Scalable Synthesis of Ruxolitinib Key Intermediate via Nickel-Catalyzed Kumada Coupling
Introduction to Advanced Ruxolitinib Intermediate Manufacturing
The pharmaceutical landscape for Janus Kinase (JAK) inhibitors continues to evolve, driven by the critical demand for efficient and cost-effective synthesis routes for active pharmaceutical ingredients (APIs) like Ruxolitinib. As detailed in the recent patent disclosure CN116082382A, a significant technological breakthrough has been achieved in the preparation of the key intermediate, 4-(1H-pyrazol-4-yl)-7-((2-(trimethylsilyl)ethoxy)methyl)-7H-pyrrolo[2,3-d]pyrimidine. This innovation addresses long-standing bottlenecks in the supply chain of JAK inhibitors, which are vital for treating myelofibrosis and polycythemia vera. The patent outlines a novel synthetic strategy that replaces traditional, cost-prohibitive methods with a streamlined, nickel-catalyzed approach. For global procurement leaders and R&D directors, this represents a pivotal shift towards more sustainable and economically viable manufacturing protocols. By leveraging a Kumada coupling reaction, the new method circumvents the reliance on expensive palladium catalysts, thereby unlocking substantial potential for cost reduction in API manufacturing while maintaining rigorous purity standards required for regulatory compliance.
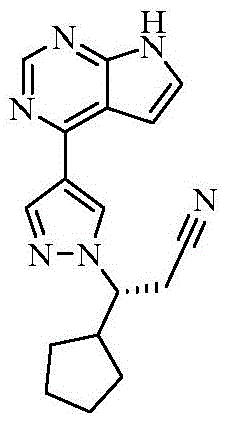
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ruxolitinib intermediates has relied heavily on Suzuki-Miyaura coupling reactions, a standard in cross-coupling chemistry but one that presents distinct disadvantages for large-scale industrial application. As illustrated in prior art routes such as those reported by Incyte Corporation (CN102348693B), the conventional pathway necessitates the use of palladium-based catalysts. Palladium is a precious metal with highly volatile market pricing, introducing significant financial uncertainty and cost instability into the production budget. Furthermore, the removal of residual palladium from the final API is a stringent regulatory requirement, often demanding additional purification steps involving specialized scavengers or chromatography, which further erodes yield and increases processing time. These traditional routes also frequently involve harsh reaction conditions or complex protection-deprotection sequences that complicate the operational workflow, making them less ideal for the high-volume production needed to meet global market demand for JAK inhibitors.
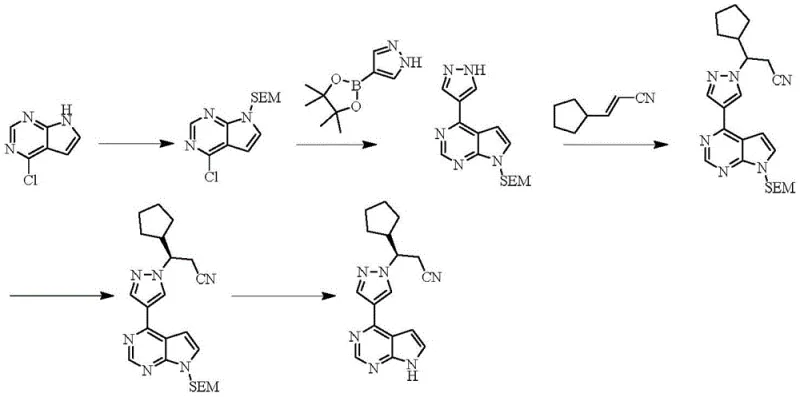
The Novel Approach
The methodology disclosed in patent CN116082382A introduces a paradigm shift by employing a Kumada cross-coupling reaction mediated by nickel catalysts. This approach fundamentally alters the economic and operational dynamics of the synthesis. Instead of palladium, the process utilizes nickel complexes such as NiCl2(dppf), which are significantly more abundant and cost-effective. The reaction proceeds under remarkably mild conditions, typically between 20°C and 60°C, which reduces energy consumption and enhances safety profiles by minimizing thermal hazards. Moreover, the new route integrates a Grignard exchange reaction to generate the necessary organometallic species in situ, avoiding the direct handling of unstable Grignard reagents. This one-pot capability simplifies the unit operations, reducing the number of isolation steps and solvent exchanges. The result is a concise, environmentally friendlier pathway that delivers high yields and superior purity, directly addressing the scalability issues inherent in the older Suzuki-based methodologies.
Mechanistic Insights into Nickel-Catalyzed Kumada Coupling
The core of this technological advancement lies in the precise execution of the Kumada coupling between the pyrazole-derived Grignard reagent (Formula 3) and the chloropyrrolopyrimidine electrophile (Formula 4). The mechanism initiates with a Grignard exchange reaction where a halogenated pyrazole precursor (Formula 2) reacts with an alkyl magnesium halide, such as isopropyl magnesium chloride-lithium chloride complex. This exchange is carefully controlled at low temperatures (-10°C to 0°C) to ensure the formation of the reactive organomagnesium species without inducing side reactions or decomposition. Once formed, this nucleophile engages with the electron-deficient chloropyrrolopyrimidine in the presence of the nickel catalyst. The nickel center facilitates the oxidative addition of the carbon-chlorine bond, followed by transmetallation with the Grignard reagent and subsequent reductive elimination to forge the critical carbon-carbon bond between the pyrazole and pyrrolopyrimidine rings. This catalytic cycle is highly efficient, allowing for the use of lower catalyst loadings (molar ratio of substrate to catalyst from 10:1 to 20:1) while maintaining robust conversion rates.
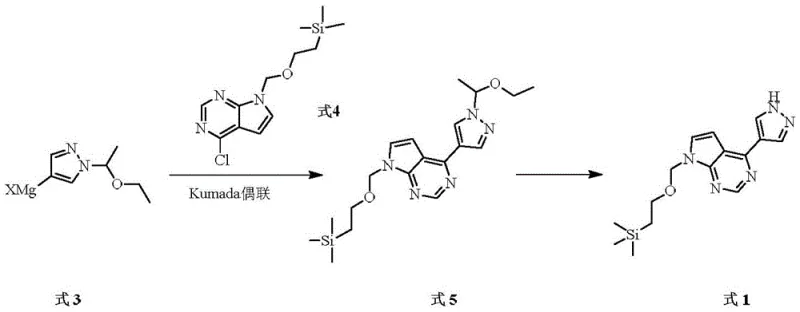
Following the coupling, the process incorporates a highly selective deprotection strategy to reveal the final active intermediate (Formula 1). The intermediate (Formula 5) contains an ethoxyethyl protecting group on the pyrazole nitrogen, which was strategically installed to prevent unwanted N-alkylation during the coupling phase. The patent demonstrates that this group can be cleanly removed using common mineral acids like hydrochloric acid, sulfuric acid, or phosphoric acid at moderate temperatures (10°C to 45°C). This selectivity is crucial; it ensures that the trimethylsilylethoxymethyl (SEM) group on the pyrrolopyrimidine nitrogen remains intact, preserving the functionality needed for downstream API synthesis. The ability to differentiate between these protecting groups under mild acidic conditions highlights the chemical robustness of the new route, minimizing the formation of des-SEM impurities or other degradation products that could compromise the quality of the final pharmaceutical product.
How to Synthesize Ruxolitinib Key Intermediate Efficiently
The synthesis protocol described in the patent offers a practical blueprint for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The procedure begins with the generation of the Grignard reagent in tetrahydrofuran, followed immediately by the addition of the nickel catalyst and the electrophilic partner, creating a seamless one-pot sequence that minimizes exposure to air and moisture. This integration reduces the overall cycle time and solvent usage, aligning with green chemistry principles. The workup involves standard extraction and washing procedures, followed by a straightforward acid-mediated deprotection. For detailed operational parameters, stoichiometry, and specific temperature ramps required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform a Grignard exchange reaction on the pyrazole precursor (Formula 2) using alkyl magnesium halide to generate the organomagnesium species (Formula 3).
- Execute a Kumada coupling reaction between the generated Formula 3 and the chloropyrrolopyrimidine derivative (Formula 4) using a nickel-based catalyst like NiCl2(dppf) to yield the protected intermediate (Formula 5).
- Conduct a selective deprotection of Formula 5 using an acid such as hydrochloric acid or sulfuric acid to remove the ethoxyethyl group, yielding the final target compound (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed route offers compelling strategic advantages beyond mere technical feasibility. The transition from palladium to nickel catalysts represents a direct intervention in the cost structure of the intermediate, decoupling production expenses from the volatile precious metals market. This stability allows for more accurate long-term budgeting and pricing contracts with API manufacturers. Furthermore, the simplified process flow, characterized by fewer unit operations and milder conditions, translates into reduced utility costs and lower capital expenditure requirements for reactor infrastructure. The elimination of complex heavy metal scavenging steps not only saves on material costs but also accelerates the batch release timeline, enhancing overall throughput. These factors collectively contribute to a more resilient supply chain capable of withstanding market fluctuations and meeting the surging demand for JAK inhibitor therapies globally.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with economical nickel-based alternatives drastically lowers the direct material cost per kilogram of the intermediate. Additionally, the high efficiency of the Kumada coupling reduces the need for excessive reagent equivalents, further optimizing the bill of materials. By avoiding the costly and time-consuming processes associated with removing trace palladium residues, manufacturers can achieve significant operational savings. This economic efficiency makes the process highly competitive for generic API production, where margin compression is a constant challenge, ensuring that the final drug product remains accessible while maintaining profitability for the supply chain partners.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including the nickel catalysts and alkyl magnesium halides, are commodity chemicals with robust global availability, unlike specialized palladium ligands which can face supply constraints. The mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity issues, leading to more predictable production schedules. This reliability is critical for maintaining continuous inventory levels for downstream API synthesis, preventing stockouts that could disrupt the manufacturing of life-saving medications. The process's tolerance to standard industrial solvents like tetrahydrofuran and isopropyl acetate further simplifies logistics and procurement, ensuring a steady flow of inputs for large-scale campaigns.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing a one-pot strategy that minimizes solvent transfers and intermediate isolations, thereby reducing waste generation. The use of nickel, a less toxic metal compared to palladium, simplifies wastewater treatment and effluent management, aiding in compliance with increasingly stringent environmental regulations. The high atom economy of the coupling reaction and the ability to recycle solvents contribute to a lower environmental footprint. This alignment with green chemistry metrics not only satisfies corporate sustainability goals but also mitigates regulatory risks associated with hazardous waste disposal, making the facility a more attractive partner for environmentally conscious pharmaceutical companies seeking long-term manufacturing alliances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. They are derived from the specific experimental data and advantageous effects reported in the patent literature, providing clarity on how this technology translates to real-world manufacturing scenarios. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this new supply source.
Q: Why is the Kumada coupling preferred over Suzuki coupling for this intermediate?
A: The Kumada coupling utilizes inexpensive nickel-based catalysts instead of costly palladium catalysts required in Suzuki coupling. This significantly reduces raw material costs and eliminates the need for complex heavy metal removal processes, making it more suitable for industrial mass production.
Q: What are the reaction conditions for the deprotection step?
A: The deprotection is carried out under mild acidic conditions, preferably using hydrochloric acid, sulfuric acid, or phosphoric acid at temperatures between 10°C and 45°C. This ensures selective removal of the ethoxyethyl group without damaging the sensitive heterocyclic core.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process is designed for scalability. It features mild reaction temperatures (20-60°C), uses readily available solvents like tetrahydrofuran, and avoids hazardous exothermic risks associated with traditional Grignard preparations by using a controlled exchange method.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruxolitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed Kumada coupling technology for the global supply of Ruxolitinib intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to rapidly adopt and optimize such advanced synthetic routes. Our facilities are equipped to handle complex heterocyclic chemistry with precision, ensuring that the transition from laboratory scale to commercial production is seamless and efficient. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and reliability. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis method for your API projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this critical intermediate, ensuring your production timelines are met without compromising on quality or cost efficiency.
