Advanced Metal-Free Synthesis of High-Purity Myrcene for Commercial Scale-Up
The global demand for high-purity terpenes, particularly myrcene, continues to surge across the fragrance and pharmaceutical sectors, driving the need for more efficient and sustainable manufacturing processes. Patent CN111454114A introduces a groundbreaking methodology for synthesizing high-purity myrcene from geraniol, specifically addressing the limitations of traditional dehydration techniques. This innovation utilizes a strategic protection-deprotection sequence involving tetrahydropyranyl (THP) groups, followed by a mild base-catalyzed elimination, effectively bypassing the harsh conditions typically required for allylic alcohol dehydration. By shifting away from energy-intensive high-pressure reactors and expensive noble metal catalysts, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains for critical fragrance intermediates like citronellal and lyral precursors.
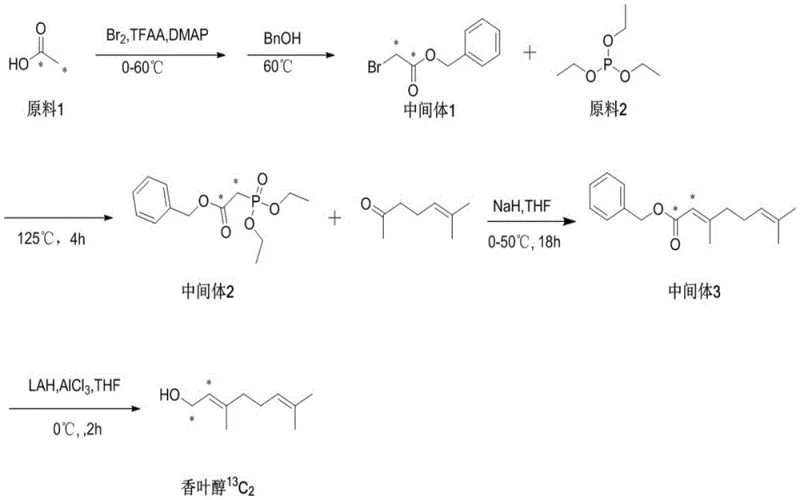
Furthermore, the versatility of this synthetic route extends beyond standard commercial grades, demonstrating exceptional efficacy in the production of isotopically labeled compounds such as 13C2-myrcene and 14C2-myrcene. The ability to maintain high isotopic abundance while achieving chemical purities exceeding 99% positions this method as a dual-purpose solution for both bulk industrial applications and specialized research markets. For procurement strategists and R&D directors alike, understanding the mechanistic advantages of this metal-free approach is essential for evaluating long-term sourcing stability and cost-efficiency in the competitive landscape of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial conversion of geraniol to myrcene has relied heavily on direct acid-catalyzed dehydration or high-temperature thermal cracking processes, both of which suffer from significant thermodynamic and kinetic drawbacks. Conventional acid dehydration often necessitates rigorous conditions involving high temperatures and pressures to overcome activation energy barriers, which inadvertently promotes extensive isomerization side reactions. These side reactions generate a complex mixture of terpene isomers, drastically reducing the yield of the desired (E)-myrcene and complicating downstream purification efforts. Moreover, alternative catalytic routes employing noble metals like palladium, while effective at lowering activation energies, introduce substantial cost burdens due to the high price of the catalysts and the stringent regulatory requirements for removing trace metal residues from final products intended for fragrance or pharmaceutical use.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN111454114A employs a sophisticated two-step strategy that prioritizes selectivity and operational simplicity over brute-force reaction conditions. The process begins by converting the hydroxyl group of geraniol into a tetrahydropyranyl (THP) ether, a robust protecting group that stabilizes the molecule against premature rearrangement. Subsequent treatment with a strong base, specifically potassium tert-butoxide in the presence of 18-crown-6, facilitates a clean E2 elimination reaction at a moderate temperature of 60°C. This mild thermal profile not only conserves energy but also virtually eliminates the formation of unwanted isomeric byproducts, resulting in a crude product of such high quality that simple silica gel chromatography suffices for final purification, thereby streamlining the entire production workflow.
Mechanistic Insights into THP-Protection and Base-Catalyzed Elimination
The core innovation of this synthesis lies in the strategic manipulation of the geraniol substrate to control regioselectivity during the elimination phase. By first reacting geraniol with 3,4-dihydro-2H-pyran (DHP) in the presence of p-toluenesulfonic acid, the primary allylic alcohol is converted into a THP ether intermediate. This protection step is crucial because it prevents the acid-catalyzed allylic rearrangements that typically plague direct dehydration attempts. The subsequent elimination step utilizes potassium tert-butoxide as a non-nucleophilic base, which abstracts a proton from the gamma-position relative to the leaving group. The presence of 18-crown-6 acts as a phase-transfer catalyst equivalent, complexing with the potassium cation to enhance the nucleophilicity and solubility of the tert-butoxide anion in the organic solvent, ensuring a rapid and complete conversion to the conjugated diene system of myrcene.
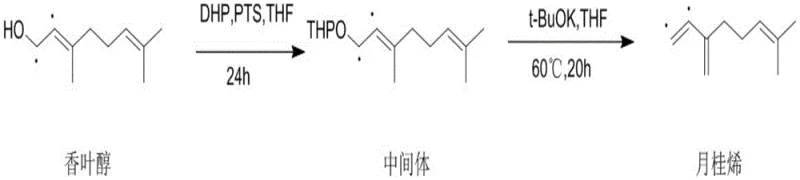
This mechanistic pathway ensures that the double bond migration is strictly controlled, preserving the structural integrity of the carbon skeleton. In the context of isotopically labeled synthesis, as depicted in the precursor preparation pathways, this level of control is paramount to prevent the scrambling of isotopic labels which would render the final tracer useless for metabolic tracking. The absence of transition metals further guarantees that the electronic environment of the double bonds remains unperturbed by coordination complexes, leading to a cleaner reaction profile. The result is a process that delivers chemical purities of up to 99.81%, as evidenced in the experimental examples, with minimal formation of the thermodynamically stable but undesired ocimene isomers.
How to Synthesize High-Purity Myrcene Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and reaction atmosphere to maximize yield and safety. The process initiates with the dissolution of the geraniol substrate in anhydrous tetrahydrofuran, followed by the addition of the protecting group reagents under ambient conditions. Once the intermediate is isolated, the critical elimination step demands an inert nitrogen atmosphere to prevent moisture interference with the strong base. Detailed standardized operating procedures regarding reagent grades, addition rates, and workup protocols are essential for reproducibility at scale. For a comprehensive breakdown of the specific molar ratios, temperature ramps, and purification gradients required to achieve the reported 84.5% yield, please refer to the technical guide below.
- Dissolve geraniol in anhydrous THF, add 3,4-dihydro-2H-pyran and p-toluenesulfonic acid, and stir to form the THP-protected intermediate.
- React the intermediate with potassium tert-butoxide and 18-crown-6 in THF under nitrogen protection at 60°C to induce elimination.
- Purify the crude reaction mixture via silica gel column chromatography to isolate high-purity myrcene with >99% chemical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this metal-free synthesis route offers transformative benefits that extend far beyond simple yield improvements. By eliminating the dependency on volatile noble metal markets, manufacturers can stabilize their raw material costs and insulate themselves from geopolitical supply shocks affecting palladium and platinum availability. Furthermore, the mild reaction conditions significantly reduce the energy footprint of the manufacturing process, aligning with increasingly stringent global sustainability mandates and reducing utility overheads. The simplification of the purification train, moving from complex distillation columns to standard chromatographic or crystallization techniques, also lowers capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of expensive noble metal catalysts from the bill of materials. Traditional methods relying on palladium complexes incur high recurring costs not just for the initial purchase, but also for the specialized ligands and the rigorous metal scavenging steps required to meet regulatory limits. By substituting these with commodity chemicals like potassium tert-butoxide and DHP, the variable cost per kilogram of myrcene is drastically reduced. Additionally, the high selectivity of the reaction minimizes waste generation, meaning less raw material is lost to byproduct streams, further enhancing the overall economic efficiency of the process.
- Enhanced Supply Chain Reliability: The reagents utilized in this novel pathway, including tetrahydrofuran, p-toluenesulfonic acid, and crown ethers, are widely available commodity chemicals with robust global supply chains. This stands in contrast to specialized catalysts which may have limited suppliers and long lead times. The operational simplicity of the process, which does not require high-pressure autoclaves or exotic reactor materials, allows for greater flexibility in manufacturing site selection. This decentralization potential reduces logistical risks and ensures a more consistent supply of high-purity myrcene to downstream customers in the fragrance and pharmaceutical industries.
- Scalability and Environmental Compliance: From an environmental health and safety (EHS) perspective, this process offers a significantly greener profile. The absence of heavy metals eliminates the need for complex wastewater treatment protocols designed to remove toxic metal ions, thereby reducing effluent disposal costs. The mild operating temperatures reduce the risk of thermal runaway incidents, enhancing plant safety. Moreover, the high purity of the crude product reduces the solvent consumption associated with extensive recrystallization or fractional distillation steps. These factors collectively make the technology highly scalable, allowing producers to ramp up from pilot batches to multi-ton commercial production with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Why is the THP protection method superior to direct acid dehydration for myrcene synthesis?
A: Direct acid dehydration often requires harsh high-temperature and high-pressure conditions, leading to significant isomerization side reactions and lower purity. The THP protection method described in patent CN111454114A allows for mild reaction conditions (60°C) that prevent skeletal rearrangement, ensuring high selectivity for the target (E)-myrcene isomer.
Q: Does this synthesis route require expensive noble metal catalysts?
A: No, this novel process completely eliminates the need for expensive noble metal catalysts such as palladium. By utilizing a base-catalyzed elimination strategy with potassium tert-butoxide, the method significantly reduces raw material costs and avoids the complex environmental and safety issues associated with heavy metal recovery and disposal.
Q: Is this method applicable for producing isotopically labeled myrcene?
A: Yes, the patent explicitly demonstrates the efficacy of this route for synthesizing 13C2-labeled and 14C2-labeled myrcene. The mild conditions preserve the isotopic label integrity, making it highly suitable for producing tracers used in metabolic studies and specialized fragrance research.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Myrcene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111454114A are fully realized in practical application. Our state-of-the-art facilities are equipped to handle the specific requirements of this metal-free process, maintaining stringent purity specifications through our rigorous QC labs to guarantee that every batch meets the exacting standards of the global fragrance and pharmaceutical markets.
We invite you to collaborate with us to leverage this innovative technology for your specific product needs. Whether you require standard high-purity myrcene or specialized isotopically labeled variants for research purposes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized supply chain can drive value for your organization.
