Advanced Metalloporphyrin Catalysis for Commercial Scale Production of Pyrazine-2-Carboxylic Acid Intermediates
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for synthesizing critical intermediates, particularly for essential medications such as anti-tuberculosis agents. Patent CN102617490A introduces a transformative methodology for the preparation of pyrazine-2-carboxylic acid, a pivotal building block in the synthesis of pyrazinamide and oltipraz. This innovation leverages advanced metalloporphyrin catalytic systems to facilitate the direct oxidation of 2-methylpyrazine using molecular oxygen, marking a significant departure from traditional stoichiometric oxidation processes. By utilizing mononuclear or binuclear metalloporphyrin structures, the process achieves high efficiency under relatively mild conditions, addressing long-standing challenges regarding waste generation and energy consumption in fine chemical manufacturing. The strategic implementation of this technology offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks aiming to enhance their production capabilities while adhering to stricter environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of pyrazine-2-carboxylic acid has been plagued by severe economic and environmental drawbacks inherent to older oxidation technologies. Prior art methods, such as those disclosed in US Patent 3154549, relied heavily on sodium dichromate as a stoichiometric oxidant, necessitating reaction temperatures as high as 225°C to drive the conversion. This reliance on heavy metal oxidants not only incurred substantial raw material costs but also generated massive quantities of toxic chromium-containing wastewater, creating a significant burden for waste treatment facilities and increasing overall operational expenditures. Furthermore, alternative methods utilizing crown ether catalysts in non-polar solvents like 1,2-dimethoxyethane required catalyst loadings as high as 2-3% by weight, which complicated downstream purification and introduced hazardous organic solvents into the manufacturing stream. These conventional approaches resulted in high energy consumption due to extreme thermal requirements and posed serious safety risks associated with handling corrosive oxidants and volatile organic compounds at elevated temperatures.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient biomimetic catalytic system that operates under significantly milder and safer conditions. By employing specific metalloporphyrin catalysts at ultra-low loadings ranging from 0.02% to 0.07% of the raw material weight, the process eliminates the need for expensive stoichiometric oxidants in favor of clean molecular oxygen. The reaction proceeds effectively in an environmentally benign solvent system comprising ethanol or an ethanol-water mixture, completely avoiding the use of toxic chlorinated or ether-based solvents. Operating temperatures are reduced to a range of 80-140°C, which drastically lowers energy requirements and enhances process safety profiles for commercial scale-up of complex pharmaceutical intermediates. Additionally, the unique property of the metalloporphyrin catalyst to degrade automatically post-reaction removes the necessity for complex catalyst recovery steps, thereby streamlining the workflow and reducing capital investment in separation equipment.
Mechanistic Insights into Metalloporphyrin-Catalyzed Oxidation
The core of this technological breakthrough lies in the sophisticated design of the metalloporphyrin catalysts, which mimic the active sites of natural enzymes like cytochrome P450 to activate molecular oxygen. The patent specifies the use of mononuclear metalloporphyrins defined by Formula (I) and (II), or binuclear structures represented by Formula (III), where the central metal ions can be manganese, iron, cobalt, copper, zinc, chromium, or nickel. These metal centers facilitate the formation of high-valent metal-oxo species that are capable of abstracting hydrogen atoms from the methyl group of 2-methylpyrazine, initiating a radical chain reaction that ultimately yields the carboxylic acid functionality. The electronic properties of the porphyrin ring, modulated by substituents such as nitro, halogen, or carboxyl groups at the meso-positions, play a critical role in tuning the redox potential of the metal center to optimize catalytic turnover and selectivity.
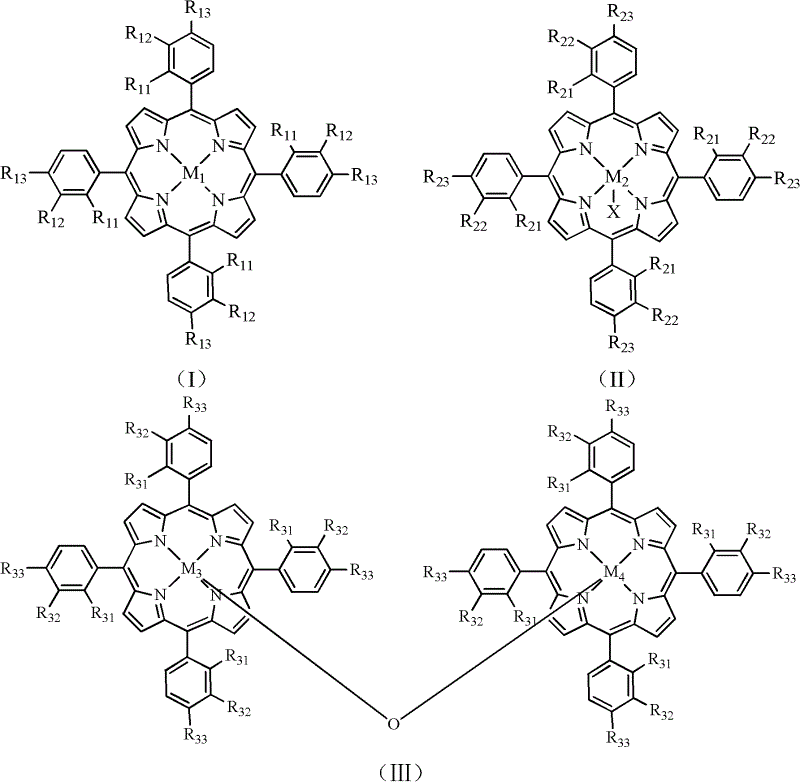
Furthermore, the mechanistic pathway ensures superior impurity control by minimizing side reactions that typically occur under harsh oxidative conditions. The use of a strong base, such as sodium hydroxide or potassium hydroxide, in conjunction with the metalloporphyrin catalyst helps to stabilize intermediate species and drives the equilibrium towards the desired carboxylate salt form. This controlled environment prevents over-oxidation or degradation of the pyrazine ring, which is a common issue in non-catalytic thermal oxidations. The binuclear catalysts, in particular, may offer cooperative effects between the two metal centers, potentially enhancing the activation of oxygen and improving the overall reaction kinetics. Understanding these mechanistic nuances is vital for R&D teams aiming to replicate high-purity pyrazine-2-carboxylic acid synthesis while maintaining strict quality standards required for API production.
How to Synthesize Pyrazine-2-Carboxylic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this green chemistry approach in a pilot or production setting. The process begins with the precise formulation of the reaction mixture, ensuring the correct molar ratios of base to substrate and the accurate dosing of the specialized metalloporphyrin catalyst. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding pressure, temperature ramping, and workup procedures that ensure maximum yield and purity.
- Prepare the reaction mixture by combining 2-methylpyrazine, ethanol or ethanol-water solvent, strong base (NaOH/KOH), and a specific mononuclear or binuclear metalloporphyrin catalyst.
- Conduct the oxidation reaction in a pressurized autoclave under 0.5-2.5 MPa oxygen pressure at temperatures between 80-140°C for 0.5 to 8 hours.
- Process the crude reaction mixture through acidification, filtration, extraction, and hot water recrystallization to isolate high-purity pyrazine-2-carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metalloporphyrin-catalyzed process translates into tangible strategic benefits that extend beyond simple chemical transformation. The shift from expensive chemical oxidants to compressed oxygen represents a fundamental change in the cost structure of raw materials, offering substantial cost savings in pharmaceutical intermediates manufacturing. By eliminating the procurement of hazardous oxidants like sodium dichromate and reducing the volume of solvent required due to higher efficiency, companies can significantly lower their direct material costs and reduce the logistical complexity associated with handling dangerous goods. This streamlined material flow enhances supply chain resilience by relying on widely available and stable commodities rather than specialty chemicals subject to market volatility.
- Cost Reduction in Manufacturing: The drastic reduction in catalyst loading to parts-per-million levels, combined with the elimination of catalyst recovery steps, leads to a simplified process flow that requires less equipment and lower utility consumption. The removal of expensive heavy metal oxidants and the ability to use cheaper, greener solvents like ethanol further contribute to a leaner cost profile, allowing for more competitive pricing in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: Utilizing ethanol and water as solvents mitigates the risks associated with the supply of specialized organic solvents, which can often face shortages or regulatory restrictions. The robustness of the reaction conditions, which tolerate a range of pressures and temperatures, ensures consistent production output even when facing minor fluctuations in utility supplies, thereby securing a steady flow of high-quality intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating at moderate pressures and temperatures that are compatible with standard stainless steel reactors found in most fine chemical plants. The generation of minimal toxic waste and the use of biodegradable solvents simplify compliance with increasingly stringent environmental regulations, reducing the liability and cost associated with waste disposal and effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on process feasibility and product quality.
Q: What are the primary advantages of using metalloporphyrin catalysts over traditional chemical oxidants?
A: Metalloporphyrin catalysts allow for the use of molecular oxygen as a clean oxidant, eliminating the need for expensive and toxic chemical oxidants like sodium dichromate, thereby significantly reducing waste treatment costs and environmental impact.
Q: Does the catalyst require complex separation procedures after the reaction?
A: No, one of the key innovations of this process is that the metalloporphyrin catalyst is used in extremely low amounts (0.02-0.07%) and automatically degrades after the reaction, removing the need for costly separation and recovery steps.
Q: How does this method improve safety compared to prior art methods?
A: This method operates at significantly lower temperatures (80-140°C) compared to conventional methods requiring up to 225°C, which drastically reduces energy consumption and minimizes operational risks associated with high-temperature processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrazine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the metalloporphyrin-catalyzed oxidation can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrazine-2-carboxylic acid meets the exacting standards required for anti-tuberculosis drug synthesis and other high-value applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this greener catalytic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production volumes and quality needs.
