Scalable Synthesis of Tedizolid Phosphate Intermediates via Novel Zinc-Catalyzed Coupling
Introduction to Patent CN110804038B and Technological Breakthroughs
The pharmaceutical industry continuously seeks robust synthetic pathways for second-generation oxazolidinone antibiotics, specifically Tedizolid phosphate, a critical agent for treating acute bacterial skin and skin tissue infections (ABSSSI). Patent CN110804038B, published in early 2021, introduces a transformative preparation method for Tedizolid phosphate and its key intermediates that fundamentally addresses the safety and efficiency limitations of prior art. This intellectual property discloses a novel two-step sequence that replaces hazardous organotin and cryogenic organolithium reagents with a safer, zinc-mediated catalytic system. For R&D directors and procurement strategists, this represents a pivotal shift towards greener chemistry that maintains rigorous purity standards while drastically simplifying the operational complexity of large-scale manufacturing. The disclosed method achieves impressive yields ranging from 72% to 74% and delivers intermediates with HPLC purity exceeding 98.0%, meeting the stringent requirements for active pharmaceutical ingredient (API) production.
The significance of this patent extends beyond mere academic interest; it offers a viable commercial solution for reliable pharmaceutical intermediate supplier networks aiming to secure the supply chain for this vital antibiotic. By utilizing abundant zinc powder and metallocene transition metal catalysts such as cobaltocene or nickelocene, the process mitigates the severe environmental and health risks associated with tin residues and pyrophoric lithium reagents. This technological advancement ensures that the production of Tedizolid phosphate can be scaled up to industrial levels without the prohibitive costs of specialized cryogenic equipment or complex heavy metal clearance protocols. As we delve deeper into the technical specifics, it becomes evident that this methodology provides a sustainable foundation for cost reduction in pharmaceutical intermediates manufacturing while enhancing overall process safety and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN110804038B, the synthesis of Tedizolid phosphate intermediates relied heavily on methodologies fraught with significant operational hazards and efficiency bottlenecks. One prominent historical approach, documented in patents like CN200480037612, utilized hexabutylditin to generate organotin intermediates which were subsequently coupled using palladium catalysis. This tin-based route is severely disadvantaged by the high toxicity of organotin reagents, which pose substantial risks to operator safety and require extensive, costly purification steps to ensure no toxic tin residues remain in the final drug substance. Furthermore, literature reports indicate that the total reaction yield for this tin-mediated pathway is disappointingly low, often hovering around 24% in standard conditions and reaching only 42% even after optimization, rendering it economically unviable for large-scale commercial production.
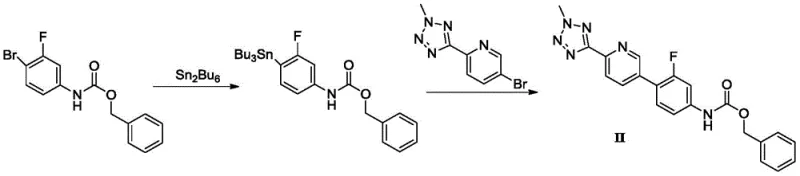
Another conventional strategy, described in documents such as CN200980140144, attempted to circumvent tin toxicity by employing n-butyl lithium for halogen-lithium exchange at extremely low temperatures. This cryogenic approach necessitates maintaining reaction environments at -78°C, requiring specialized and energy-intensive cooling infrastructure that is not readily available in many standard pharmaceutical manufacturing facilities. Additionally, n-butyl lithium is highly flammable and pyrophoric, presenting severe safety challenges for bulk handling and increasing the risk of catastrophic incidents during scale-up. The cumulative yield for this borate intermediate route is reported to be merely 55%, further compounding the economic inefficiencies. These legacy methods collectively highlight a critical industry need for a safer, more efficient, and industrially scalable synthetic route that eliminates toxic metals and extreme thermal conditions.
The Novel Approach
The inventive method disclosed in CN110804038B elegantly resolves these historical challenges by introducing a zinc-mediated activation strategy that operates under mild, ambient-friendly conditions. Instead of relying on toxic tin or dangerous lithium reagents, the process utilizes commercially available zinc powder in conjunction with a metallocene transition metal catalyst to activate the aryl bromide substrate. This transformation occurs in common organic solvents such as tetrahydrofuran or acetonitrile at temperatures ranging from 0°C to 60°C, completely eliminating the need for energy-draining cryogenic cooling systems. The resulting organozinc intermediate is then seamlessly coupled with the tetrazole-pyridine fragment using a palladium catalyst, achieving a robust total yield of 72% to 74%. This substantial improvement in yield, coupled with the elimination of hazardous reagents, signifies a major leap forward in process chemistry, offering a pathway that is both economically superior and environmentally responsible.
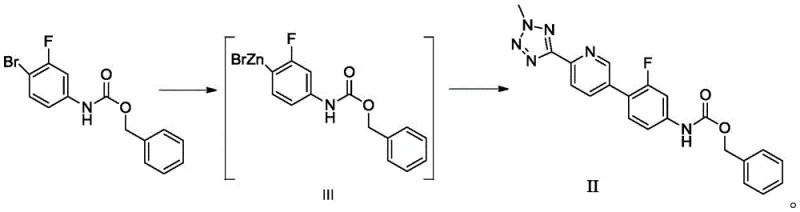
Furthermore, the novel approach ensures exceptional product quality, with the prepared Tedizolid phosphate intermediate II demonstrating an HPLC purity of greater than 98.0%. The final API derived from this intermediate achieves a purity exceeding 99.5%, with maximum single impurities controlled below 0.10%, fully satisfying regulatory standards for raw medicinal materials. By shifting the paradigm from hazardous stoichiometric reagents to a catalytic system based on abundant metals, this method facilitates the commercial scale-up of complex pharmaceutical intermediates with reduced regulatory burden and lower waste disposal costs. The operational simplicity allows for implementation in standard reactor setups, making it an ideal candidate for contract development and manufacturing organizations (CDMOs) seeking to optimize their production portfolios for high-value antibiotics.
Mechanistic Insights into Zinc-Catalyzed Activation and Coupling
The core mechanistic innovation of this patent lies in the efficient generation of the organozinc species (Intermediate III) through a catalytic cycle involving metallocene transition metal complexes. In the first step, 3-fluoro-4-bromophenyl benzyl carbamate reacts with zinc powder in the presence of a catalyst such as cobaltocene, nickelocene, or ferrocene derivatives. The metallocene catalyst facilitates the electron transfer necessary to activate the carbon-bromine bond, allowing zinc to insert effectively at moderate temperatures between 20°C and 30°C. This avoids the harsh conditions typically required for direct zinc insertion and prevents the formation of excessive homocoupling byproducts. The molar ratio of zinc powder to substrate is carefully optimized between 2.0 and 8.0, ensuring complete conversion while minimizing excess metal waste. The choice of solvent, preferably ethers like THF or nitriles like acetonitrile, plays a crucial role in stabilizing the organozinc species and maintaining catalyst activity throughout the reaction duration of 4 to 8 hours.
Following the formation of Intermediate III, the second step involves a palladium-catalyzed cross-coupling reaction with 2-methyl-5-(5-bromopyridine-2-yl)tetrazole. This step utilizes robust palladium sources such as tris(dibenzylideneacetone)dipalladium or tetrakis(triphenylphosphine)palladium in the presence of a base like sodium carbonate or triethylamine. The reaction proceeds at elevated temperatures between 55°C and 85°C, driving the coupling to completion within 3 to 8 hours. Crucially, the mild nature of the preceding zinc activation step ensures that the sensitive tetrazole and carbamate functionalities remain intact, minimizing degradation pathways that often plague harsher lithiation methods. Impurity control is achieved through careful monitoring via HPLC or TLC, ensuring that the starting material is consumed to less than 1% before workup. The subsequent purification via recrystallization from ethyl acetate further refines the crystal lattice, removing trace palladium and zinc residues to deliver a product of exceptional chemical integrity.
How to Synthesize Tedizolid Phosphate Intermediate II Efficiently
The synthesis of Tedizolid phosphate intermediate II via this patented route involves a streamlined two-step protocol that prioritizes safety and yield. The process begins with the activation of the aryl bromide using zinc powder and a metallocene catalyst in an inert atmosphere, followed immediately by the palladium-catalyzed coupling with the heterocyclic partner. Detailed operational parameters, including specific solvent volumes, catalyst loadings, and temperature profiles, are critical for reproducing the high yields reported in the patent examples. For process chemists looking to implement this technology, adherence to the specified molar ratios and reaction times is essential to maximize the formation of the desired biaryl product while suppressing side reactions. The standardized synthesis steps outlined below provide a foundational guide for translating this laboratory-scale innovation into a robust manufacturing process.
- React 3-fluoro-4-bromophenyl benzyl carbamate with zinc powder and a metallocene transition metal catalyst (e.g., cobaltocene) in an organic solvent like THF at 0-60°C to generate Intermediate III.
- Perform a coupling reaction between the Intermediate III solution and 2-methyl-5-(5-bromopyridine-2-yl)tetrazole using a palladium catalyst and base at 45-85°C.
- Purify the crude product through filtration, extraction, and recrystallization from ethyl acetate to achieve HPLC purity greater than 98.0%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the zinc-catalyzed synthesis route described in CN110804038B offers profound strategic advantages that extend well beyond simple chemical yield improvements. The elimination of hexabutylditin and n-butyl lithium removes two of the most significant cost drivers and risk factors associated with the traditional production of Tedizolid intermediates. Organotin reagents are not only expensive but also subject to increasingly stringent environmental regulations regarding disposal and residual limits in APIs, which can lead to costly delays and batch rejections. Similarly, the requirement for cryogenic cooling in lithium-based routes demands specialized infrastructure and high energy consumption, inflating the overhead costs of production. By transitioning to a zinc-based system that operates at near-ambient temperatures, manufacturers can significantly reduce utility costs and capitalize on the widespread availability and low price point of zinc powder.
- Cost Reduction in Manufacturing: The shift to zinc catalysis eliminates the need for expensive, toxic tin reagents and the complex purification steps required to remove heavy metal residues, leading to substantial cost savings in raw materials and waste treatment. Furthermore, the avoidance of cryogenic conditions (-78°C) removes the necessity for specialized low-temperature reactors and the high energy expenditure associated with maintaining such extreme environments, thereby lowering the overall operational expenditure per kilogram of product. The higher reaction yields (72-74% compared to 24-55% in prior art) directly translate to better material efficiency, reducing the amount of starting material required to produce a fixed quantity of API and optimizing the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Zinc powder and metallocene catalysts are commodity chemicals with stable global supply chains, unlike specialized organotin or pyrophoric organolithium reagents which may face supply disruptions or strict transportation regulations. The mild reaction conditions reduce the risk of batch failures due to temperature excursions or equipment malfunctions, ensuring a more consistent and predictable production schedule. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting market demand for this essential antibiotic without interruption, thereby strengthening the resilience of the entire pharmaceutical supply network.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard reactor configurations and avoiding hazardous reagents that complicate safety audits and environmental permitting. The reduction in toxic waste generation aligns with green chemistry principles, facilitating easier compliance with environmental protection agencies and reducing the liability associated with hazardous waste disposal. This environmental stewardship not only protects the corporate reputation but also future-proofs the manufacturing site against tightening global regulations on chemical emissions and effluent quality, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel zinc-catalyzed synthesis for Tedizolid phosphate intermediates. These answers are derived directly from the experimental data and technical disclosures within patent CN110804038B, providing clarity on reaction conditions, safety profiles, and scalability potential. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the zinc-catalyzed method superior to traditional tin-based routes for Tedizolid synthesis?
A: Traditional tin-based routes utilize highly toxic hexabutylditin, which poses significant environmental hazards and leaves difficult-to-remove tin residues in the final API. The novel zinc-catalyzed method described in CN110804038B eliminates toxic tin reagents entirely, uses abundant zinc powder, and operates under much milder conditions, resulting in higher yields (72-74% vs 24-42%) and superior purity profiles suitable for pharmaceutical standards.
Q: What are the critical reaction conditions for the zinc activation step?
A: The zinc activation step requires precise control of the catalyst system, typically employing metallocene transition metal complexes such as cobaltocene or nickelocene at molar ratios between 0.001 and 0.1 relative to the substrate. The reaction is conducted in ether or nitrile solvents like tetrahydrofuran or acetonitrile at temperatures ranging from 0°C to 60°C, avoiding the extreme cryogenic conditions (-78°C) required by prior art lithiation methods.
Q: How does this process impact the supply chain stability for Tedizolid Phosphate?
A: By replacing hazardous n-butyl lithium and expensive tin reagents with stable zinc powder and robust palladium catalysts, the process significantly reduces operational risks and equipment costs associated with cryogenic cooling. This simplification enhances supply chain reliability by minimizing batch failures due to temperature excursions and ensuring consistent availability of key starting materials, thereby supporting continuous commercial-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tedizolid Phosphate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-quality pharmaceutical intermediates. Our technical team has thoroughly analyzed the zinc-catalyzed route disclosed in CN110804038B and possesses the expertise to implement this technology effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Tedizolid phosphate intermediate meets the highest international standards for safety and efficacy.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this zinc-mediated process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain security and drive down manufacturing costs for this vital antibiotic therapy.
