Optimizing Afatinib Manufacturing: A Novel Convergent Synthesis Strategy for Commercial Scale-Up
Introduction to Advanced Afatinib Synthesis Technologies
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally sustainable production methods. A pivotal development in this domain is detailed in patent CN103254182A, which discloses a novel method for preparing Afatinib, a potent irreversible inhibitor of EGFR and HER2 tyrosine kinases used in the treatment of non-small cell lung cancer. This patent represents a significant departure from legacy synthetic routes, proposing a convergent strategy that merges condensation and cyclization steps to streamline the production of this critical active pharmaceutical ingredient (API). By leveraging the principles of green chemistry and atom economy, this approach addresses the longstanding challenges of low total recovery and complex purification associated with earlier methodologies. For R&D directors and supply chain stakeholders, understanding this technological shift is crucial for optimizing procurement strategies and ensuring long-term supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
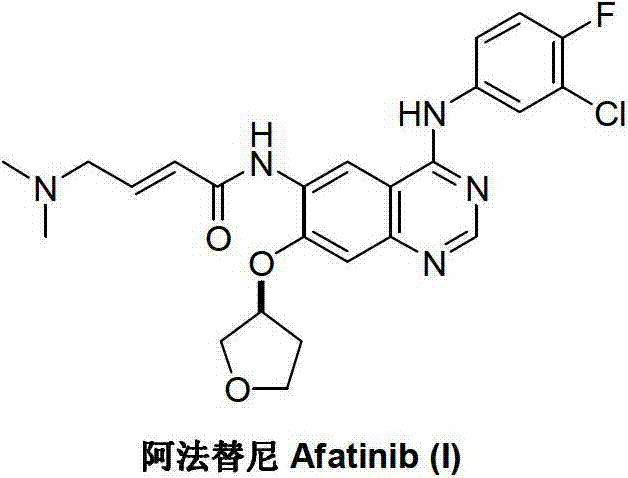
The Limitations of Conventional Methods
Historically, the industrial preparation of Afatinib has relied on the methodology established by Boehringer Ingelheim, as documented in patents such as WO0250043A1 and WO03094921A2. This traditional pathway initiates with a functionalized quinazoline parent nucleus, specifically 4-[(3-chloro-4-fluorophenyl)amino]-6-nitro-7-fluoroquinazoline (IV). The synthesis proceeds through a linear sequence involving nucleophilic substitution with (S)-tetrahydrofuran-3-ol, followed by the reduction of the 6-position nitro group to an amine. Subsequent steps involve amidation with bromocrotonyl chloride and final amination with dimethylamine. This linear approach is inherently inefficient; it requires multiple isolation and purification stages, often relying on column chromatography which is notoriously difficult to scale and economically burdensome in a GMP environment. Furthermore, the functionalization of the pre-formed heterocyclic core often suffers from moderate yields at each step, compounding losses and increasing the cost of goods sold (COGS).
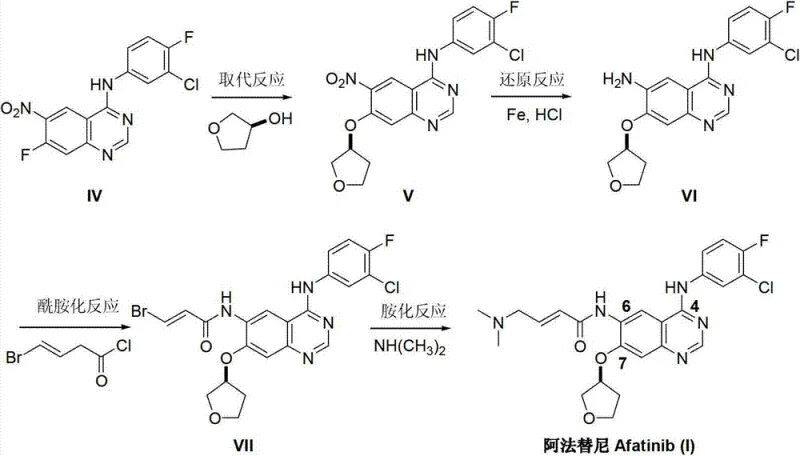
The Novel Approach
In stark contrast, the methodology outlined in CN103254182A introduces a convergent synthesis that fundamentally reimagines the construction of the quinazoline scaffold. Instead of building the molecule around a pre-existing core, this route synthesizes the core de novo through the condensation of 4-fluoro-3-chloroaniline with N,N-dimethylformamide dimethylacetal (DMF-DMA). This reaction generates a Schiff base intermediate (III) which is not isolated but immediately subjected to a cyclization reaction with a highly functionalized aniline derivative (II). This "one-pot" philosophy drastically reduces the number of unit operations. By merging the formation of the Schiff base and the subsequent ring closure, the process eliminates the need for handling unstable intermediates and removes the reliance on chromatographic purification. This shift from a linear, functionalization-heavy pathway to a convergent, condensation-driven pathway offers a clear trajectory for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Aniline Condensation and Cyclization
The core chemical innovation lies in the efficient formation of the quinazoline ring system via the reaction of an ortho-amino nitrile equivalent (generated in situ) with an amidine species. In the first stage, 4-fluoro-3-chloroaniline reacts with DMF-DMA under acidic catalysis (typically acetic acid) to form an enaminone or Schiff base species. This transformation activates the aniline nitrogen for the subsequent nucleophilic attack. The mechanism proceeds through the elimination of methanol, driving the equilibrium forward. The use of toluene as a solvent facilitates the removal of methanol via azeotropic distillation, pushing the reaction to near-completion, as evidenced by the high yields reported in the patent embodiments.
Following the formation of the Schiff base, the addition of the key intermediate, 2-cyano-4-[4-(N,N-dimethylamino)-1-oxo-2-buten-1-yl]amino-5-[(S)-(tetrahydrofuran-3-yl)oxy]aniline, triggers the cyclization. The amino group of this aniline attacks the electrophilic carbon of the Schiff base, followed by intramolecular cyclization involving the nitrile group to close the pyrimidine ring of the quinazoline system. This cascade reaction is highly atom-economical. Crucially, the stereochemistry at the tetrahydrofuran ring is preserved throughout this harsh thermal process (120-130°C), indicating robust process control. The final workup involves simple pH adjustment with ammoniacal liquor to precipitate the product, leveraging the solubility differences between the target API and organic impurities, thereby ensuring high purity without complex downstream processing.
How to Synthesize Afatinib Efficiently
The implementation of this synthesis route requires precise control over reaction temperatures and stoichiometry to maximize the efficiency of the telescoped steps. The process begins with the condensation of the chlorofluoroaniline and DMF-DMA, followed immediately by the introduction of the cyano-aniline partner for ring closure. Detailed standardized operating procedures regarding specific molar ratios, heating ramps, and crystallization parameters are essential for reproducibility.
- Condense 4-fluoro-3-chloroaniline with DMF-DMA in toluene using acetic acid catalyst at 100-110°C to form the Schiff base intermediate.
- Directly react the crude Schiff base with 2-cyano-4-[sidechain]amino-5-[(S)-(tetrahydrofuran-3-yl)oxy]aniline in acetic acid/toluene at 120-130°C.
- Neutralize the reaction mixture with ammoniacal liquor, induce crystallization, and purify via recrystallization to obtain high-purity Afatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthesis route described in CN103254182A presents compelling economic and operational benefits. The primary advantage stems from the drastic simplification of the manufacturing workflow. By eliminating the multi-step functionalization of the quinazoline core and removing the dependency on column chromatography, the process significantly lowers the barrier to entry for large-scale production. This simplification translates directly into reduced capital expenditure (CapEx) for equipment and lower operational expenditure (OpEx) due to decreased solvent consumption and waste generation.
- Cost Reduction in Manufacturing: The novel route utilizes readily available starting materials such as 4-fluoro-3-chloroaniline and DMF-DMA, which are commodity chemicals with stable supply chains. The elimination of expensive transition metal catalysts and the reduction in solvent usage for purification purposes lead to substantial cost savings. Furthermore, the high yield of the initial condensation step (reported up to 97%) minimizes raw material waste, enhancing the overall material throughput and reducing the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: Traditional routes often face bottlenecks due to the complexity of intermediate synthesis and the need for specialized purification services. The convergent nature of this new method decouples the synthesis of the two main fragments, allowing for parallel manufacturing of the aniline intermediate and the Schiff base precursor. This modularity enhances supply chain resilience, reducing lead times for high-purity pharmaceutical intermediates and mitigating the risk of production delays caused by single-point failures in a linear synthesis chain.
- Scalability and Environmental Compliance: The replacement of column chromatography with crystallization is a critical factor for industrial scale-up. Chromatography is difficult to validate and scale beyond pilot plant levels, whereas crystallization is a standard, robust unit operation in the fine chemical industry. Additionally, the process aligns with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product). This environmental compliance simplifies regulatory filings and reduces the costs associated with hazardous waste disposal, making the process more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Afatinib synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: How does this new synthesis route improve upon the traditional Boehringer Ingelheim method?
A: The traditional method relies on functionalizing a pre-formed quinazoline parent nucleus (IV), requiring multiple steps including nitro reduction and halogen substitution which often necessitate column chromatography. The novel route described in CN103254182A constructs the quinazoline ring via condensation of a functionalized aniline, significantly reducing step count and eliminating the need for complex purification techniques.
Q: What are the key yield advantages observed in the experimental embodiments?
A: Experimental data indicates a highly efficient condensation step with yields reaching approximately 97% for the Schiff base intermediate. The subsequent cyclization step achieves yields around 70%, resulting in a robust overall process suitable for industrial application without the losses associated with intermediate isolations.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for industrial scalability. It utilizes common solvents like toluene and acetic acid, avoids expensive transition metal catalysts, and replaces column chromatography with standard crystallization and filtration unit operations, making it ideal for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Afatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of optimized synthetic routes like the one described in CN103254182A for the global oncology market. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific thermal and solvent requirements of this condensation-cyclization protocol, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Afatinib meets the highest international pharmacopoeia standards.
We invite pharmaceutical partners to leverage our technical expertise to further refine this cost-effective manufacturing process. By collaborating with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your supply chain for this critical cancer therapy is both robust and economically optimized.
