Optimizing Mesosulfuron-methyl Production: A Technical Breakdown of Patented Synthetic Routes
Optimizing Mesosulfuron-methyl Production: A Technical Breakdown of Patented Synthetic Routes
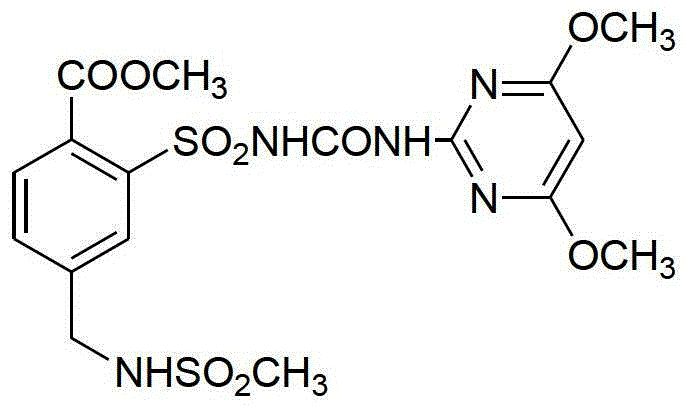
The global demand for high-efficiency sulfonylurea herbicides continues to drive innovation in intermediate manufacturing, specifically for Mesosulfuron-methyl. Patent CN103333120A introduces a transformative seven-step synthesis pathway that addresses critical bottlenecks in traditional production methods. By shifting the starting material to p-toluic acid and optimizing reaction conditions, this technology offers a robust alternative to legacy processes plagued by low yields and hazardous reagents. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for securing a stable supply of high-purity agrochemical intermediates. The following analysis details how this patented methodology enhances process safety, improves overall yield to approximately 28%, and simplifies equipment requirements for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of the route disclosed in CN103333120A, the industry relied heavily on two primary synthetic strategies, both of which presented significant operational and safety challenges. The first method, documented in DE433297, utilized 5-methyl-2-methoxycarbonylbenzenecyclic acid chloride as a starting material. This pathway was inherently flawed due to the difficulty in sourcing raw materials and the reliance on sodium azide during the azidation step. Sodium azide is a highly toxic and explosive reagent, posing severe safety risks during large-scale manufacturing and requiring specialized waste treatment protocols. Furthermore, the multi-step nature involving NBS bromination and de-tert-butylation resulted in cumbersome operations that negatively impacted overall throughput and cost efficiency.
The second conventional approach, described in US6538150, started with p-toluonitrile. While the raw materials were more accessible, the process suffered from a critically low total yield of only 12.4%. A major bottleneck in this route was the reduction step, which necessitated extremely high hydrogen pressures of 1700 kPa. Such high-pressure requirements mandate the use of expensive, specialized autoclaves and rigorous safety monitoring, drastically increasing capital expenditure. Additionally, this method involved diazotization-sulfonation steps that released toxic sulfur dioxide gas, creating significant environmental compliance burdens and necessitating complex scrubbing systems to protect worker health and meet emission standards.
The Novel Approach
The synthetic strategy outlined in CN103333120A represents a paradigm shift by utilizing p-toluic acid as the foundational building block. This route eliminates the need for hazardous sodium azide and avoids the extreme high-pressure conditions of 1700 kPa, instead operating hydrogenation steps at a much milder 0.1 MPa to 1 MPa range. By replacing the dangerous diazotization-sulfonation sequence with a controlled chlorosulfonation-ammonolysis followed by oxidation, the process significantly reduces the generation of toxic gaseous byproducts. The result is a streamlined seven-step sequence that achieves a total yield of approximately 28%, more than doubling the efficiency of the p-toluonitrile route. This improvement not only lowers the cost per kilogram of the final active ingredient but also simplifies the equipment footprint, making it highly suitable for industrial application without the need for ultra-high-pressure reactors.
Mechanistic Insights into Chlorosulfonation-Ammonolysis and Oxidative Cyclization
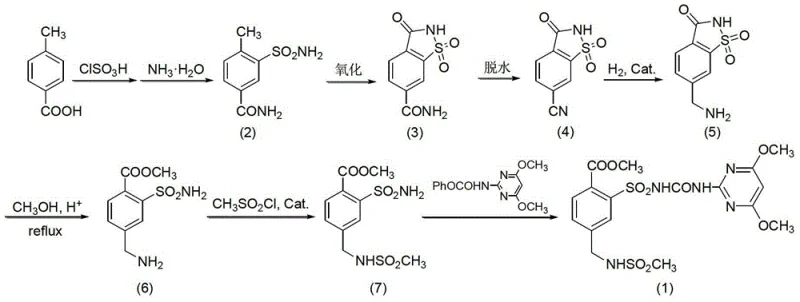
The core innovation of this synthesis lies in the initial construction of the benzenesulfonamide scaffold. The process begins with the chlorosulfonation of p-toluic acid using chlorosulfonic acid and thionyl chloride. This electrophilic aromatic substitution is carefully controlled at temperatures between 30°C and 80°C to ensure regioselectivity, favoring the formation of the 2-methyl-5-aminocarbonylbenzenesulfonamide intermediate. Following this, the immediate ammonolysis step converts the sulfonyl chloride moiety into the sulfonamide without isolating the unstable acid chloride, thereby minimizing hydrolysis side reactions. This tandem operation is crucial for maintaining high purity levels early in the synthesis, preventing the carryover of chlorinated impurities that could complicate downstream purification.
Subsequent steps involve a sophisticated oxidative cyclization to form the saccharin core, followed by dehydration and reduction. The oxidation utilizes dichromate salts in concentrated sulfuric acid, a robust system that effectively converts the methyl group into the requisite cyclic imide structure. Crucially, the dehydration of the resulting amide to the nitrile (5-cyano-saccharin) employs agents such as phosphorus oxychloride or phosphorus pentoxide. This transformation is pivotal as it activates the molecule for the subsequent catalytic hydrogenation. The reduction of the nitrile to the aminomethyl group is performed using Raney-Ni or Pd/C catalysts under mild hydrogen pressure. This specific sequence ensures that the sensitive ester and sulfonamide functionalities remain intact while selectively reducing the nitrile, demonstrating a high degree of chemoselectivity that is vital for achieving the stringent purity specifications required for agrochemical registration.
How to Synthesize Mesosulfuron-methyl Efficiently
The execution of this seven-step synthesis requires precise control over reaction parameters to maximize the 28% overall yield. The process integrates chlorosulfonation, oxidation, dehydration, reduction, alcoholysis, mesylation, and final condensation into a cohesive workflow. Each step has been optimized to use commercially available solvents and reagents, reducing supply chain friction. For technical teams looking to implement this route, attention to the recrystallization protocols described in the patent—specifically using tetrahydrofuran and petroleum ether mixtures—is essential for removing trace metal catalysts and organic impurities. The detailed standardized synthesis steps see the guide below.
- Perform chlorosulfonation-ammonolysis on p-toluic acid using chlorosulfonic acid and thionyl chloride to form 2-methyl-5-aminocarbonylbenzenesulfonamide.
- Oxidize the sulfonamide intermediate using dichromate oxidants in concentrated sulfuric acid to yield 5-aminocarbonyl saccharin.
- Execute dehydration using agents like POCl3 or P2O5 to convert the amide to 5-cyano-saccharin, followed by catalytic hydrogenation.
- Complete the synthesis through alcoholysis, mesylation with methanesulfonyl chloride, and final condensation with the pyrimidine carbamate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the CN103333120A synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from high-pressure hydrogenation and toxic azide chemistry directly translates to reduced operational risk and lower insurance costs for manufacturing facilities. By eliminating the need for 1700 kPa reactors, producers can utilize standard hydrogenation equipment, significantly lowering the barrier to entry for contract manufacturing organizations (CMOs) and expanding the potential supplier base. This diversification is critical for mitigating supply chain disruptions and ensuring consistent availability of this key herbicide intermediate.
- Cost Reduction in Manufacturing: The doubling of the overall yield from 12.4% to approximately 28% fundamentally alters the cost structure of Mesosulfuron-methyl production. Higher yields mean less raw material consumption per unit of output, directly reducing the variable cost of goods sold. Furthermore, the avoidance of expensive high-pressure equipment maintenance and the elimination of complex waste treatment systems for sulfur dioxide and azides result in substantial operational expenditure savings. These efficiencies allow for more competitive pricing structures in the global agrochemical market without compromising margin integrity.
- Enhanced Supply Chain Reliability: The reliance on p-toluic acid as a starting material leverages a widely available commodity chemical, reducing dependency on specialized or scarce precursors like 5-methyl-2-methoxycarbonylbenzenecyclic acid chloride. This abundance ensures that raw material shortages are unlikely to halt production. Additionally, the milder reaction conditions reduce the likelihood of unplanned shutdowns due to equipment failure or safety incidents, leading to more predictable lead times and improved on-time delivery performance for downstream formulators.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard unit operations such as filtration, distillation, and crystallization that are easily transferred from pilot plant to commercial scale. The reduction in hazardous waste generation, particularly the absence of toxic sulfur dioxide gas and explosive azides, simplifies environmental permitting and compliance reporting. This "green chemistry" aspect not only aligns with increasingly strict global environmental regulations but also enhances the brand reputation of manufacturers committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in CN103333120A, offering clarity on yield expectations, safety profiles, and scalability factors for stakeholders evaluating this technology.
Q: How does the CN103333120A route improve yield compared to prior art?
A: The patented route achieves a total yield of approximately 28%, significantly outperforming the 12.4% yield associated with older p-toluonitrile-based methods that require harsh conditions.
Q: Does this synthesis method eliminate high-risk reagents like sodium azide?
A: Yes, unlike the DE433297 route which utilizes hazardous sodium azide, this method relies on safer chlorosulfonation and catalytic hydrogenation steps, enhancing operational safety.
Q: What are the pressure requirements for the hydrogenation step?
A: The process operates at mild hydrogenation pressures between 0.1 MPa and 1 MPa, avoiding the extreme 1700 kPa high-pressure requirements found in legacy synthesis pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mesosulfuron-methyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the modern agrochemical landscape. Our technical team has extensively analyzed the CN103333120A pathway and possesses the expertise to execute this complex seven-step sequence with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless. Our facilities are equipped with the necessary reactor configurations to handle chlorosulfonation and mild hydrogenation safely, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals for high-purity Mesosulfuron-methyl supply.
