Revolutionizing O-Phthalaldehyde Production: A Technical Analysis of Halogen-Free Synthesis
The chemical industry continuously seeks advancements in the synthesis of critical intermediates that balance high purity with economic viability, and the technology disclosed in patent CN1891680A represents a significant leap forward in the production of o-phthalaldehyde (OPA). This versatile compound serves as a foundational building block across diverse sectors, ranging from pharmaceutical intermediates and agrochemical formulations to specialized polymer additives and photographic materials. Traditional manufacturing routes have long struggled with issues related to halogen contamination, color instability, and complex purification requirements, which often compromise the quality of the final active ingredient. The improved process detailed in this patent addresses these historical bottlenecks by introducing a refined three-step methodology that ensures the production of high-purity, halogen-free OPA without the need for aggressive decolorization agents. For R&D directors and procurement specialists evaluating supply chain partners, understanding the technical nuances of this synthesis route is crucial for assessing the reliability and quality consistency of potential suppliers in the global fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing o-phthalaldehyde have been plagued by inherent chemical inefficiencies that directly impact commercial feasibility and product quality. For instance, the ozonolysis of naphthalene described in EP-B-0147593 generates ester by-products that are notoriously difficult to separate from the target aldehyde, leading to purity profiles that are often insufficient for high-end pharmaceutical applications. Furthermore, alternative hydrolysis methods, such as those disclosed in US 5107032, typically result in products that retain residual halogens, necessitating additional and costly purification stages to meet regulatory standards for halogen-free specifications. The thermal and oxidative instability of OPA further complicates storage and handling, as conventional crude products tend to agglomerate and discolor over time, requiring extensive recrystallization and treatment with activated carbon or Tonsil earth. These downstream processing steps not only increase the overall manufacturing cost but also introduce variability in batch-to-batch quality, creating significant risks for supply chain managers who require consistent material specifications for continuous production lines.
The Novel Approach
The innovative process outlined in patent CN1891680A circumvents these traditional drawbacks through a strategically designed sequence of hydrolysis, protection, and deprotection reactions that prioritize purity and yield. By conducting the initial hydrolysis of tetrahalo-o-xylene at elevated temperatures between 155-160°C and pressures of 2-5 bar, the method ensures complete conversion while utilizing a phase transfer catalyst to enhance reaction kinetics. The subsequent conversion of the crude aldehyde into a dialkoxyphthalane acetal serves as a critical purification handle, allowing for the distillation of the intermediate in a stable form that is resistant to oxidation and agglomeration. This acetal protection strategy effectively isolates the reactive aldehyde functionality from impurities, enabling the removal of halogenated residues before the final regeneration of the OPA. The result is a streamlined workflow that eliminates the need for post-synthesis decolorization, thereby reducing waste generation and simplifying the overall manufacturing footprint while delivering a product with exceptional color stability and chemical integrity.
Mechanistic Insights into High-Temperature Hydrolysis and Acetal Protection
The core of this technological advancement lies in the precise control of reaction conditions during the hydrolysis of the tetrahalo-o-xylene precursor, which dictates the success of the entire synthetic pathway. 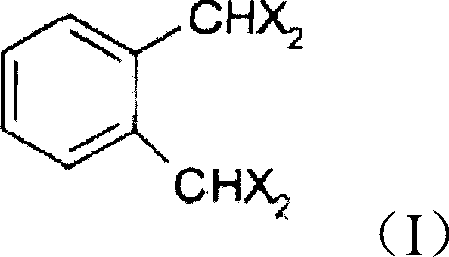 The reaction is performed in an aqueous system comprising C1-C4 carboxylic acids, preferably acetic acid, in the presence of a strong base such as sodium hydroxide. The use of a phase transfer catalyst, such as tetrabutylammonium chloride, is instrumental in facilitating the interaction between the organic halide substrate and the aqueous hydrolysis medium, significantly accelerating the reaction rate without requiring excessive energy input. Maintaining the temperature within the narrow window of 155-160°C is critical; temperatures below this range may lead to incomplete conversion, while higher temperatures risk thermal degradation of the sensitive aldehyde groups. This careful balance ensures that the tetrahalo precursor is efficiently transformed into the corresponding o-phthalaldehyde with minimal formation of side products, setting a high-quality foundation for the subsequent purification steps.
The reaction is performed in an aqueous system comprising C1-C4 carboxylic acids, preferably acetic acid, in the presence of a strong base such as sodium hydroxide. The use of a phase transfer catalyst, such as tetrabutylammonium chloride, is instrumental in facilitating the interaction between the organic halide substrate and the aqueous hydrolysis medium, significantly accelerating the reaction rate without requiring excessive energy input. Maintaining the temperature within the narrow window of 155-160°C is critical; temperatures below this range may lead to incomplete conversion, while higher temperatures risk thermal degradation of the sensitive aldehyde groups. This careful balance ensures that the tetrahalo precursor is efficiently transformed into the corresponding o-phthalaldehyde with minimal formation of side products, setting a high-quality foundation for the subsequent purification steps.
Following hydrolysis, the immediate conversion of the crude OPA into a dialkoxyphthalane acetal is a masterstroke of process chemistry that addresses the inherent instability of the free aldehyde. By reacting the crude mixture with a C1-C4 alcohol under acidic conditions at a pH of 0-3, the reactive aldehyde groups are masked as stable acetals, which can then be purified via distillation. This step is pivotal for impurity control, as it allows for the physical separation of non-volatile salts and halogenated by-products that would otherwise contaminate the final product. The distilled acetal exhibits extremely high halogen-free quality, often exceeding 99.5% GC area purity, before it is even subjected to the final hydrolysis. The final decomposition of the acetal is conducted under mild acidic conditions at a pH greater than 1.5, which gently regenerates the free aldehyde without inducing the polymerization or discoloration commonly seen in harsher acidic environments. This mechanistic approach ensures that the final OPA retains its pristine physical properties, including a fixed color and high solubility, making it ideal for sensitive downstream applications.
How to Synthesize High Purity Halogen-Free O-Phthalaldehyde Efficiently
Implementing this synthesis route requires strict adherence to the patented parameters to replicate the high yields and purity levels demonstrated in the experimental examples. The process begins with the preparation of the hydrolysis mixture, followed by the extraction and acetalization of the crude intermediate, and concludes with the controlled hydrolytic release of the final product. Each stage demands precise monitoring of temperature, pressure, and pH to ensure optimal conversion and minimal by-product formation. For technical teams looking to adopt this methodology, the following standardized steps outline the critical operational parameters derived directly from the patent data to ensure successful scale-up and reproducibility.
- Hydrolyze tetrahalo-o-xylene at 155-160°C and 2-5 bar pressure in the presence of a phase transfer catalyst.
- Convert the resulting crude o-phthalaldehyde into dialkoxyphthalane via acid-catalyzed acetalization in an alcoholic solution.
- Decompose the purified acetal through acid hydrolysis at a pH greater than 1.5 to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic advantages for procurement managers and supply chain heads focused on cost optimization and risk mitigation. The elimination of decolorization steps, which traditionally require expensive adsorbents like activated carbon and generate significant solid waste, translates directly into reduced operational expenditures and a smaller environmental footprint. Furthermore, the ability to produce a halogen-free product without complex multi-stage extractions simplifies the supply chain logistics, reducing the dependency on specialized waste disposal services and lowering the overall cost of goods sold. The enhanced stability of the intermediate acetal also allows for more flexible inventory management, as the protected form can be stored and transported with reduced risk of degradation, ensuring that production schedules are not disrupted by material spoilage. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the rigorous demands of global pharmaceutical and agrochemical manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this process significantly lowers manufacturing costs by removing the need for energy-intensive decolorization and recrystallization steps that are standard in conventional OPA production. By achieving high purity directly through the acetal distillation step, manufacturers can avoid the purchase of costly purification aids and reduce the labor hours associated with multiple filtration and washing cycles. Additionally, the high yield of the acetal intermediate, reported to be greater than 92%, ensures that raw material utilization is maximized, further driving down the unit cost of the final active ingredient. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on margin, providing a tangible financial benefit to downstream buyers seeking to optimize their bill of materials.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain reliability by minimizing the variables that typically cause batch failures or delays in chemical manufacturing. The use of commercially available starting materials, such as tetrahalo-o-xylene, ensures that raw material sourcing is stable and not subject to the volatility of exotic reagents. Moreover, the process's tolerance for continuous operation means that production capacity can be scaled up or adjusted more responsively to market demand fluctuations compared to batch-limited legacy methods. This flexibility is crucial for supply chain heads who must guarantee uninterrupted material flow to production facilities, reducing the risk of stockouts and the associated costs of emergency sourcing or production line stoppages.
- Scalability and Environmental Compliance: Scalability is inherently supported by the shortened reaction times and the simplified workup procedures described in the patent, which facilitate the transition from pilot plant to full commercial scale. The reduction in solvent usage and the elimination of solid waste from decolorization agents align with increasingly stringent environmental regulations, reducing the compliance burden on manufacturing sites. This environmental advantage not only mitigates regulatory risk but also enhances the corporate sustainability profile of the supply chain, a factor that is becoming progressively important for multinational corporations evaluating their vendor networks. The ability to produce high-purity chemicals with a lower environmental impact positions this process as a future-proof solution for long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced o-phthalaldehyde synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and quality assurances provided by this method. Understanding these details is essential for stakeholders evaluating the technical feasibility and commercial value of integrating this material into their existing product portfolios.
Q: How does this process ensure the product is halogen-free?
A: The process utilizes a specific hydrolysis condition followed by acetal purification which effectively separates halogenated by-products, ensuring the final OPA is free from halogen contamination unlike previous methods.
Q: What are the purity levels achievable with this method?
A: The patented methodology consistently yields o-phthalaldehyde with a purity exceeding 99.5% GC area, eliminating the need for extensive decolorization steps typically required in conventional synthesis.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the shortened reaction times and the ability to operate under continuous process conditions make this method highly scalable for industrial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Phthalaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic routes like CN1891680A to deliver superior chemical intermediates to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in large-scale manufacturing environments. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that the halogen-free and high-purity attributes of this process are consistently maintained. We understand that for R&D and procurement leaders, consistency is key, and our infrastructure is designed to provide the reliability and technical support necessary for successful product integration.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain efficiency and product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic ally committed to driving innovation and value in your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
