Optimizing Tedizolid Production: A Technical Analysis of Novel Catalytic Routes for Commercial Scale-Up
Introduction to Advanced Tedizolid Synthesis
The global demand for next-generation oxazolidinone antibiotics continues to rise, driven by the urgent need to combat resistant Gram-positive bacterial infections. Patent CN108368100B introduces a transformative preparation method for Tedizolid and its key phosphate prodrug, addressing critical bottlenecks in existing manufacturing protocols. This intellectual property outlines a robust synthetic strategy that replaces costly and hazardous reagents with more accessible catalytic systems, specifically leveraging copper-mediated coupling reactions. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and economically viable production of high-purity pharmaceutical intermediates. The technology focuses on optimizing the construction of the biaryl core and its subsequent coupling with the oxazolidinone moiety, ensuring that the final active pharmaceutical ingredient meets rigorous quality specifications while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tedizolid has been plagued by complex multi-step sequences that rely on expensive transition metals and extreme reaction conditions. Early routes, such as those disclosed by Dong-A Pharmaceutical, utilized silver trifluoroacetate for iodination and required multiple palladium-catalyzed steps, leading to inflated production costs and difficult scale-up due to heavy metal contamination risks. Furthermore, alternative approaches developed by Trius Therapeutics necessitated the use of n-butyllithium at cryogenic temperatures of -65°C, imposing severe safety hazards and requiring specialized infrastructure that many contract manufacturing organizations lack. These legacy methods often suffered from low total yields and generated significant waste streams, making them less attractive for large-scale commercial manufacturing in a cost-sensitive generic market environment.
The Novel Approach
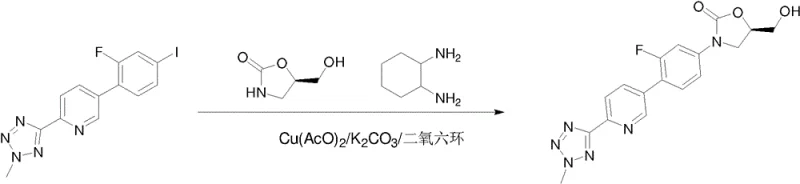
The methodology described in CN108368100B offers a streamlined alternative by introducing a versatile coupling strategy that tolerates a wide range of reaction conditions and catalysts. By utilizing copper catalysts such as cuprous iodide or copper acetate in conjunction with diamine ligands, the process achieves efficient C-N bond formation under moderate thermal conditions, typically between 60°C and 110°C. This eliminates the need for cryogenic cooling and highly pyrophoric reagents, drastically simplifying the engineering requirements for production facilities. Additionally, the route allows for the use of diverse leaving groups, including halides and sulfonates, providing flexibility in raw material sourcing and enabling process chemists to select the most cost-effective precursors for their specific supply chain configurations.
Mechanistic Insights into Copper-Catalyzed Ullmann-Type Coupling
The core innovation of this patent lies in the application of copper-catalyzed coupling reactions to link the functionalized biaryl system with the chiral oxazolidinone ring. Mechanistically, this involves the oxidative addition of the aryl halide or pseudohalide to the copper center, followed by coordination with the nitrogen nucleophile of the oxazolidinone. The presence of ligands such as cyclohexanediamine stabilizes the copper species and facilitates the transmetallation and reductive elimination steps necessary to form the C-N bond. This catalytic cycle is highly efficient and demonstrates excellent tolerance for the sensitive tetrazole and pyridine motifs present in the molecule, preventing decomposition side reactions that often plague palladium-catalyzed systems in similar contexts.
Impurity control is meticulously managed through the selection of specific bases and solvents that minimize side reactions such as dehalogenation or homocoupling. The patent details the use of inorganic bases like potassium carbonate in polar aprotic solvents such as dioxane or DMF, which optimize the solubility of reactants while maintaining the stability of the catalytic complex. Post-reaction workup procedures involving aqueous washes and recrystallization from specific solvent pairs ensure that residual copper levels are reduced to trace amounts, meeting strict regulatory limits for heavy metals in drug substances. This level of process control is essential for ensuring the safety and efficacy of the final antibiotic product.
How to Synthesize Tedizolid Intermediates Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing the key oxazolidinone intermediate with high fidelity. The process begins with the preparation of the biaryl scaffold via Suzuki coupling, followed by conversion to a reactive leaving group, and concludes with the critical copper-catalyzed coupling step. Detailed operational parameters regarding temperature, stoichiometry, and purification are provided to ensure reproducibility. For a comprehensive guide on executing these steps in a GMP environment, please refer to the standardized synthesis instructions below.
- Prepare the biaryl core via Suzuki coupling of a tetrazolyl-pyridine boronate with a fluoro-substituted aniline or phenol derivative.
- Convert the amino or hydroxy group on the biaryl intermediate into a leaving group such as iodide or triflate.
- Perform a copper-catalyzed coupling reaction between the leaving group intermediate and (R)-5-(hydroxymethyl)oxazolidin-2-one.
- Finalize the synthesis by phosphorylating the hydroxymethyl group using phosphorus oxychloride to yield Tedizolid Phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere technical feasibility. The shift away from precious metal catalysts like palladium and silver towards base metals like copper results in a direct reduction in raw material expenditure, which is critical for maintaining margin integrity in competitive generic drug markets. Furthermore, the elimination of cryogenic processing steps reduces energy consumption and capital investment in specialized cooling equipment, thereby lowering the overall cost of goods sold. These efficiencies translate into a more resilient supply chain capable of withstanding fluctuations in the availability of exotic reagents.
- Cost Reduction in Manufacturing: The substitution of expensive silver salts and palladium catalysts with economical copper sources significantly lowers the direct material costs associated with each batch. Additionally, the simplified workup procedures reduce solvent usage and waste disposal fees, contributing to a leaner manufacturing budget without compromising product quality.
- Enhanced Supply Chain Reliability: By relying on commercially available and stable reagents such as copper iodide and common inorganic bases, manufacturers can mitigate the risk of supply disruptions often associated with specialized organometallic reagents. The robustness of the reaction conditions also allows for sourcing from a broader range of qualified vendors, enhancing supply security.
- Scalability and Environmental Compliance: The moderate reaction temperatures and absence of hazardous pyrophoric reagents make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced environmental footprint aligns with modern green chemistry principles, facilitating smoother regulatory approvals and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided within the patent documentation, offering clarity on performance metrics and operational requirements.
Q: What are the primary cost advantages of the copper-catalyzed route over traditional palladium methods?
A: The novel route utilizes abundant copper catalysts like CuI or Cu(OAc)2 instead of expensive palladium or silver reagents, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: How does this method improve safety compared to previous synthetic strategies?
A: Unlike prior art requiring cryogenic conditions (-65°C) and hazardous reagents like n-BuLi, this process operates at moderate temperatures (60-110°C), eliminating the need for specialized low-temperature equipment and enhancing operational safety.
Q: Is the purity of the intermediate suitable for pharmaceutical grade applications?
A: Yes, the patent examples demonstrate high purity levels (often exceeding 97-99% by HPLC) achieved through optimized crystallization and pulping steps, ensuring compliance with stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tedizolid Phosphate Supplier
As a leading CDMO specializing in complex pharmaceutical intermediates, NINGBO INNO PHARMCHEM possesses the technical expertise to adapt and optimize this advanced catalytic route for commercial production. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing floor is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Tedizolid intermediate meets the highest global pharmacopoeia standards.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a secure and efficient supply of this critical antibiotic intermediate for your pipeline.
