Advanced Metal-Free Synthesis of High-Purity Myrcene and Isotopically Labeled Derivatives for Commercial Scale-Up
The global demand for high-purity terpenes, particularly myrcene, has surged due to its critical role as a building block in the fragrance, flavor, and pharmaceutical industries. Patent CN111454114A introduces a groundbreaking methodology for the synthesis of high-purity myrcene and its isotopically labeled variants (13C2 and 14C2) starting from geraniol. This innovation addresses long-standing inefficiencies in terpene processing by replacing traditional harsh dehydration protocols with a sophisticated, mild, two-step protection-elimination sequence. By utilizing a tetrahydropyranyl (THP) protection strategy followed by base-mediated elimination, the process achieves exceptional selectivity without the need for toxic heavy metal catalysts. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing that ensures consistent quality and supply stability for complex fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of myrcene from geraniol or beta-pinene has been plagued by significant technical and economic hurdles. Traditional acid-catalyzed dehydration typically necessitates extreme conditions, including high temperatures and elevated pressures, which drive up energy consumption and impose severe stress on reactor equipment. Under these harsh thermal conditions, the reaction lacks specificity, leading to a complex mixture of isomers, polymers, and cyclization by-products that are difficult to separate. Furthermore, existing catalytic methods often rely on noble metals such as palladium, which not only inflate raw material costs but also introduce stringent requirements for metal removal to meet pharmaceutical grade specifications. The accumulation of heavy metal waste and the difficulty in recovering expensive catalysts create substantial environmental liabilities and supply chain bottlenecks for large-scale manufacturers.
The Novel Approach
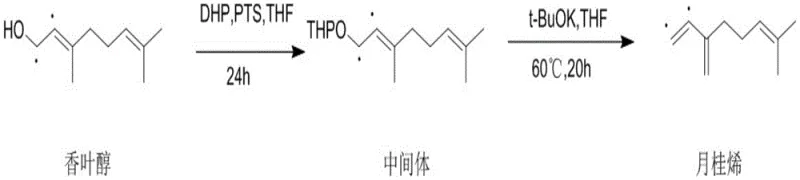
In stark contrast, the methodology disclosed in CN111454114A employs a clever synthetic detour that circumvents these pitfalls entirely. The process begins by converting the hydroxyl group of geraniol into a tetrahydropyranyl (THP) ether using 3,4-dihydro-2H-pyran (DHP) and a catalytic amount of p-toluenesulfonic acid. This protection step stabilizes the molecule, allowing for a subsequent elimination reaction under remarkably mild alkaline conditions using potassium tert-butoxide and 18-crown-6. This approach eliminates the need for high-pressure reactors and noble metal catalysts, significantly lowering the barrier to entry for production. The result is a streamlined workflow that delivers myrcene with purity levels exceeding 99%, effectively removing the need for energy-intensive fractional distillation and ensuring a cleaner product profile suitable for sensitive applications.
Mechanistic Insights into THP Protection and Base-Mediated Elimination
The core of this technological advancement lies in the precise control of reaction kinetics through functional group manipulation. In the first stage, the nucleophilic attack of the geraniol hydroxyl group on the activated DHP creates a robust THP ether linkage. This transformation is crucial because it converts a poor leaving group (hydroxide) into a stable intermediate that can withstand the subsequent reaction conditions without premature degradation. The use of p-toluenesulfonic acid (PTS) as a mild organocatalyst ensures that the acid-sensitive double bonds in the geraniol backbone remain intact, preventing the acid-catalyzed rearrangements that typically plague direct dehydration attempts. This step sets the stage for a highly controlled elimination, preserving the structural integrity of the carbon skeleton.
The second stage involves an E2-type elimination mechanism facilitated by the strong non-nucleophilic base, potassium tert-butoxide. The addition of 18-crown-6 acts as a phase transfer catalyst, complexing with the potassium cation to enhance the nucleophilicity and basicity of the tert-butoxide anion in the organic solvent (THF). This activation allows the base to abstract a proton from the allylic position adjacent to the THP ether, triggering the simultaneous expulsion of the THP group and the formation of the conjugated diene system characteristic of myrcene. Because this elimination occurs at moderate temperatures (around 60°C), the thermodynamic drive towards isomerization is minimized. This mechanistic precision is what allows the process to achieve such high regioselectivity and stereochemical purity, effectively suppressing the formation of unwanted ocimene or linalool isomers.
How to Synthesize High-Purity Myrcene Efficiently
Implementing this synthesis route requires careful attention to solvent dryness and stoichiometric ratios to maximize yield and purity. The process is divided into two distinct operational phases: the protection of the starting alcohol and the subsequent elimination to form the diene. Operators must ensure that the THF solvent is anhydrous to prevent hydrolysis of the intermediate or deactivation of the strong base. The reaction progress is conveniently monitored via TLC, allowing for real-time adjustments to reaction times. While the laboratory scale examples demonstrate excellent results, scaling this process requires maintaining efficient mixing and temperature control during the exothermic protection step. The detailed standardized synthetic steps for replicating this high-efficiency protocol are outlined in the guide below.
- Protect the hydroxyl group of geraniol using 3,4-dihydro-2H-pyran (DHP) and p-toluenesulfonic acid (PTS) in THF to form a stable THP ether intermediate.
- Perform an elimination reaction on the intermediate using potassium tert-butoxide (t-BuOK) and 18-crown-6 in anhydrous THF at moderate temperatures (60°C).
- Purify the crude reaction mixture via silica gel column chromatography to isolate high-purity myrcene with minimal isomeric impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers profound strategic advantages beyond mere technical performance. By eliminating the dependency on volatile noble metal markets and complex recovery infrastructure, companies can stabilize their cost structures and reduce exposure to raw material price fluctuations. The simplified purification process, which relies on standard silica gel chromatography rather than high-vacuum distillation, translates directly into lower capital expenditure (CAPEX) for equipment and reduced operational expenditure (OPEX) for energy. Furthermore, the absence of heavy metals simplifies regulatory compliance and waste disposal, accelerating the time-to-market for new products derived from this intermediate.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of expensive palladium or platinum catalysts from the bill of materials. In traditional processes, the cost of these metals and the specialized ligands required can constitute a significant portion of the total production cost. By substituting these with inexpensive organic reagents like t-BuOK and DHP, the variable cost per kilogram of myrcene is drastically reduced. Additionally, the mild reaction conditions eliminate the need for high-pressure autoclaves and the associated safety systems, further lowering the depreciation costs of the manufacturing facility and reducing energy consumption for heating and cooling cycles.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of commodity chemicals that are readily available in the global market. Reagents such as tetrahydrofuran, potassium tert-butoxide, and dihydropyran are produced at massive scales for various industries, ensuring a stable supply even during market disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the inputs for this process can be sourced from multiple vendors, mitigating the risk of production stoppages. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of downstream pharmaceutical and fragrance clients.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The absence of heavy metal waste streams removes the burden of complex wastewater treatment and hazardous sludge disposal, which are often major bottlenecks in scaling up fine chemical processes. The mild operating temperatures and atmospheric pressure conditions reduce the risk of thermal runaway incidents, enhancing overall plant safety. These factors collectively make the technology highly scalable, allowing manufacturers to ramp up production from pilot batches to multi-ton commercial volumes with minimal regulatory friction and environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these nuances is essential for R&D teams evaluating the technology for potential licensing or internal development.
Q: Why is the THP protection strategy superior to direct acid-catalyzed dehydration for myrcene production?
A: Direct acid-catalyzed dehydration often requires harsh high-temperature and high-pressure conditions, leading to significant isomerization and polymerization side reactions. The THP protection strategy described in patent CN111454114A allows for mild reaction conditions that preserve the double bond geometry, resulting in significantly higher chemical purity (>99%) and eliminating the need for complex distillation purification.
Q: Does this synthesis method involve expensive noble metal catalysts?
A: No, this process is explicitly designed to be metal-free. Unlike traditional industrial methods that may rely on palladium or other noble metals which pose recovery challenges and contamination risks, this route utilizes organic bases (t-BuOK) and phase transfer catalysts (18-crown-6), drastically reducing raw material costs and environmental hazards associated with heavy metal waste.
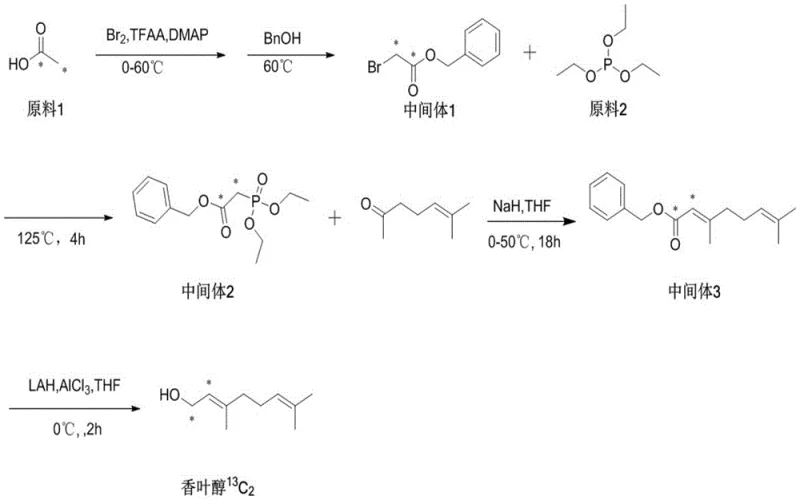
Q: Is this method applicable for synthesizing isotopically labeled myrcene?
A: Yes, the patent specifically highlights the versatility of this route for synthesizing 13C and 14C labeled myrcene. The mild conditions prevent the scrambling of isotopic labels that can occur under harsh thermal cracking conditions, making it an ideal method for producing high-specific-activity tracers for metabolic studies and ADME profiling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Myrcene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the metal-free synthesis route described in CN111454114A for the production of high-value terpene derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of batch size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise quantification of isomeric impurities and residual solvents, guaranteeing that every shipment meets the exacting standards required by the global fragrance and pharmaceutical sectors.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Whether you require custom synthesis of isotopically labeled standards for metabolic research or bulk quantities of high-purity myrcene for fragrance formulation, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and accelerate your product development timelines.
