Revolutionizing O-Phthalaldehyde Production: Advanced Halogen-Free Synthesis for Global Pharma
The landscape of fine chemical manufacturing is constantly evolving, driven by the relentless demand for higher purity intermediates that can withstand the rigorous scrutiny of modern pharmaceutical regulatory bodies. Patent CN1891680B represents a significant technological leap in the production of o-phthalaldehyde (OPA), a versatile compound critical for applications ranging from biocides to complex pharmaceutical synthesis. This specific intellectual property outlines a robust methodology for preparing high-purity, halogen-free OPA, addressing long-standing industry pain points regarding impurity profiles and storage stability. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this process is essential for securing a competitive edge in the market. The technology described herein not only promises superior product quality but also offers a pathway to more efficient and sustainable manufacturing operations that align with modern green chemistry principles. By leveraging this advanced synthetic route, companies can mitigate the risks associated with traditional production methods that often suffer from inconsistent batch quality and difficult purification challenges.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-phthalaldehyde has been plagued by significant technical hurdles that compromise both yield and purity, creating bottlenecks for downstream applications in the life sciences sector. Prior art methods, such as the ozonolysis of naphthalene described in EP-B-0147593, frequently result in the formation of ester by-products that are notoriously difficult to separate from the target aldehyde, leading to extensive and costly purification workflows. Furthermore, alternative approaches like those disclosed in US 5107032, which involve the hydrolysis of tetrahalo-o-xylene at lower temperatures, often fail to completely eliminate halogen residues, rendering the final product unsuitable for sensitive electronic or pharmaceutical applications. The inherent instability of free OPA also poses a major logistical challenge, as the compound is prone to agglomeration and discoloration upon long-term storage, which can drastically alter its reactivity and dissolution properties. These cumulative deficiencies in conventional manufacturing protocols necessitate additional processing steps, such as recrystallization and decolorization with activated carbon, which erode profit margins and extend lead times for high-purity intermediates. Consequently, the industry has been in urgent need of a process that can deliver consistent, halogen-free quality without relying on inefficient separation techniques.
The Novel Approach
The patented process introduces a transformative three-step strategy that fundamentally alters the production paradigm by utilizing a high-temperature hydrolysis of tetrahalo-o-xylene followed by a protective acetalization sequence. Central to this innovation is the use of specific starting materials, such as the tetrahalo-o-xylene structure depicted below, which serves as the foundational backbone for the subsequent chemical transformations. 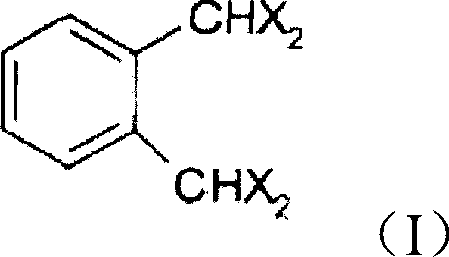 . By conducting the initial hydrolysis at elevated temperatures between 155-160°C and pressures of 2-5 bar, the reaction kinetics are optimized to ensure complete conversion while facilitating the removal of halogen impurities early in the workflow. The subsequent conversion of the crude aldehyde into a dialkoxyphthalane acetal acts as a crucial stabilization mechanism, protecting the reactive aldehyde groups from oxidation and polymerization during isolation and storage. This strategic use of a protected intermediate allows for high-efficiency distillation purification, which is otherwise impossible with the unstable free aldehyde, thereby ensuring that the final deprotected product meets stringent purity specifications exceeding 99.5% GC area. This novel approach effectively bypasses the limitations of prior art by integrating purification into the synthesis flow rather than treating it as a post-reaction remediation step.
. By conducting the initial hydrolysis at elevated temperatures between 155-160°C and pressures of 2-5 bar, the reaction kinetics are optimized to ensure complete conversion while facilitating the removal of halogen impurities early in the workflow. The subsequent conversion of the crude aldehyde into a dialkoxyphthalane acetal acts as a crucial stabilization mechanism, protecting the reactive aldehyde groups from oxidation and polymerization during isolation and storage. This strategic use of a protected intermediate allows for high-efficiency distillation purification, which is otherwise impossible with the unstable free aldehyde, thereby ensuring that the final deprotected product meets stringent purity specifications exceeding 99.5% GC area. This novel approach effectively bypasses the limitations of prior art by integrating purification into the synthesis flow rather than treating it as a post-reaction remediation step.
Mechanistic Insights into High-Temperature Hydrolysis and Acetal Protection
The core of this technological advancement lies in the precise control of reaction conditions during the hydrolysis phase, where the interplay between temperature, pressure, and catalytic activity dictates the overall success of the transformation. Operating at a temperature range of 155-160°C under a pressure of 2-5 bar creates a thermodynamic environment that favors the rapid cleavage of carbon-halogen bonds while minimizing side reactions that could lead to tar formation or over-oxidation. The inclusion of a phase transfer catalyst, such as quaternary ammonium salts, further enhances the reaction rate by facilitating the interaction between the organic tetrahalo-o-xylene substrate and the aqueous alkaline phase, ensuring homogeneous reaction conditions throughout the vessel. This mechanistic efficiency is critical for achieving the high yields reported in the patent data, as it prevents the accumulation of partially hydrolyzed intermediates that could contaminate the final product stream. Moreover, the use of carboxylic acids like acetic acid in the aqueous system helps to buffer the reaction medium, providing a controlled environment that protects the newly formed aldehyde functionality from immediate degradation. For technical teams evaluating cost reduction in fine chemical manufacturing, understanding these kinetic parameters is vital for replicating the process at a commercial scale with consistent results.
Equally important is the mechanism of impurity control achieved through the acetalization and subsequent hydrolysis steps, which serve as a chemical filter for the entire process. By converting the crude OPA into a dialkoxyphthalane derivative in an acidic alcohol solution, the reactive aldehyde groups are masked, rendering the molecule stable enough to undergo rigorous distillation without decomposition. This distillation step is the key to achieving the reported halogen-free quality, as it allows for the physical separation of non-volatile inorganic salts and high-boiling organic impurities that would otherwise persist in the final product. The final deprotection step, conducted via acid hydrolysis at a controlled pH greater than 1.5, is carefully tuned to regenerate the free aldehyde without inducing the polymerization or discoloration typically associated with acidic conditions. This multi-stage purification strategy ensures that the final OPA exhibits a fixed, light color, eliminating the need for post-synthesis decolorization treatments that often result in significant product loss. For R&D Directors focused on purity and impurity profiles, this mechanism offers a robust solution for producing API intermediates that meet the most demanding regulatory standards without compromising on yield.
How to Synthesize High Purity O-Phthalaldehyde Efficiently
Implementing this synthesis route requires a disciplined approach to process engineering, beginning with the careful selection of raw materials and the precise calibration of reactor conditions to match the patent specifications. The initial hydrolysis step demands equipment capable of withstanding elevated pressures and temperatures, along with efficient mixing systems to maintain the efficacy of the phase transfer catalyst throughout the reaction duration. Following the hydrolysis, the extraction and acetalization phases must be managed with strict pH control to ensure optimal conversion rates and to prevent premature decomposition of the acetal intermediate. . Adhering to these standardized operational parameters is essential for realizing the full potential of this technology, as even minor deviations in temperature or acidity can impact the purity profile and color of the final crystalline product. Manufacturers aiming for commercial scale-up of complex aldehydes should prioritize the automation of these control loops to ensure batch-to-batch consistency and operational safety.
- Hydrolyze tetrahalo-o-xylene at 155-160°C and 2-5 bar pressure in the presence of a phase transfer catalyst and base.
- Convert the resulting crude o-phthalaldehyde into a stable dialkoxyphthalane acetal using acidic alcohol solution at 0 to reflux temperature.
- Decompose the purified acetal via acid hydrolysis at pH greater than 1.5 to recover high-purity halogen-free o-phthalaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented manufacturing process offers substantial advantages that extend far beyond simple technical metrics, directly impacting the bottom line and supply chain resilience. The elimination of halogen residues and the inherent stability of the intermediate acetal form significantly reduce the complexity of the purification train, which translates into lower energy consumption and reduced solvent usage per kilogram of finished product. This streamlined workflow inherently supports cost reduction in fine chemical manufacturing by removing the need for expensive adsorbents like Tonsil or activated carbon, which are standard requirements in legacy processes to achieve acceptable color grades. Furthermore, the ability to produce a stable intermediate that can be stored and transported without degradation provides a critical buffer against supply chain disruptions, allowing for larger batch production runs and more flexible inventory management. For Supply Chain Heads concerned with continuity, this means a more reliable source of high-quality material that is less susceptible to the variability often seen in multi-step batch processes involving unstable intermediates. The overall efficiency gains also contribute to a smaller environmental footprint, aligning with the increasing corporate mandates for sustainable sourcing and green chemistry compliance in the global supply network.
- Cost Reduction in Manufacturing: The process architecture fundamentally lowers production costs by integrating purification into the synthesis sequence, thereby removing unit operations dedicated solely to impurity removal and decolorization. By avoiding the use of costly filtration media and reducing the number of recrystallization cycles required to meet purity specs, the overall operational expenditure is significantly optimized without sacrificing product quality. The high yield reported in the patent data indicates efficient atom economy, meaning less raw material is wasted as by-product, which further drives down the variable cost per unit. Additionally, the potential for continuous processing suggested by the shortened reaction times allows for better asset utilization and lower fixed cost allocation per ton of output. These factors combine to create a highly competitive cost structure that can be leveraged in negotiations with downstream pharmaceutical clients seeking value-driven partnerships.
- Enhanced Supply Chain Reliability: The chemical stability of the dialkoxyphthalane intermediate serves as a powerful tool for risk mitigation in the supply chain, as it allows manufacturers to build strategic stockpiles without fear of product degradation over time. Unlike free OPA which requires cold storage and has a limited shelf life, the protected form can be managed with standard warehousing protocols, simplifying logistics and reducing the risk of spoilage during transit. This robustness ensures that delivery schedules can be met consistently, even in the face of fluctuating demand or unexpected production delays, providing buyers with greater certainty in their planning cycles. The use of commercially available starting materials like tetrahalo-o-xylene also reduces dependency on niche precursors, diversifying the supply base and minimizing the risk of raw material shortages. For procurement managers, this translates to a more resilient supply partner capable of maintaining steady flows of critical intermediates essential for uninterrupted drug manufacturing.
- Scalability and Environmental Compliance: The reaction conditions described, particularly the use of aqueous systems and common organic solvents, are well-suited for translation from pilot plant to full-scale commercial production without requiring exotic equipment or hazardous reagents. The high purity of the final product reduces the burden on wastewater treatment facilities, as there are fewer heavy organic contaminants and halogenated waste streams to manage compared to traditional synthesis routes. This alignment with environmental regulations facilitates smoother permitting processes and reduces the long-term liability associated with hazardous waste disposal, making the facility more sustainable in the eyes of regulators and investors. The scalability is further supported by the robust nature of the hydrolysis step, which tolerates variations in feed quality better than sensitive catalytic hydrogenation or ozonolysis processes. Consequently, this technology offers a future-proof platform for expanding capacity to meet growing global demand for high-performance chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced o-phthalaldehyde production technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and technical evaluators. Understanding these details is crucial for making informed decisions about integrating this material into your existing supply chain or development pipeline. . We encourage stakeholders to review these points closely as they highlight the distinct competitive advantages offered by this proprietary manufacturing route over generic market alternatives.
Q: How does this process ensure the product is halogen-free compared to prior art?
A: Unlike previous methods such as US 5107032 which resulted in halogenated byproducts, this process utilizes a specific high-temperature hydrolysis step followed by acetal purification that effectively separates halogen impurities before the final deprotection stage.
Q: What are the storage stability advantages of the intermediate acetal form?
A: The intermediate dialkoxyphthalane acetal is significantly more stable against heat and oxidation than free o-phthalaldehyde, preventing agglomeration and discoloration during long-term storage and transport.
Q: Does this method require expensive decolorization steps like activated carbon?
A: No, the o-phthalaldehyde produced via this patented route exhibits a fixed, light color inherently, eliminating the need for additional decolorization treatments with Tonsil or activated carbon typically required in conventional processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Phthalaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and a commitment to quality excellence. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the CN1891680B process are executed with precision at every scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs designed to validate every batch against the highest international standards, guaranteeing that the halogen-free o-phthalaldehyde you receive is ready for immediate use in sensitive applications. We understand the critical nature of API intermediates in the pharmaceutical value chain and are dedicated to maintaining the integrity of the supply through advanced process control and comprehensive documentation. Our team is prepared to collaborate closely with your technical staff to optimize the integration of this material into your specific manufacturing workflows.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements and cost structures. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this higher-efficiency production method for your supply chain. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your exacting standards for purity and consistency. Let us help you secure a stable, high-quality supply of o-phthalaldehyde that supports your long-term growth and innovation goals in the global marketplace. Contact us today to initiate a dialogue about partnership opportunities and technical collaboration.
