Revolutionizing OLED Material Production: One-Step Synthesis of Tricarbazole Derivatives
The landscape of organic optoelectronic materials is constantly evolving, driven by the demand for higher efficiency and purer emissive layers in Organic Light-Emitting Diodes (OLEDs). A pivotal advancement in this domain is documented in patent CN103421012A, which discloses a novel, one-step method for preparing tricarbazole materials directly from aryl indolinone precursors. This technology represents a significant departure from conventional multi-step syntheses, offering a streamlined pathway to highly conjugated, three-fold symmetric molecules that serve as excellent hole transport or blue-light emitting materials. By utilizing phosphorus oxychloride (POCl3) as both a dehydrating and cyclizing agent in a toluene medium, this process eliminates the need for intermediate isolation and complex coupling reactions. For R&D directors and procurement strategists, understanding this shift is crucial, as it directly impacts the cost structure and scalability of producing high-performance display and lighting components. The ability to introduce diverse aryl substituents—such as phenyl, tert-butylphenyl, and fluorenyl groups—directly at the precursor stage allows for precise tuning of electronic properties without the burden of extensive downstream modification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tricarbazole cores has been plagued by inefficiency and structural rigidity. Early methodologies, such as those reported in 1986, relied on the acid-catalyzed coupling of indole and 3-bromoindole, followed by oxidative dehydrogenation using trifluoroacetic acid. As illustrated in the reaction scheme below, this pathway is fraught with complications, including numerous side reactions and difficult separation processes, resulting in dismal yields of merely 23%. Furthermore, alternative approaches involving the direct bromination of indole to form hexabromotricarbazole, while simpler, offer limited versatility for introducing functional aryl groups necessary for advanced OLED applications. These traditional routes often necessitate harsh conditions and generate significant chemical waste, creating bottlenecks for supply chain managers who require consistent, high-volume production. The inability to easily incorporate bulky or electron-rich aryl substituents in these early stages severely restricts the molecular engineering potential required for next-generation optoelectronic devices.
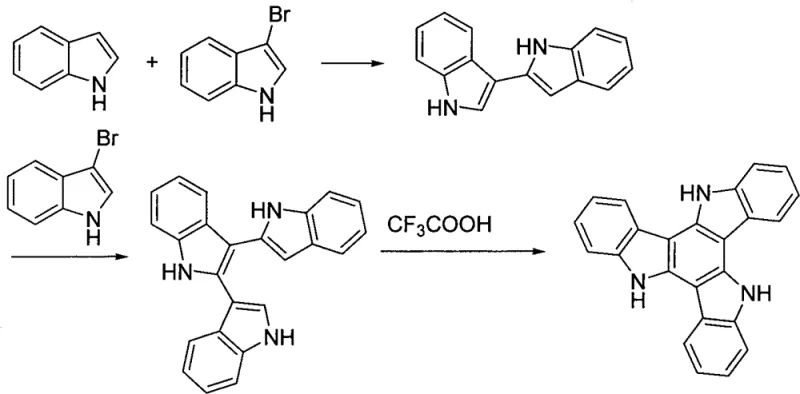
The Novel Approach
In stark contrast, the methodology outlined in CN103421012A utilizes aryl indolinone as the starting material, enabling a direct condensation reaction that constructs the tricarbazole skeleton in a single operational step. This approach fundamentally alters the economic and technical feasibility of manufacturing these compounds. By dissolving the aryl indolinone in toluene and treating it with POCl3 under reflux conditions, the reaction proceeds through a concerted cyclization and dehydration mechanism. This not only simplifies the workflow but also dramatically expands the scope of accessible derivatives. Unlike previous methods restricted to small alkyl or halogen groups, this novel route successfully incorporates substantial aryl branches like fluorenyl and triphenylamino groups directly into the core structure. The visual representation of this transformation highlights the elegance of the design, where three indolinone units converge to form the central benzene ring of the tricarbazole system, preserving the substituents introduced at the indolinone stage.
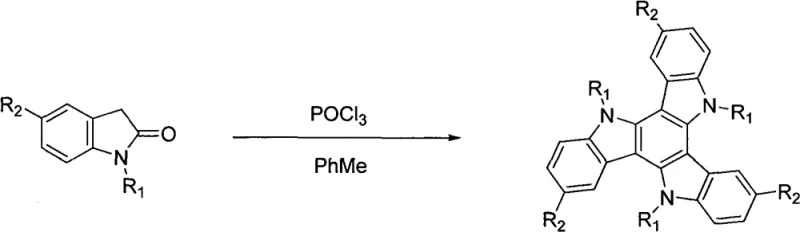
Mechanistic Insights into POCl3-Mediated Cyclization
The core of this technological breakthrough lies in the dual role of phosphorus oxychloride (POCl3) acting as a potent dehydrating agent and an activator for electrophilic aromatic substitution. In the reaction medium, POCl3 facilitates the activation of the carbonyl group on the indolinone, promoting the formation of reactive intermediates that undergo intermolecular cyclization. The mechanism likely involves the initial formation of a Vilsmeier-Haack type intermediate or a similar activated species, which then attacks the electron-rich positions of adjacent indolinone molecules. This cascade reaction effectively stitches three molecular units together while eliminating oxygen atoms as water (sequestered by POCl3), driving the equilibrium towards the formation of the fully aromatic tricarbazole system. For process chemists, understanding this mechanism is vital for optimizing reaction parameters such as temperature (100-110°C) and duration (10-30 hours) to maximize conversion while minimizing the formation of oligomeric byproducts. The robustness of this mechanism allows it to tolerate a wide variety of electronic environments on the aryl rings, ensuring consistent performance across different derivative batches.
Furthermore, the impurity profile of this reaction is inherently cleaner than multi-step alternatives due to the absence of intermediate isolation and purification steps. In traditional syntheses, each isolation step introduces opportunities for product loss and contamination with residual solvents or catalysts. Here, the reaction mixture is simply quenched into ice water and neutralized with sodium carbonate, followed by standard extraction and chromatography. This simplicity suggests that on a commercial scale, the purification train could be significantly shortened, potentially replacing column chromatography with crystallization or continuous extraction techniques. The high stability of the resulting tricarbazole molecule ensures that once formed, it resists degradation during workup, contributing to the observed yields of 28.6% to 40.5%. This mechanistic efficiency translates directly into a more predictable and controllable manufacturing process, a key metric for quality assurance teams in the fine chemical sector.
How to Synthesize Tricarbazole Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and thermal management of the POCl3 addition. The standard protocol involves dissolving the specific aryl indolinone precursor in toluene at a concentration of roughly 10 mmol per 10 mL, followed by the addition of an equimolar volume of POCl3. The mixture is then heated to reflux, maintaining a temperature between 100°C and 110°C for a period ranging from 10 to 30 hours, depending on the steric bulk of the substituents. Upon completion, the reaction is carefully quenched into ice water to hydrolyze excess POCl3, and the pH is adjusted to neutral using sodium carbonate before extraction with dichloromethane. The detailed standardized synthesis steps for scaling this process are provided in the guide below.
- Dissolve the specific aryl indolinone precursor in toluene solvent within a reaction vessel equipped with stirring capabilities.
- Add an equal volume of phosphorus oxychloride (POCl3) to the solution and initiate heating to reflux conditions between 100-110°C.
- Maintain the reaction for 10 to 30 hours, then quench into ice water, neutralize with sodium carbonate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-step synthesis offers profound strategic benefits beyond mere technical novelty. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By collapsing what was previously a multi-step sequence involving coupling, oxidation, and purification into a single reactor operation, manufacturers can significantly lower labor costs, energy consumption, and solvent usage. This reduction in process complexity also minimizes the risk of batch-to-batch variability, ensuring a more reliable supply of critical OLED intermediates. Furthermore, the ability to source diverse aryl indolinones allows for a flexible supply chain strategy where different grades of tricarbazole can be produced on demand without retooling the entire synthesis line, enhancing responsiveness to market fluctuations in the display industry.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the reduction in total reaction time lead to substantial cost savings in utilities and manpower. Since the process avoids the use of expensive transition metal catalysts often required for cross-coupling modifications in traditional routes, the raw material costs are further optimized. The use of common solvents like toluene and readily available reagents like POCl3 ensures that the input costs remain stable and predictable, shielding the production budget from the volatility associated with precious metal catalysts.
- Enhanced Supply Chain Reliability: The reliance on aryl indolinone precursors, which are generally stable and commercially accessible, mitigates the risk of supply disruptions. Unlike complex brominated intermediates that may have limited suppliers, aryl indolinones can be sourced from a broader vendor base or synthesized via established nitro-reduction pathways. This diversification of the raw material base strengthens the supply chain resilience, ensuring that production schedules for high-purity OLED materials are maintained even when specific niche reagents face global shortages.
- Scalability and Environmental Compliance: The straightforward nature of the reaction—simply heating a mixture under reflux—makes it highly amenable to scale-up from kilogram to tonne quantities without requiring specialized high-pressure or cryogenic equipment. Additionally, the simplified workup reduces the volume of aqueous and organic waste streams generated per kilogram of product. This aligns with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities and lowering the overall environmental footprint of the manufacturing site, which is a critical factor for long-term operational licensing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tricarbazole synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their existing product lines or R&D pipelines.
Q: What is the primary advantage of the aryl indolinone route over traditional indole coupling?
A: The aryl indolinone route allows for the direct introduction of diverse aryl substituents in a single step, whereas traditional methods require complex multi-step coupling or post-synthesis modification, significantly reducing process time and impurity profiles.
Q: What yields can be expected from this one-step cyclization process?
A: According to patent data, the process achieves yields ranging from approximately 28% to 40% depending on the specific aryl substituent, which is competitive given the significant reduction in synthetic steps compared to prior art methods that yielded only 23%.
Q: Can this method accommodate bulky substituents for OLED applications?
A: Yes, the method successfully accommodates bulky groups such as fluorenyl and triphenylamino moieties, which are critical for tuning the photophysical properties of OLED materials, overcoming the limitations of earlier methods restricted to small alkyl or halogen groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the one-step aryl indolinone cyclization route for the next generation of OLED materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to mass market deployment is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade chemicals, guaranteeing that every batch of tricarbazole derivative performs consistently in your final device architecture. We understand that in the fast-paced world of optoelectronics, time-to-market is critical, and our optimized processes are designed to accelerate your development cycles.
We invite you to collaborate with our technical team to explore how this efficient synthesis can enhance your product portfolio. Whether you require custom substitution patterns to tune emission wavelengths or need to secure a stable supply of core intermediates, we are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you engineer a more competitive and sustainable supply chain for your high-performance materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
