipment. The ability to operate without excessive amounts of toxic solvents or additives, and the potential to recycle the solvent medium, aligns with modern green chemistry principles and regulatory requirements for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners and technical stakeholders.
Q: What is the key innovation in patent CN101675020A regarding catalyst stability?
A: The patent introduces a method where the catalyst is pretreated with a CO/H2 mixture, excess CO is removed, and then the reaction proceeds with a strictly controlled low ppm level of CO (100-1200 ppm). This stabilizes the homogeneous catalyst without poisoning the reaction, significantly extending catalyst life.
Q: Which transition metals are preferred for this asymmetric hydrogenation process?
A: The process preferably utilizes transition metals from Group VIII, specifically Rhodium (Rh) and Iridium (Ir), complexed with chiral phosphine ligands such as (R,R)-chiraphos to ensure high enantioselectivity.
Q: Can this method be scaled for industrial production of fragrances?
A: Yes, the patent explicitly describes continuous processing capabilities using gas circulation reactors, making it highly suitable for the commercial scale-up of complex fragrance intermediates like D-citronellal.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Carbonyl Compounds Supplier
The technological potential of patent CN101675020A represents a paradigm shift in the production of chiral aldehydes and ketones, offering a pathway to high-purity intermediates with unmatched efficiency. NINGBO INNO PHARMCHEM stands at the forefront of adopting such advanced synthetic methodologies to serve the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess and residual metal content. We understand that consistency is key in the fragrance and pharmaceutical sectors, and our process controls are designed to deliver batch-after-batch reliability.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of complex chiral intermediates or scale-up of existing routes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure a supply chain that is not only cost-effective but also technologically superior, ensuring your products reach the market with the highest quality standards.
