Advanced Isocyanate Coupling Strategy for Commercial Scale-up of High-Purity Mesosulfuron-methyl
Advanced Isocyanate Coupling Strategy for Commercial Scale-up of High-Purity Mesosulfuron-methyl
The global demand for high-efficiency sulfonylurea herbicides continues to drive innovation in agrochemical intermediate manufacturing, specifically for critical compounds like Mesosulfuron-methyl. A pivotal advancement in this domain is detailed in Chinese patent CN114539165A, which discloses a novel preparation method that fundamentally shifts the synthetic paradigm from traditional transesterification to a highly efficient isocyanate coupling route. This technological breakthrough addresses long-standing industry pain points regarding byproduct management and final product stability, offering a robust pathway for reliable agrochemical intermediate supplier networks to enhance their portfolio quality. By converting the aminopyrimidine precursor into a reactive isocyanate intermediate prior to condensation with the sulfonamide core, the process achieves exceptional yields exceeding 92% and purities reaching 99.1%, setting a new benchmark for commercial scale-up of complex herbicide intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Mesosulfuron-methyl has relied heavily on transesterification reactions involving pyrimidine aminophenyl esters, a method fraught with significant environmental and quality control challenges that hinder cost reduction in herbicide manufacturing. The most critical defect of this legacy approach is the stoichiometric generation of phenol as a byproduct, with data indicating that approximately 190 kilograms of phenol are produced for every ton of final product synthesized. While theoretical recycling of phenol is possible, its physicochemical properties make recovery energetically expensive and operationally difficult, often leading to residual phenol contamination in the final active ingredient. This residue is not merely an impurity; it acts as a catalyst for product degradation, causing the herbicide to turn red and deteriorate during storage, thereby severely compromising the shelf-life and market value of the agrochemical product.
The Novel Approach
In stark contrast to the problematic transesterification pathways, the novel methodology presented in CN114539165A employs a strategic isocyanate coupling mechanism that completely circumvents the formation of phenolic waste streams. This innovative route involves the initial activation of 2-amino-4,6-dimethoxypyrimidine using a phosgene equivalent to form a highly reactive isocyanate intermediate, which is subsequently condensed with the sulfonamide component under mild conditions. By eliminating the phenol-generating step entirely, the process not only simplifies the downstream purification workflow but also drastically improves the color stability and chemical integrity of the final Mesosulfuron-methyl. Furthermore, unlike alternative carbamate routes that suffer from low purity caps around 96% due to high-temperature reflux requirements, this isocyanate method operates at controlled low temperatures, effectively suppressing thermal side reactions and enabling the production of ultra-high purity material suitable for premium agricultural applications.
Mechanistic Insights into Isocyanate Coupling Reaction
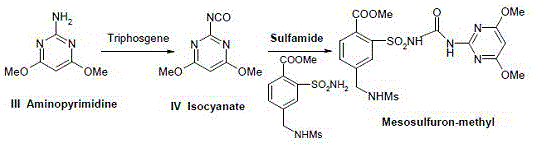
The core of this synthetic breakthrough lies in the precise generation and utilization of the isocyanate intermediate, a species that offers superior electrophilicity for the subsequent urea bond formation without the baggage of difficult-to-remove leaving groups. The reaction initiates by dispersing a phosgene source, such as bis(trichloromethyl) carbonate (triphosgene), in an aprotic solvent like dichloromethane and cooling the mixture to a rigorous 0-5°C to manage the exothermic nature of the activation. Upon the dropwise addition of the aminopyrimidine solution and a tertiary amine acid scavenger, the amine is converted into the corresponding isocyanate with high fidelity, a transformation that must be carefully monitored to prevent oligomerization or hydrolysis. This activated intermediate is then introduced to the sulfonamide nucleophile, where the nitrogen of the sulfonamide attacks the electrophilic carbon of the isocyanate group, forming the characteristic sulfonylurea bridge that defines the herbicide's biological activity.
Impurity control in this mechanism is predominantly governed by thermal management and stoichiometric precision, ensuring that the reactive isocyanate species reacts exclusively with the intended sulfonamide rather than undergoing self-polymerization or solvent interaction. The patent specifies maintaining the condensation temperature below 30°C during the addition of the isocyanate solution, a critical parameter that kinetically favors the desired cross-coupling over potential side reactions that could generate urea dimers or other structural analogs. Additionally, the use of specific acid binding agents like triethylamine or diisopropylethylamine in molar ratios of 3-5:1 ensures complete neutralization of the hydrogen chloride generated during the isocyanate formation, preventing acid-catalyzed decomposition of the sensitive pyrimidine ring. This meticulous control over the reaction environment results in a crude product profile that is exceptionally clean, requiring minimal recrystallization to achieve the reported 99% purity levels.
How to Synthesize Mesosulfuron-methyl Efficiently
The operational protocol for this synthesis is designed for reproducibility and safety, leveraging standard chemical engineering unit operations that are easily adaptable to existing multipurpose reactors in fine chemical facilities. The process begins with the preparation of the isocyanate solution in a dedicated vessel, followed by its controlled transfer into the sulfonamide reaction mixture, ensuring that the exotherm is managed through jacketed cooling systems. Detailed standardized operating procedures regarding solvent selection, addition rates, and workup protocols are essential for maintaining the high yield and purity profiles demonstrated in the patent examples. For a comprehensive breakdown of the specific reagent quantities, timing, and isolation techniques required to replicate this high-performance route, please refer to the structured guide below.
- Prepare the isocyanate intermediate by reacting 2-amino-4,6-dimethoxypyrimidine with a phosgene equivalent like triphosgene in dichloromethane at 0-5°C, using triethylamine as an acid scavenger.
- In a separate vessel, dissolve 5-methanesulfonamidomethyl-2-methoxycarbonylbenzenesulfonamide in dichloromethane and cool to 0-5°C.
- Slowly add the isocyanate solution to the sulfonamide mixture while maintaining the temperature below 30°C, then stir, quench with water, acidify, and filter to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this isocyanate-based synthesis route represents a strategic opportunity to optimize the total cost of ownership for Mesosulfuron-methyl sourcing while mitigating regulatory and environmental risks. The elimination of phenol byproducts removes a significant hazardous waste stream from the manufacturing balance sheet, thereby reducing the burden on wastewater treatment facilities and lowering the overall environmental compliance costs associated with production. Moreover, the ability to consistently achieve purities above 99% reduces the need for extensive downstream purification steps such as repeated recrystallizations or chromatographic separations, which directly translates to shorter batch cycle times and higher throughput capacity for the manufacturer. These efficiency gains allow suppliers to offer more competitive pricing structures without compromising on the stringent quality specifications required by global agrochemical registrants.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of phenol recovery units and the associated energy costs required to distill and purify phenolic waste from the reaction mixture. By utilizing readily available reagents like triphosgene and common solvents such as dichloromethane or toluene, the raw material cost structure remains stable and predictable, avoiding the volatility associated with specialized ester reagents used in older methods. The high reaction yield, consistently demonstrated above 92% across multiple examples, ensures that raw material utilization is maximized, minimizing the cost per kilogram of the final active ingredient and providing a clear margin advantage for large-scale producers.
- Enhanced Supply Chain Reliability: From a supply continuity perspective, this method relies on commodity chemicals that are widely available in the global market, reducing the risk of bottlenecks caused by the shortage of niche intermediates. The robustness of the reaction conditions, which tolerate slight variations in temperature and addition rates without significant yield loss, makes the process highly resilient to operational fluctuations in a busy multi-product plant. This reliability ensures that suppliers can maintain consistent delivery schedules for high-purity agrochemical intermediates, safeguarding the production timelines of downstream formulators who depend on steady inputs for their herbicide blending operations.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred-gram batches with linear scalability to tonnage production without the need for exotic high-pressure or cryogenic equipment. The absence of phenolic waste simplifies the environmental permitting process for manufacturing sites, as the effluent load is significantly lower and easier to treat biologically compared to phenol-laden streams. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the sustainable agriculture value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology, derived directly from the comparative data and experimental results presented in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer or for procurement specialists assessing the quality differentiators of this specific manufacturing route. The answers provided reflect the specific advantages of the isocyanate coupling method over traditional transesterification and carbamate pathways.
Q: What are the primary limitations of conventional transesterification methods for Mesosulfuron-methyl?
A: Conventional transesterification methods generate significant amounts of phenol byproducts (approximately 190 kg per ton of product), which are difficult to recover and often cause product discoloration and stability issues during storage.
Q: How does the isocyanate coupling route improve product purity?
A: By utilizing a low-temperature isocyanate coupling mechanism instead of high-temperature reflux, this method minimizes thermal degradation and side reactions, achieving purities exceeding 99% compared to the 96% limit of older carbamate routes.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like dichloromethane or toluene and avoids the handling of difficult-to-remove phenolic waste, making it highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mesosulfuron-methyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the isocyanate coupling method requires a partner with deep technical expertise and proven infrastructure for handling reactive intermediates safely. Our facility is equipped with state-of-the-art reactor trains capable of managing exothermic reactions under strict temperature control, ensuring that the high purity and yield potential of this patent are fully realized in commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, backed by stringent purity specifications and rigorous QC labs that utilize HPLC and NMR to verify every batch against the highest industry standards. Our commitment to quality assurance means that every kilogram of Mesosulfuron-methyl we produce meets the exacting requirements for modern agrochemical formulations.
We invite global agrochemical companies and trading partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the specific economic advantages of switching to this phenol-free manufacturing method. We encourage you to contact us directly to obtain specific COA data from our recent pilot runs and to receive detailed route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-quality intermediates into your global sourcing strategy.
