Advanced Catalytic Synthesis of Chiral 2-Azabicyclo[2.2.1]Heptane Derivatives for Commercial Scale-Up
Advanced Catalytic Synthesis of Chiral 2-Azabicyclo[2.2.1]Heptane Derivatives for Commercial Scale-Up
The pharmaceutical industry constantly seeks robust, scalable pathways for complex chiral intermediates, particularly those serving as cores for antiviral agents and adenosine agonists. Patent CN1385422A presents a groundbreaking methodology for the preparation of 2-azadihydroxybicyclo[2.2.1]heptane compounds, addressing critical bottlenecks in stereochemical control and process efficiency. This technology leverages a sophisticated sequence involving catalytic asymmetric dihydroxylation followed by a novel resolution strategy using L-tartaric acid, ultimately delivering lactam intermediates with exceptional enantiomeric purity. For R&D directors and procurement specialists, this patent represents a pivotal shift away from inefficient enzymatic resolutions toward a chemically driven, highly controllable synthesis that ensures supply chain stability and cost predictability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral bicyclic lactams relied heavily on enzymatic resolution or stoichiometric oxidation methods, both of which present significant drawbacks for large-scale manufacturing. Enzymatic processes, while selective, often suffer from narrow substrate specificity, requiring extensive optimization for each new derivative and posing risks of batch-to-batch variability due to biological catalyst sensitivity. Furthermore, traditional stoichiometric oxidations utilizing heavy metals in large quantities generate substantial toxic waste, complicating environmental compliance and driving up disposal costs. Previous attempts to resolve diastereomeric mixtures often resulted in poor yields or required cumbersome chromatographic separations that are economically unfeasible for multi-kilogram production runs, creating a barrier to reliable commercial supply.
The Novel Approach
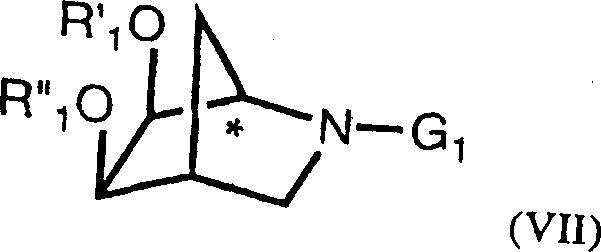
The methodology disclosed in CN1385422A overcomes these hurdles by introducing a catalytic dihydroxylation protocol that operates with remarkably low metal loading, specifically utilizing osmium tetroxide at concentrations as low as 0.06mol%. This approach is coupled with a chemical resolution step using L-tartaric acid in an aqueous-isopropanol system, which allows for the efficient isolation of the desired (1R) diastereomer through crystallization rather than chromatography. By integrating a subsequent ruthenium-catalyzed oxidation step, the process achieves enantiomeric excess values greater than 95%, ensuring high-quality output suitable for stringent pharmaceutical applications. This chemical logic provides a deterministic pathway that is far more robust and scalable than biological alternatives.
![General formula for the dihydroxylation of bicyclo[2.2.1]heptane compounds showing chiral centers](/insights/img/2-azabicyclo-heptane-synthesis-pharma-supplier-20260307064139-02.webp)
Mechanistic Insights into Osmium-Catalyzed Dihydroxylation and Ruthenium Oxidation
The core of this synthetic strategy lies in the precise control of stereochemistry during the functionalization of the bicyclic alkene. The dihydroxylation reaction proceeds via a concerted [3+2] cycloaddition mechanism where the osmium tetroxide species coordinates with the olefinic bond. The use of a co-oxidant such as N-methylmorpholine oxide (NMO) is critical, as it regenerates the active Os(VIII) species from the reduced Os(VI) osmate ester, allowing the reaction to proceed catalytically. The patent highlights that maintaining the osmium loading between 0.06mol% and 0.1mol% is sufficient to drive the reaction to completion while minimizing residual metal contamination, a key concern for downstream processing. The solvent system, typically a mixture of water, acetone, and ethers like tert-butyl methyl ether, is optimized to balance solubility and reaction rate, ensuring the exo-selectivity required for the target stereochemistry.
Following the dihydroxylation and resolution, the transformation of the protected diol into the corresponding lactam is achieved through a ruthenium-catalyzed oxidation. This step utilizes ruthenium dioxide (RuO2) or its hydrate at loadings of approximately 0.1mol% to 1mol% in the presence of a stoichiometric oxidant such as sodium periodate or sodium bromate. The mechanism involves the generation of volatile ruthenium tetroxide in situ, which selectively oxidizes the secondary alcohol or amine functionality to the lactam carbonyl without affecting the acid-sensitive acetal protecting groups. This chemoselectivity is paramount, as it preserves the integrity of the chiral centers established in earlier steps. The result is a lactam compound with an enantiomeric excess of greater than or equal to 95%, demonstrating the high fidelity of the catalytic cycle in preserving optical purity throughout the synthesis.
How to Synthesize 2-Azabicyclo[2.2.1]Heptane Derivatives Efficiently
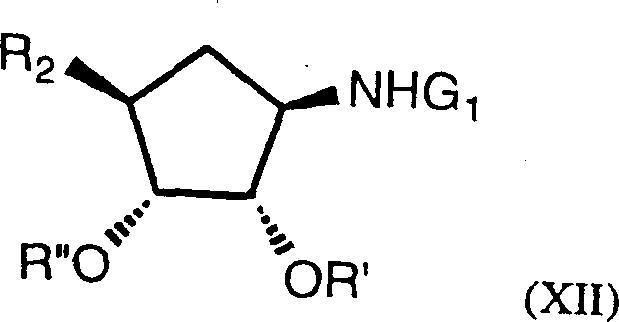
The synthesis of these high-value intermediates requires strict adherence to the optimized conditions regarding temperature, solvent ratios, and catalyst loading to ensure maximum yield and purity. The process begins with the preparation of the bicyclic alkene precursor, followed by the critical dihydroxylation step where temperature control is vital to prevent over-oxidation or side reactions. Subsequent resolution via salt formation demands precise control of the water-to-isopropanol ratio to maximize the crystallization efficiency of the L-tartrate salt. For a comprehensive understanding of the operational parameters, including specific workup procedures and purification techniques, please refer to the standardized synthesis guide below.
- Perform asymmetric dihydroxylation on the bicyclo[2.2.1]heptene precursor using catalytic osmium tetroxide (0.06mol% to 0.1mol%) and N-methylmorpholine oxide in a water-acetone solvent system.
- Resolve the resulting diastereomeric mixture by forming the L-tartrate salt in an aqueous-isopropanol mixture to isolate the desired (1R) diastereomer with high optical purity.
- Protect the dihydroxy moiety via acetalization using 2,2-dimethoxypropane and TFA, followed by oxidation with catalytic RuO2 to yield the high-ee lactam compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative benefits in terms of cost structure and supply reliability. The drastic reduction in precious metal catalyst loading directly translates to lower raw material costs, as osmium and ruthenium are among the most expensive reagents in organic synthesis. By operating at sub-stoichiometric levels (0.06mol% for Os and <1mol% for Ru), the process minimizes the financial exposure to volatile metal markets. Furthermore, the reliance on crystallization for purification instead of preparative chromatography significantly reduces solvent consumption and processing time, leading to substantial cost savings in manufacturing overheads. This efficiency makes the production of these complex intermediates economically viable even at large scales.
- Cost Reduction in Manufacturing: The implementation of catalytic rather than stoichiometric oxidation methods eliminates the need for massive quantities of expensive oxidants and metals. This shift not only lowers the direct bill of materials but also simplifies the waste treatment process, as the volume of heavy metal-contaminated waste is significantly diminished. The ability to recover and recycle solvents like isopropanol and acetone further enhances the economic profile, ensuring that the cost reduction in pharmaceutical intermediate manufacturing is sustained over long production campaigns without compromising quality.
- Enhanced Supply Chain Reliability: Unlike enzymatic processes that depend on the availability and stability of biological cultures, this purely chemical synthesis relies on stable, commercially available reagents. This independence from biological variables ensures consistent batch-to-batch performance and reduces the risk of supply disruptions caused by fermentation failures or enzyme deactivation. The robustness of the crystallization-based purification also means that the process is less sensitive to minor fluctuations in reaction conditions, providing a reliable source of high-purity pharmaceutical intermediates that can meet tight delivery schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common industrial solvents and standard reactor configurations. The low toxicity profile resulting from minimal heavy metal usage simplifies regulatory compliance and environmental permitting, facilitating faster technology transfer from pilot plant to commercial production. This alignment with green chemistry principles not only reduces the environmental footprint but also future-proofs the supply chain against increasingly stringent global regulations on hazardous waste discharge and residual solvent limits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: What is the primary advantage of the osmium loading described in this patent?
A: The patent specifies an ultra-low osmium tetroxide loading of approximately 0.06mol% to 0.1mol%, which drastically reduces the cost of goods and simplifies the removal of toxic heavy metal residues compared to stoichiometric methods.
Q: How is high enantiomeric excess achieved in the lactam formation?
A: High enantiomeric excess (greater than 95% ee) is achieved through a combination of stereoselective dihydroxylation, diastereomeric resolution via L-tartaric acid crystallization, and a highly selective ruthenium-catalyzed oxidation step.
Q: Can this process be scaled for industrial API production?
A: Yes, the process utilizes robust crystallization steps for purification rather than chromatography, and employs catalytic amounts of expensive metals, making it highly suitable for commercial scale-up in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Azabicyclo[2.2.1]Heptane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated catalytic methodologies described in CN1385422A can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-azabicyclo[2.2.1]heptane derivative meets the exacting standards required for API synthesis. Our commitment to quality assurance ensures that our clients receive materials that facilitate smooth downstream processing and regulatory approval.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this catalytic route can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your project timelines and secure a competitive advantage in the marketplace.
