Advanced Resolution Technology for High-Purity 1-Phenylethylamines: Commercial Scale-Up and Cost Efficiency
Introduction to Advanced Chiral Resolution Technologies
The production of optically active amines is a cornerstone of modern fine chemical synthesis, particularly for the development of high-performance agrochemicals and pharmaceuticals. Patent CN1128785C introduces a groundbreaking methodology for the preparation of optically active 1-phenylethylamines, addressing critical bottlenecks in chiral separation efficiency and cost-effectiveness. This technology leverages a sophisticated two-step crystallization process using (S)-(-)-N-phenylcarbamate lactate as a resolving agent, enabling the simultaneous isolation of both (R) and (S) enantiomers with exceptional optical purity. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic nuances of this patent is essential for optimizing supply chains and reducing manufacturing overheads.
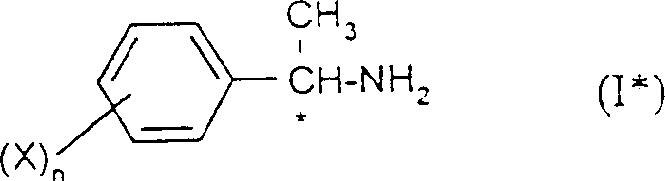
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional resolution techniques for 1-phenylethylamines often suffer from significant economic and operational inefficiencies. Prior art, such as the methods described in EP-A 0341475, typically involves reacting racemic amines with chiral acids in ethanol, followed by filtration. A major drawback of these legacy processes is the difficulty in recovering the resolving agent from the mother liquor after the initial crystallization. The resolving agent often remains dissolved in complex mixtures, requiring energy-intensive distillation or complex extraction procedures to separate, which drastically increases the cost of goods sold (COGS). Furthermore, single-step crystallization frequently fails to achieve high optical purity for the second enantiomer remaining in the solution, leading to substantial yield losses and waste generation.
The Novel Approach
The methodology disclosed in CN1128785C revolutionizes this landscape by implementing a controlled, two-stage precipitation strategy. By carefully tuning the molar ratio of the resolving agent to the racemic amine—specifically using between 0.25 and 0.5 moles of resolving agent per mole of racemate in the first step—the process selectively precipitates the less soluble diastereomeric salt of the (R)-amine. Crucially, the subsequent treatment of the mother liquor allows for the recovery of the resolving agent and the isolation of the (S)-amine with high purity. This approach not only simplifies the downstream processing but also ensures that the valuable chiral auxiliary can be recycled almost quantitatively, representing a paradigm shift in cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Diastereomeric Salt Crystallization
The core of this innovation lies in the differential solubility behavior of diastereomeric salts in specific solvent systems comprising aliphatic or aromatic hydrocarbons and lower aliphatic alcohols. When racemic 1-phenylethylamine reacts with (S)-(-)-N-phenylcarbamate lactate, two diastereomeric salts are formed: the (R)-amine/(S)-acid salt and the (S)-amine/(S)-acid salt. In a hydrocarbon-rich environment (after distilling off the alcohol), the (R)-amine/(S)-acid salt exhibits significantly lower solubility compared to its counterpart. This thermodynamic preference drives the selective crystallization of the (R)-enantiomer salt. The patent data highlights that this behavior is surprisingly distinct from one-step reactions, where such clear differentiation is often obscured by eutectic formations.
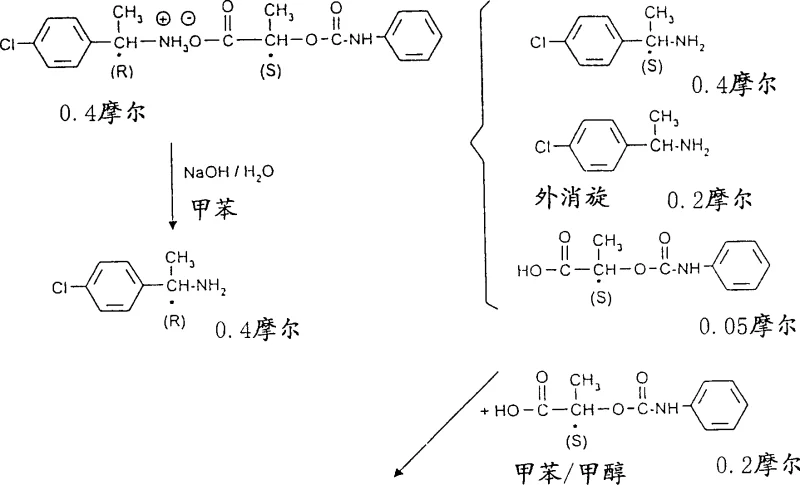
Furthermore, the process incorporates a unique mother liquor treatment step that maximizes atom economy. After isolating the (R)-salt, the remaining solution is treated with additional resolving agent—specifically calculated to be twice the molar amount of the remaining (R)-amine. This induces the precipitation of a racemic salt, effectively scavenging the unwanted (R)-isomer from the solution. Consequently, the final mother liquor becomes highly enriched with the desired (S)-amine, which can then be isolated via simple distillation. This precise control over stoichiometry and phase behavior ensures that impurities are systematically removed at each stage, resulting in a final product that meets stringent purity specifications without the need for extensive chromatographic purification.
How to Synthesize Optically Active 1-Phenylethylamines Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production. It emphasizes the importance of solvent selection, specifically recommending toluene or methylcyclohexane mixed with methanol, to facilitate the differential solubility required for effective resolution. The detailed standardized synthesis steps below guide the operator through the critical parameters of temperature control, molar dosing, and crystallization kinetics necessary to replicate the high yields and optical purities reported in the intellectual property.
- React racemic amine with 0.25-0.5 equivalents of resolving agent in toluene/methanol, distill off alcohol, and crystallize the (R)-amine salt.
- Treat the isolated salt with aqueous alkali to liberate the pure (R)-amine and recover the resolving agent from the aqueous phase.
- Process the mother liquor with additional resolving agent to precipitate racemic salt, allowing isolation of high-purity (S)-amine from the final filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology offers tangible strategic benefits beyond mere technical feasibility. The ability to recycle the resolving agent is a primary driver for cost optimization, as chiral auxiliaries often represent a significant portion of raw material expenses. By eliminating the loss of these expensive reagents into waste streams, manufacturers can achieve substantial cost savings and stabilize their input costs against market volatility. Additionally, the use of common, non-halogenated solvents like toluene and methanol simplifies regulatory compliance and waste disposal, further enhancing the environmental profile of the manufacturing process.
- Cost Reduction in Manufacturing: The closed-loop recovery of the (S)-(-)-N-phenylcarbamate lactate resolving agent fundamentally alters the cost structure of producing chiral amines. In traditional processes, the resolving agent is often a single-use consumable or requires complex regeneration. Here, the acidification of the aqueous phase post-liberation allows the resolving agent to precipitate and be reused directly. This circular material flow drastically reduces the net consumption of chiral starting materials, leading to a leaner and more economically resilient production model that protects margins even when raw material prices fluctuate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as toluene, methanol, and sodium hydroxide ensures that the supply chain is not vulnerable to the shortages often associated with exotic catalysts or specialized reagents. The robustness of the crystallization process, which tolerates variations in temperature and pressure within defined ranges, means that production can be maintained consistently across different manufacturing sites. This reliability is critical for maintaining continuous supply to downstream customers in the pharmaceutical and agrochemical sectors, minimizing the risk of stockouts or delivery delays.
- Scalability and Environmental Compliance: The process operates at mild temperatures, typically between 0°C and 40°C, and at atmospheric or slightly reduced pressure, which reduces the energy load required for heating and cooling compared to high-temperature distillation or cryogenic reactions. The elimination of chlorinated solvents like dichloromethane, which were used in prior art methods for extraction, aligns the process with modern green chemistry principles. This facilitates easier permitting and reduces the environmental footprint, making the technology scalable from kilogram to multi-ton production without encountering significant regulatory hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on process capabilities and limitations for potential partners and licensees.
Q: What is the primary advantage of this resolution method over conventional single-step crystallization?
A: Unlike conventional methods where the resolving agent is difficult to recover from the mother liquor, this two-step process allows for the quantitative recovery and reuse of the expensive (S)-(-)-N-phenylcarbamate lactate, significantly lowering material costs.
Q: Can this process be scaled for industrial production of fungicide intermediates?
A: Yes, the patent explicitly describes the use of common industrial solvents like toluene and methanol, and the crystallization steps operate at mild temperatures (0°C to 40°C), making it highly suitable for large-scale commercial manufacturing.
Q: What optical purity can be achieved for the (R)-enantiomer using this technique?
A: Experimental data within the patent indicates that the (R)-1-(4-chloro-phenyl)-ethylamine can be isolated with optical purities ranging from 96% to 98%, which meets the stringent requirements for active pharmaceutical and agrochemical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenylethylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation fungicides and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate crystallization dynamics described in CN1128785C are perfectly translated to an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optically active amine meets the exacting standards required for global regulatory filings.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
