Advanced Electrooxidation Strategy for Commercial 2-Aminoindole Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing critical heterocyclic scaffolds, particularly the 2-aminoindole motif which serves as a privileged structure in numerous bioactive molecules. Patent CN111676487B introduces a groundbreaking electrooxidation amination method that fundamentally shifts the paradigm from traditional transition-metal catalysis to a metal-free, electricity-driven synthesis. This innovation addresses the growing demand for greener manufacturing processes by utilizing a simple aqueous phase system that eliminates the need for expensive and toxic metal catalysts. By leveraging constant current electrolysis, the process generates nitrogen free radicals directly from sulfonamide compounds, which then attack the indole core to form the desired C-N bond with high selectivity. This technical breakthrough not only simplifies the operational procedure but also aligns perfectly with modern environmental regulations and cost-reduction strategies essential for global supply chains. For R&D directors and procurement managers alike, this patent represents a viable route to high-purity pharmaceutical intermediates with significantly reduced downstream processing burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N bonds in aromatic systems has relied heavily on transition-metal catalyzed cross-coupling reactions, such as the renowned Ullmann or Buchwald-Hartwig aminations. These traditional methodologies, while effective in laboratory settings, present substantial challenges when translated to commercial manufacturing scales due to their reliance on precious metals like palladium or copper. The presence of these metals necessitates rigorous and costly purification steps to ensure the final product meets stringent pharmaceutical impurity specifications, often requiring specialized scavengers or multiple recrystallization cycles. Furthermore, these reactions frequently demand harsh conditions, including high temperatures and strong bases, which can compromise the stability of sensitive functional groups on the substrate. The environmental footprint of these processes is also significant, generating heavy metal waste that requires complex disposal protocols, thereby increasing the overall operational expenditure and regulatory compliance burden for chemical manufacturers.
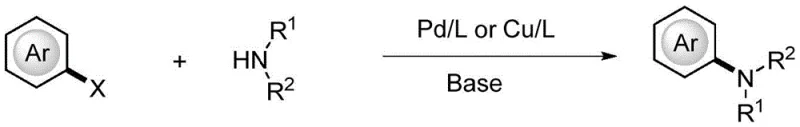
The Novel Approach
In stark contrast to the metal-dependent legacy methods, the electrooxidation strategy disclosed in the patent offers a streamlined and economically superior alternative that utilizes electricity as the primary oxidant. This approach operates under mild conditions in a green water-phase system, drastically reducing the need for volatile organic solvents and eliminating the risk of heavy metal contamination entirely. The reaction mechanism bypasses the need for pre-functionalization of the aromatic ring, allowing for direct amination which simplifies the synthetic route and improves overall atom economy. By employing a constant current system with accessible electrode materials, the process ensures consistent reaction kinetics that are easier to control and scale compared to exothermic chemical oxidations. This shift towards electro-organic synthesis not only enhances the safety profile of the manufacturing process but also delivers substantial cost savings by removing the procurement and recovery costs associated with precious metal catalysts.
Mechanistic Insights into Electrooxidation Amination
The core of this innovative synthesis lies in the precise generation and manipulation of radical species through anodic oxidation, a process that offers unparalleled control over reaction selectivity. Under the influence of a constant current, the sulfonamide substrate undergoes deprotonation in the presence of a base to form a nitrogen anion, which is subsequently oxidized at the anode to generate a highly reactive nitrogen free radical. This radical species selectively attacks the electron-rich 2-position of the substituted indole, forming a thermodynamically stable benzyl radical intermediate that is crucial for the reaction's success. The intermediate then undergoes a second oxidation event at the anode to form a carbonium ion, which finally proceeds through dehydroaromatization to yield the stable 2-amino substituted indole product. This stepwise electron transfer mechanism ensures that the reaction proceeds without the need for external chemical oxidants, minimizing side reactions and byproduct formation that typically complicate purification workflows in traditional organic synthesis.
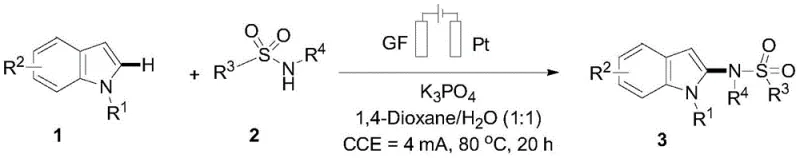
Controlling the impurity profile in the synthesis of complex heterocycles is a critical concern for R&D teams, and this electrochemical method provides inherent advantages in this regard. The use of a graphite felt anode and a platinum cathode creates a controlled electrochemical environment where the potential can be finely tuned to favor the desired radical pathway over competing oxidation processes. The aqueous solvent system further aids in suppressing the formation of organic byproducts that are often soluble in non-polar organic solvents, leading to cleaner reaction mixtures. Additionally, the open system design allows for the safe release of hydrogen gas generated at the cathode, preventing pressure buildup and ensuring a stable reaction environment that minimizes the risk of thermal runaways. These factors collectively contribute to a robust process capable of delivering high-purity intermediates that require minimal downstream processing, thereby enhancing the overall efficiency of the production line.
How to Synthesize 2-Aminoindole Efficiently
Implementing this electrooxidation protocol requires careful attention to the reaction parameters to ensure optimal conversion and yield, as detailed in the patent examples. The process begins with the precise charging of substituted indole and sulfonamide compounds into a reaction vessel containing a mixed solvent system of 1,4-Dioxane and water, along with potassium phosphate as the base. The electrode configuration is critical, with graphite felt serving as the anode and a platinum sheet as the cathode, maintained at a constant current of 4mA for a duration of approximately 20 hours at 80°C. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving substituted indole and sulfonamide compounds in a 1: 1 volume ratio of 1,4-Dioxane and water with potassium phosphate base.
- Set up the electrolytic cell using a Graphite Felt (GF) anode and a Platinum (Pt) cathode, maintaining a constant current of 4mA.
- Heat the reaction system to 80°C under an air atmosphere for 20 hours to facilitate radical generation and dehydroaromatization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free electrochemical process offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of transition metal catalysts removes a significant variable from the raw material cost structure, shielding the production process from the volatility of precious metal markets and reducing the capital tied up in catalyst inventory. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, translates into reduced labor costs and shorter production cycles, allowing for faster turnaround times on customer orders. The use of water as a co-solvent also enhances the safety profile of the facility, potentially lowering insurance premiums and reducing the regulatory burden associated with hazardous waste disposal. These operational efficiencies combine to create a more resilient supply chain capable of sustaining long-term production volumes without the bottlenecks typically associated with traditional catalytic methods.
- Cost Reduction in Manufacturing: The removal of expensive palladium or copper catalysts from the synthesis route results in direct material cost savings that accumulate significantly over large production batches. Without the need for specialized metal scavengers or extensive purification protocols to meet residual metal limits, the downstream processing costs are drastically simplified, leading to a lower cost of goods sold. This economic advantage allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins. The reliance on electricity as a reagent also provides a more predictable cost model compared to chemical oxidants which may fluctuate in price and availability.
- Enhanced Supply Chain Reliability: The raw materials required for this electrooxidation process, such as substituted indoles and sulfonamides, are readily available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive metal-catalyzed reactions, ensures consistent batch-to-batch quality even in varying manufacturing environments. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical clients who require strict adherence to delivery schedules. The simplified equipment requirements also mean that production can be easily scaled or transferred between facilities without significant requalification efforts.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system aligns perfectly with increasingly stringent environmental regulations regarding solvent emissions and waste discharge. Scaling this process is straightforward as the electrochemical parameters can be adjusted linearly, and the use of durable electrode materials ensures long equipment lifecycles with minimal maintenance. The absence of heavy metal waste simplifies the environmental compliance process, reducing the administrative overhead and costs associated with waste treatment and disposal. This green chemistry approach not only future-proofs the manufacturing process against tighter regulations but also enhances the corporate sustainability profile for partners seeking eco-friendly supply chain solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrooxidation technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: Why is electrooxidation preferred over traditional metal catalysis for 2-aminoindoles?
A: Traditional methods rely on expensive transition metals like Palladium or Copper which require complex removal steps. Electrooxidation uses electricity as the reagent, eliminating metal contamination risks and reducing purification costs significantly.
Q: What are the safety advantages of the aqueous reaction system?
A: The process utilizes a water-based solvent system which is inherently safer than volatile organic solvents. Additionally, the open system design prevents hydrogen accumulation, mitigating explosion risks during scale-up.
Q: Can this method be scaled for industrial pharmaceutical production?
A: Yes, the use of simple electrode materials like graphite felt and platinum, combined with mild reaction conditions (80°C), makes the process highly adaptable for large-scale continuous flow or batch reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrooxidation technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required by global pharmaceutical companies. We are committed to leveraging such advanced synthetic strategies to deliver superior quality intermediates that support our clients' drug development pipelines.
We invite you to collaborate with us to optimize your supply chain for 2-aminoindole derivatives and other complex heterocycles. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can enhance your production efficiency and reduce overall manufacturing costs. Let us help you navigate the transition to greener, more cost-effective synthesis methods.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
