Advanced Two-Step One-Pot Synthesis of Methyl 4-(3-bromopropyl) Benzoate for Commercial Scale-Up
Advanced Two-Step One-Pot Synthesis of Methyl 4-(3-bromopropyl) Benzoate for Commercial Scale-Up
The pharmaceutical industry constantly seeks efficient, scalable, and cost-effective pathways for producing critical drug intermediates, particularly those serving high-demand therapeutic areas such as osteoporosis treatment. A significant breakthrough in this domain is detailed in patent CN112321425B, which discloses a novel synthesis method for methyl 4-(3-bromopropyl) benzoate. This compound serves as a pivotal building block in the manufacture of medications designed to combat bone density loss and fracture risks. The patented technology introduces a streamlined two-step one-pot approach that fundamentally alters the economic and operational landscape of producing this specific pharmaceutical intermediate. By leveraging a Friedel-Crafts acylation strategy followed by direct esterification, the process bypasses the cumbersome isolation steps typical of legacy methods. For R&D directors and procurement specialists alike, this innovation represents a tangible opportunity to optimize supply chains, reduce manufacturing overheads, and secure a more reliable source of high-purity materials essential for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of methyl 4-(3-bromopropyl) benzoate was fraught with inefficiencies that hindered large-scale commercial viability. Existing literature, including patents such as CN102757444A and US2002065308a1, outlines synthetic routes that typically require a minimum of three distinct reaction steps. These traditional pathways often rely on expensive and specialized catalysts that drive up the raw material costs significantly. Furthermore, a critical bottleneck in these conventional methods is the absolute necessity for purification via chromatographic columns after each individual step. This reliance on chromatography not only consumes vast quantities of solvents but also drastically extends the production cycle time, creating a bottleneck for manufacturers aiming for high throughput. The cumulative effect of these multi-step processes results in relatively low overall yields, reported to be approximately 24% to 52% depending on the specific route employed. Such low efficiency translates directly into higher waste generation and increased environmental compliance burdens, making these older methods less attractive for modern, green chemistry-focused manufacturing facilities.
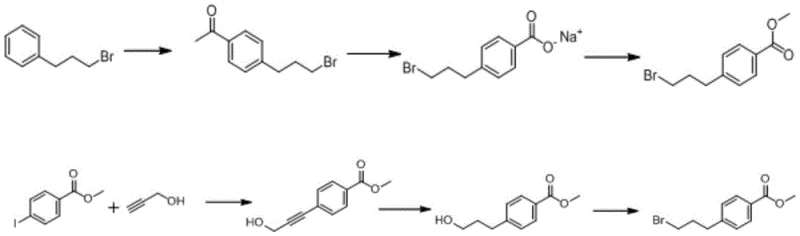
The Novel Approach
In stark contrast to the fragmented nature of previous techniques, the methodology presented in CN112321425B consolidates the synthesis into a highly efficient two-step one-pot sequence. This novel approach utilizes 1-bromo-3-phenylpropane as the starting material, reacting it with oxalyl chloride in the presence of aluminum trichloride to form an intermediate acyl chloride, which is then directly converted to the target methyl ester without isolation. By eliminating the intermediate purification step, the process dramatically reduces solvent usage and labor intensity. The reaction conditions are notably mild, primarily proceeding at room temperature after an initial cooling phase, which simplifies the thermal management requirements for reactor systems. This consolidation of steps not only accelerates the production timeline but also inherently improves the mass balance of the process. The result is a robust synthetic route that offers a superior yield profile compared to the prior art, providing a compelling value proposition for reliable pharmaceutical intermediate suppliers looking to enhance their portfolio competitiveness through process intensification.
Mechanistic Insights into AlCl3-Catalyzed Friedel-Crafts Acylation
The core chemical transformation driving this synthesis is a classic yet meticulously optimized Friedel-Crafts acylation. The mechanism initiates with the interaction between oxalyl chloride and the Lewis acid catalyst, aluminum trichloride (AlCl3). In the aprotic solvent environment, typically trichloroethylene, the aluminum trichloride coordinates with the carbonyl oxygen of the oxalyl chloride. This coordination weakens the carbon-chlorine bond, facilitating the departure of a chloride ion and the generation of a highly reactive acylium ion species. This electrophilic species is the key driver of the subsequent aromatic substitution. The 1-bromo-3-phenylpropane substrate acts as the nucleophile, where the electron-rich benzene ring attacks the acylium ion. The choice of aluminum trichloride is critical here, as it provides the necessary Lewis acidity to activate the acylating agent effectively without promoting excessive side reactions that could degrade the bromo-alkyl chain. Understanding this mechanistic pathway is vital for process chemists aiming to replicate the high selectivity observed in the patent data.
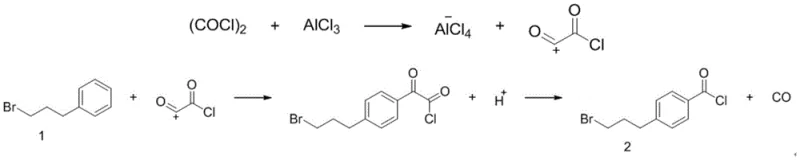
Regioselectivity is a paramount concern in electrophilic aromatic substitutions, particularly when aiming for the para-substituted product required for the osteoporosis drug intermediate. The patent data elucidates that the steric bulk of the 3-bromopropyl chain on the benzene ring naturally directs the incoming acyl group towards the para position. However, the precision of this selectivity is further enhanced by strictly controlling the stoichiometry of the reagents. The optimal molar ratio of 1-bromo-3-phenylpropane to oxalyl chloride and aluminum trichloride is maintained at approximately 1:1.1:1.05. Deviating from this narrow window, particularly by increasing the excess of oxalyl chloride and catalyst, can lead to the formation of ortho-isomers or di-acylated by-products, which complicates downstream purification. By adhering to these specific mechanistic parameters, the process ensures that the intermediate 4-(3-bromopropyl) benzoyl chloride is formed with high fidelity, setting the stage for the subsequent high-yield esterification step.
How to Synthesize Methyl 4-(3-bromopropyl) Benzoate Efficiently
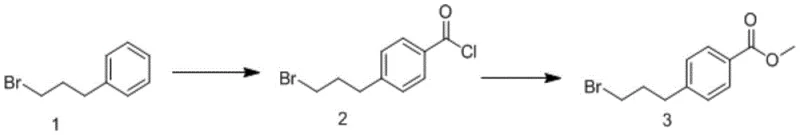
Implementing this synthesis requires precise adherence to the reaction parameters outlined in the patent to achieve the reported yields of over 40%. The process begins by dissolving the starting material, 1-bromo-3-phenylpropane, in an aprotic solvent such as trichloroethylene at a concentration of roughly 50 g/L. To this solution, oxalyl chloride is added, followed by the slow introduction of aluminum trichloride at a controlled temperature of -5°C to manage the exotherm. Once the addition is complete, the reaction mixture is allowed to warm to room temperature and stirred for 0.5 to 3 hours to ensure complete conversion to the acyl chloride intermediate. Following this acylation phase, the reaction vessel is cooled back down to -5°C, and methanol is added directly to the same pot. The mixture is then stirred at room temperature for an additional 1 to 3 hours to effect the esterification. This seamless transition from acylation to esterification without workup is the hallmark of the process efficiency. For a detailed breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Mix 1-bromo-3-phenylpropane, oxalyl chloride, and aluminum trichloride in an aprotic solvent like trichloroethylene at -5°C to room temperature.
- React the mixture for 0.5 to 3 hours to form the intermediate 4-(3-bromopropyl) benzoyl chloride.
- Without isolation, add methanol directly to the cooled reaction mixture and stir at room temperature for 1 to 3 hours to obtain the final ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers profound strategic advantages beyond mere chemical elegance. The shift from a three-step sequence requiring chromatography to a two-step one-pot process fundamentally reshapes the cost structure of manufacturing this pharmaceutical intermediate. By eliminating the need for column chromatography, the process removes a major cost center associated with silica gel, massive solvent volumes, and extended equipment occupancy time. This simplification allows for a drastic reduction in the overall production footprint and energy consumption, as the reaction proceeds largely at ambient temperatures. Furthermore, the reliance on common commercially available raw materials such as oxalyl chloride and aluminum trichloride mitigates supply risk. Unlike exotic catalysts that may have long lead times or single-source dependencies, these reagents are commodity chemicals with robust global supply chains, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the significant reduction in unit operations. By combining the acylation and esterification steps into a single vessel, manufacturers save on capital expenditure related to reactor trains and reduce the labor hours required for material handling and transfer. The avoidance of chromatographic purification is perhaps the most significant cost saver, as it eliminates the high variable costs associated with solvent recovery and waste disposal. Additionally, the improved yield profile compared to legacy routes means that less starting material is wasted per kilogram of final product, directly lowering the cost of goods sold (COGS) and improving margin potential for the final drug product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the reaction conditions. The process does not require extreme cryogenic conditions or high-pressure equipment, reducing the likelihood of unplanned downtime due to equipment failure. The use of trichloroethylene or other common aprotic solvents ensures that solvent availability is not a bottleneck. Moreover, the shorter reaction timeline—completing the synthesis in a matter of hours rather than days—allows for faster turnover of production batches. This agility enables suppliers to respond more rapidly to demand spikes from downstream pharmaceutical clients, reducing lead times and improving inventory management dynamics across the value chain.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the one-pot nature of this synthesis aligns well with green chemistry principles. The reduction in solvent usage and the elimination of silica waste from chromatography columns significantly lower the environmental footprint of the manufacturing process. This makes it easier for facilities to maintain compliance with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and hazardous waste generation. The process is inherently scalable; the linear relationship between reagent input and product output, combined with the lack of complex purification bottlenecks, facilitates a smooth transition from pilot plant scales to multi-ton commercial production, ensuring that supply can grow in tandem with market demand for osteoporosis treatments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of methyl 4-(3-bromopropyl) benzoate. These answers are derived directly from the experimental data and technical disclosures found in patent CN112321425B. They are intended to provide clarity on the feasibility, optimization, and practical application of this novel synthetic route for industry stakeholders evaluating its potential for integration into their supply chains.
Q: What are the key advantages of the one-pot synthesis method described in CN112321425B?
A: The primary advantage is the reduction of synthetic steps from three to two within a single pot, eliminating the need for intermediate purification via chromatography. This significantly lowers solvent consumption, reduces processing time, and utilizes common, cost-effective raw materials like oxalyl chloride and aluminum trichloride.
Q: How does this method improve regioselectivity compared to conventional routes?
A: By carefully controlling the molar ratio of reactants, specifically maintaining a ratio of 1-bromo-3-phenylpropane to oxalyl chloride and aluminum trichloride around 1:1.1:1.05, the process favors the formation of the para-substituted product. This minimizes ortho-isomer by-products, thereby enhancing the overall yield and purity of the final pharmaceutical intermediate.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable due to its use of standard reagents and moderate reaction conditions, primarily operating at room temperature after initial cooling. The elimination of complex purification steps like column chromatography makes it economically viable for manufacturing multi-ton quantities required for the pharmaceutical supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-(3-bromopropyl) Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive landscape of pharmaceutical intermediates. The technology described in CN112321425B exemplifies the kind of process innovation we champion to deliver value to our global partners. As a leading CDMO and supplier, we possess the technical expertise to not only replicate but also further optimize such pathways to meet the rigorous demands of the international market. Our facilities are equipped to handle complex organic syntheses, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key, which is why our stringent purity specifications and rigorous QC labs ensure that every batch of methyl 4-(3-bromopropyl) benzoate meets the highest standards required for API synthesis.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthesis technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes both quality and cost-efficiency. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your drug development and commercialization goals.
