Advanced Catalytic Cyclization of Citronellal to Isopulegol for Industrial Scale Menthol Production
The global demand for high-purity menthol and its precursors continues to surge across the pharmaceutical and flavor industries, driving the need for more efficient synthetic routes. Patent CN107188781B introduces a groundbreaking methodology for the preparation of isopulegol from citronellal, utilizing a novel class of aryl benzenesulfonic acid aluminum compounds as catalysts. This technology represents a significant leap forward in asymmetric synthesis, addressing long-standing challenges regarding stereoselectivity and process scalability. By employing specifically designed ligands that create optimal steric environments around the catalytic center, this method achieves exceptional enantiomeric excess while operating under remarkably mild reaction conditions. For procurement and technical teams seeking a reliable flavor & fragrance intermediates supplier, understanding the mechanistic advantages of this patent is crucial for securing a stable supply chain of high-value chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
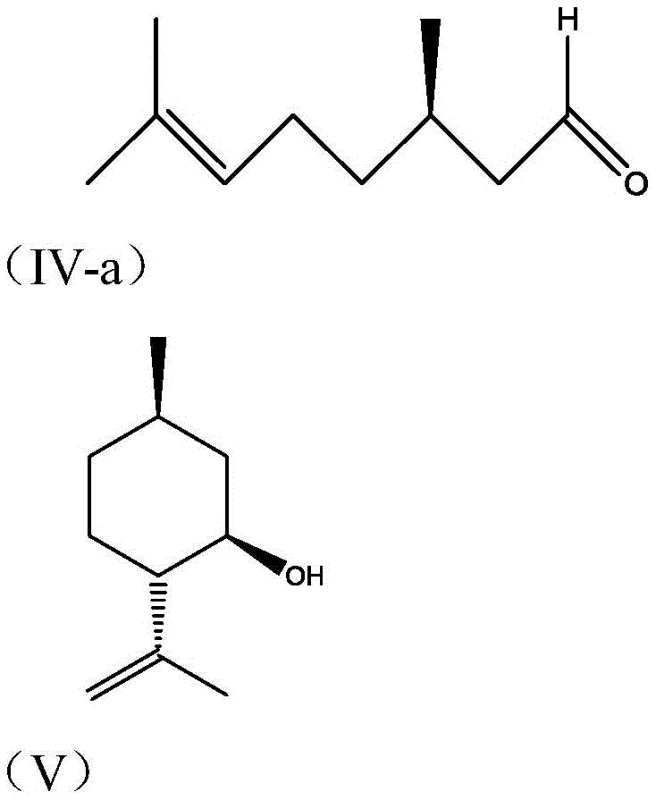
Historically, the industrial synthesis of isopulegol has relied heavily on Lewis acid catalysts such as zinc bromide or unstable aluminum phenolates, each presenting distinct operational bottlenecks. Traditional zinc bromide catalysis, while effective in achieving conversion, suffers from moderate enantioselectivity, typically yielding a ratio of only 94:6 between the desired isopulegol and its unwanted isomers. Furthermore, the corrosive nature of bromide ions necessitates the use of specialized, expensive corrosion-resistant reactor materials, driving up capital expenditure and maintenance costs. Alternative methods using tris(2,6-diphenylphenol)aluminum have demonstrated higher selectivity but fail in practical application due to catalyst instability and the inability to recycle the ligand, leading to prohibitive costs for large-scale operations. These legacy processes often require rigorous low-temperature controls and complex downstream purification to remove metal residues, creating significant inefficiencies in manufacturing throughput.
The Novel Approach
The innovative process described in CN107188781B overcomes these hurdles by introducing aryl benzenesulfonic acid aluminum compounds that combine high activity with exceptional recoverability. This novel catalytic system operates effectively at temperatures ranging from 5°C to 15°C, eliminating the need for energy-intensive cryogenic cooling systems required by older technologies. The core breakthrough lies in the ligand design, where bulky aryl groups provide the necessary steric hindrance to direct the cyclization pathway exclusively towards the desired L-(-)-n-isopulegol isomer. Unlike previous generations of catalysts, this system allows for a simplified workup procedure where the catalyst ligand can be separated into an aqueous phase and recovered with high efficiency. This shift not only enhances the economic viability of the process but also aligns with modern green chemistry principles by reducing waste generation and solvent consumption during the isolation of the final product.
Mechanistic Insights into Aryl Benzenesulfonic Acid Aluminum Catalysis
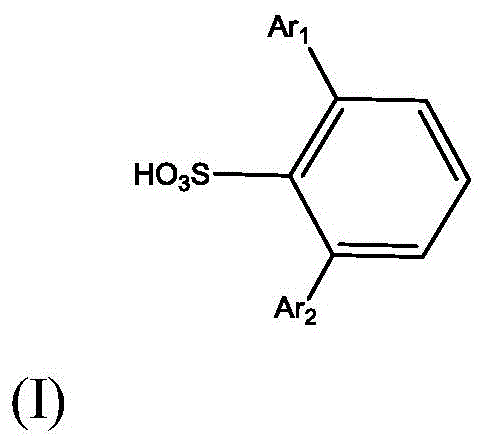
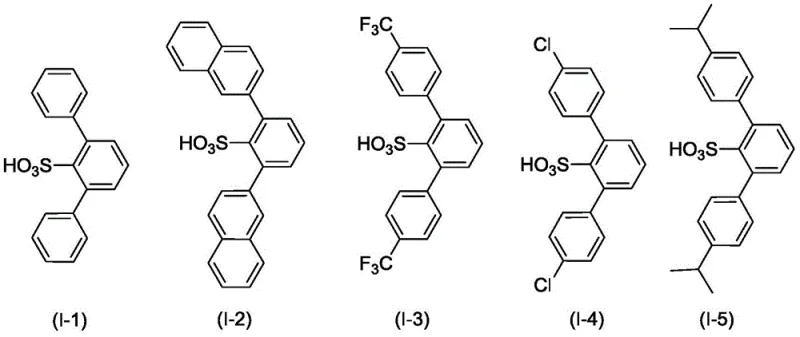
The superior performance of this catalytic system is fundamentally rooted in the precise spatial arrangement of the ligand surrounding the aluminum center. The aryl groups (Ar1 and Ar2) attached to the benzene ring are selected to possess significant steric bulk, such as phenyl, beta-naphthyl, or substituted phenyl groups containing trifluoromethyl or isopropyl moieties. These large substituents create a confined chiral pocket that sterically hinders the approach of the citronellal substrate in unfavorable orientations. Consequently, the reaction transition state is stabilized in a conformation that favors intramolecular cyclization over intermolecular polymerization or the formation of diastereomers like neo-isopulegol. This steric control is critical for achieving the reported enantioselectivity ratios of up to 99.73:0.27, ensuring that the resulting isopulegol meets the stringent purity specifications required for downstream hydrogenation into menthol.
Furthermore, the presence of the sulfonic acid group on the ligand backbone introduces a unique solubility switch that facilitates catalyst management. In its active form, the aluminum complex is soluble in organic solvents like toluene or hexane, allowing for homogeneous catalysis and efficient contact with the substrate. However, upon completion of the reaction, treatment with an aqueous sodium hydroxide solution converts the ligand into its corresponding sodium sulfonate salt. This salt exhibits high water solubility, enabling a clean phase separation where the organic product remains in the upper layer while the catalyst components migrate to the aqueous layer. This mechanism prevents the contamination of the product with aluminum residues and allows for the ligand to be regenerated by acidifying the aqueous phase, thereby closing the loop on catalyst usage and significantly reducing raw material costs over multiple batches.
How to Synthesize Isopulegol Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing isopulegol with minimal operational complexity. The process begins with the in situ generation of the catalyst by reacting the arylbenzenesulfonic acid ligand with an aluminum source such as triethylaluminum or lithium aluminum hydride under an inert atmosphere. Once the catalyst suspension is formed, it is introduced to a solution of citronellal, optionally supplemented with auxiliaries like acetic anhydride or methyl pyruvate to further enhance reaction kinetics and selectivity. The reaction proceeds smoothly at near-ambient temperatures, monitoring conversion via gas chromatography until the desired endpoint is reached. Following the reaction, a straightforward aqueous workup isolates the product, leaving the catalyst ready for recovery.
- Prepare the catalyst by reacting an arylbenzenesulfonic acid ligand (Formula I) with an aluminum compound (Formula II or III) in an inert solvent like toluene under nitrogen protection.
- Add the prepared catalyst solution to a cooled solution of citronellal (R-(+)-citronellal preferred) containing optional auxiliaries like acetic anhydride or methyl pyruvate.
- Maintain the reaction temperature between 5°C and 15°C for 4 to 12 hours, then quench with aqueous NaOH to separate the water-soluble catalyst ligand from the organic product phase.
Commercial Advantages for Procurement and Supply Chain Teams
For organizations focused on cost reduction in flavor & fragrance intermediates manufacturing, this technology offers compelling economic benefits driven by process intensification and waste minimization. The ability to operate at mild temperatures significantly lowers the energy burden associated with refrigeration, translating directly into reduced utility costs per kilogram of product. Moreover, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the load on distillation columns and decreases the loss of valuable material during purification. The simplified catalyst recovery process eliminates the need for complex chromatographic separations or extensive washing steps often required to remove metal contaminants, streamlining the overall production timeline and increasing facility throughput.
- Cost Reduction in Manufacturing: The implementation of this catalytic system removes the dependency on corrosive reagents like zinc bromide, thereby extending the lifespan of standard stainless-steel reactors and reducing maintenance expenditures. The high recovery rate of the ligand means that the effective cost of the catalyst per batch diminishes significantly over time, as the expensive chiral inducer is reused rather than discarded. Additionally, the high conversion rates achieved (often exceeding 90%) ensure that raw material utilization is maximized, preventing the financial loss associated with unreacted starting materials that must be recycled or disposed of.
- Enhanced Supply Chain Reliability: By utilizing a catalyst system that is stable and easy to handle, manufacturers can reduce the risk of batch failures caused by catalyst degradation or sensitivity to moisture. The robustness of the process allows for consistent production schedules, ensuring that downstream customers receive their shipments of isopulegol without delay. The availability of diverse ligand structures (such as those shown in Formula I) provides flexibility in sourcing, as the synthesis of the ligand itself relies on readily available aromatic sulfonic acids and halides, mitigating the risk of supply chain bottlenecks for specialized reagents.
- Scalability and Environmental Compliance: The aqueous workup procedure generates a waste stream that is easier to treat compared to heavy metal-containing effluents from traditional methods. The separation of the catalyst into the water phase allows for centralized treatment and recovery, aligning with increasingly strict environmental regulations regarding metal discharge. This environmental compatibility facilitates smoother regulatory approvals for new manufacturing sites and supports corporate sustainability goals by reducing the overall ecological footprint of the synthesis process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this catalytic technology in industrial settings. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: How does the new arylbenzenesulfonic acid aluminum catalyst compare to traditional zinc bromide catalysts?
A: Unlike zinc bromide, which requires corrosive-resistant equipment and offers lower enantioselectivity (approx. 94/6), the novel aluminum catalyst achieves enantioselectivity ratios up to 99.73:0.27 under milder conditions without severe corrosion risks.
Q: What is the advantage of the ligand recovery process in this method?
A: The sulfonic acid group on the ligand renders its sodium salt water-soluble. This allows for simple phase separation using aqueous NaOH, followed by acidification to precipitate and recover the ligand with high purity, significantly simplifying the workflow compared to non-soluble ligands.
Q: Can this process be scaled for industrial production of menthol intermediates?
A: Yes, the process operates at mild temperatures (5-15°C) rather than extreme cryogenic conditions, reducing energy consumption. The simplified workup and high catalyst stability make it highly suitable for large-scale commercial manufacturing of isopulegol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isopulegol Supplier
As the market for high-purity chiral intermediates expands, partnering with an experienced CDMO is vital for translating patented laboratory methods into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of isopulegol meets the exacting standards required by the global flavor and pharmaceutical industries. We understand the critical nature of supply continuity and have optimized our processes to deliver consistent quality regardless of order volume.
We invite potential partners to engage with our technical procurement team to discuss how this advanced catalytic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this greener, more efficient synthesis route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific production requirements, ensuring a competitive edge in the marketplace.
