Advanced Two-Step One-Pot Synthesis of Methyl 4-(3-bromopropyl) Benzoate for Commercial Scale
The pharmaceutical industry's relentless pursuit of effective treatments for systemic skeletal diseases like osteoporosis has placed a premium on the efficient production of key chemical intermediates. Patent CN112321425A introduces a groundbreaking synthesis method for Methyl 4-(3-bromopropyl) Benzoate, a critical precursor in the development of osteoporosis medications. This innovation addresses the longstanding inefficiencies associated with traditional manufacturing pathways by proposing a novel two-step one-pot strategy. By leveraging common raw materials such as 1-bromo-3-phenylpropane and optimizing reaction conditions through precise catalytic control, this technology promises to redefine the economic and operational landscape for reliable pharmaceutical intermediates supplier networks. The shift towards streamlined synthesis not only enhances chemical efficiency but also aligns with the global demand for cost reduction in pharmaceutical intermediates manufacturing, ensuring that life-saving treatments remain accessible through optimized supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
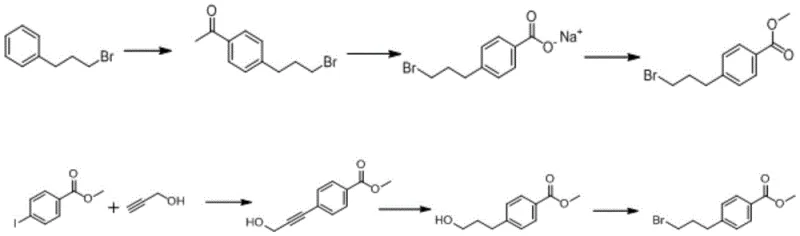
Historically, the production of Methyl 4-(3-bromopropyl) Benzoate has been plagued by cumbersome multi-step processes that hinder scalability and inflate production costs. Existing patents, such as CN102757444A and US2002065308a1, disclose synthetic routes that invariably require three distinct reaction steps, each necessitating rigorous purification protocols including column chromatography. These traditional pathways suffer from inherently low total yields, often ranging between 24% and 52%, which translates to significant material waste and elevated unit costs. Furthermore, the reliance on expensive catalysts and auxiliary raw materials in the second step of these conventional routes creates bottlenecks in commercial scale-up of complex pharmaceutical intermediates. The cumulative effect of these inefficiencies is a fragile supply chain vulnerable to raw material fluctuations and extended lead times, making it difficult for procurement teams to secure high-purity pharmaceutical intermediates at a competitive price point without compromising on quality or delivery schedules.
The Novel Approach
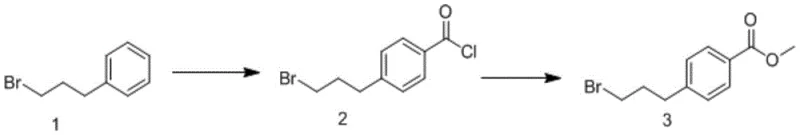
In stark contrast to the convoluted legacy methods, the innovative approach detailed in CN112321425A utilizes a sophisticated two-step one-pot methodology that dramatically simplifies the production workflow. This novel route initiates with the Friedel-Crafts acylation of 1-bromo-3-phenylpropane using oxalyl chloride and aluminum trichloride, followed immediately by esterification with methanol in the same reaction vessel. By eliminating the isolation and purification of the intermediate acid chloride, the process removes the need for resource-intensive column chromatography, thereby accelerating throughput. The reaction conditions are meticulously tuned to operate at mild temperatures, specifically initiating at -5°C and proceeding at room temperature, which reduces energy consumption and enhances safety profiles. This strategic simplification not only boosts the overall yield significantly but also ensures reducing lead time for high-purity pharmaceutical intermediates, providing a robust foundation for manufacturers aiming to optimize their operational expenditure while maintaining stringent quality standards required for drug substance production.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts Acylation
The core of this technological breakthrough lies in the precise mechanistic control of the Friedel-Crafts acylation, where aluminum trichloride acts as a potent Lewis acid catalyst to facilitate the electrophilic aromatic substitution. The reaction mechanism involves the generation of an acylium ion from oxalyl chloride, which subsequently attacks the aromatic ring of 1-bromo-3-phenylpropane. Critical to the success of this pathway is the regioselectivity achieved, favoring the formation of the para-substituted product, 4-(3-bromopropyl) benzoyl chloride, over ortho-isomers. This selectivity is governed by the steric hindrance of the propyl chain and the electronic properties of the solvent system, particularly when using trichloroethylene. Understanding this mechanistic nuance is vital for R&D directors focused on high-purity pharmaceutical intermediates, as minimizing isomeric impurities at the source reduces the burden on downstream purification processes and ensures the structural integrity of the final API intermediate.
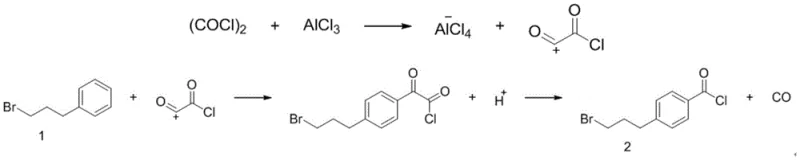
Furthermore, the control of impurity profiles is intrinsically linked to the molar ratios of the reactants and the choice of aprotic solvent. The patent data indicates that maintaining a specific molar ratio of 1-bromo-3-phenylpropane to oxalyl chloride and aluminum trichloride, ideally around 1:1.1:1.05, is crucial for maximizing yield and minimizing by-products such as upper ortho-acid chlorides. Deviations from this optimal stoichiometry can lead to decreased reaction selectivity and lower overall efficiency. Additionally, the solvent effect plays a pivotal role; trichloroethylene provides a superior medium compared to dichloromethane or ethyl acetate due to its ability to stabilize the transition state and influence the reaction equilibrium favorably. For technical teams, mastering these parameters ensures the consistent production of high-purity pharmaceutical intermediates that meet the rigorous specifications demanded by regulatory bodies, thereby mitigating the risk of batch failures and ensuring supply continuity.
How to Synthesize Methyl 4-(3-bromopropyl) Benzoate Efficiently
Implementing this advanced synthesis route requires strict adherence to the optimized reaction parameters to achieve the reported benefits in yield and purity. The process begins with the preparation of a mixed solution containing the starting material and reagents in an aprotic solvent under controlled low-temperature conditions to manage the exothermic nature of the acylation. Following the formation of the intermediate, the direct addition of methanol allows for immediate conversion to the final ester without intermediate isolation. This seamless transition between reaction stages is the key to the process's efficiency, eliminating unit operations that traditionally consume time and resources. Detailed standardized synthesis steps see the guide below for specific operational protocols that ensure reproducibility and safety during scale-up activities.
- Prepare the reaction mixture by adding aluminum trichloride to a solution of 1-bromo-3-phenylpropane and oxalyl chloride in an aprotic solvent at -5°C.
- Allow the mixture to react at room temperature for 0.5 to 3 hours to form the intermediate acid chloride.
- Directly add methanol to the cooled reaction liquid and stir at room temperature for 1 to 3 hours to obtain the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this two-step one-pot synthesis offers transformative advantages for procurement managers and supply chain heads tasked with optimizing cost structures and ensuring material availability. The elimination of multiple isolation steps and chromatographic purifications directly translates to a drastic reduction in processing time and solvent consumption, which are major cost drivers in fine chemical manufacturing. By utilizing common, commercially available raw materials like 1-bromo-3-phenylpropane and oxalyl chloride, the supply chain becomes more resilient against shortages of exotic reagents, ensuring enhanced supply chain reliability for long-term production contracts. This robustness is essential for maintaining the continuity of API production schedules, preventing costly delays that can ripple through the entire pharmaceutical value chain and impact patient access to critical medications.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently lowers the cost of goods sold by reducing the number of unit operations and minimizing waste generation. Eliminating the need for expensive catalysts in subsequent steps and avoiding chromatographic purification significantly cuts down on consumable costs and labor hours. This efficiency gain allows for substantial cost savings that can be passed down the supply chain, making the final osteoporosis medication more economically viable to produce. The qualitative improvement in process efficiency ensures that manufacturers can maintain competitive pricing margins while adhering to strict quality compliance standards without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials mitigates the risk of supply disruptions often associated with specialized or custom-synthesized reagents. The mild reaction conditions, operating primarily at room temperature after initial cooling, reduce the energy load on manufacturing facilities and decrease the dependency on complex utility infrastructure. This operational simplicity enhances the agility of the supply chain, allowing for faster response times to market demand fluctuations. Consequently, partners can rely on a more stable and predictable supply of pharmaceutical intermediates, fostering stronger strategic relationships between suppliers and multinational pharmaceutical corporations.
- Scalability and Environmental Compliance: The simplified workflow facilitates easier scale-up from laboratory to commercial production volumes, as fewer process variables need to be controlled compared to multi-step sequences. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations, reducing the burden of waste treatment and disposal. This environmental compatibility not only lowers compliance costs but also enhances the sustainability profile of the manufacturing process. Such attributes are increasingly valued by global stakeholders who prioritize green chemistry principles, positioning this synthesis method as a forward-looking solution for sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the patent data and practical considerations for industrial application, providing clarity on how this method outperforms traditional routes. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios to achieve better economic and operational outcomes.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method reduces the synthesis from three steps to a two-step one-pot process, eliminating the need for column chromatography purification and significantly improving overall yield compared to prior art which typically achieves only 24% to 52%.
Q: Which solvents are compatible with this Friedel-Crafts acylation process?
A: The process demonstrates high flexibility with various aprotic solvents including trichloroethylene, dichloromethane, and chloroform, with trichloroethylene showing optimal yield performance due to favorable solvent effects on reaction equilibrium.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of common commercially available raw materials like 1-bromo-3-phenylpropane and oxalyl chloride, combined with mild reaction conditions at room temperature, makes this route highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-(3-bromopropyl) Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN112321425A method are translated into tangible supply chain solutions. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We understand that consistency and reliability are paramount for our clients, and our infrastructure is designed to support the complex requirements of modern drug development and commercial manufacturing.
We invite you to collaborate with us to leverage this advanced technology for your osteoporosis treatment programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain resilience and drive down manufacturing costs effectively.
