Advanced Heavy-Metal-Free Synthesis of Bortezomib for Commercial Scale-Up
Advanced Heavy-Metal-Free Synthesis of Bortezomib for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and compliant synthetic routes for critical oncology therapeutics. Patent CN103897028A introduces a transformative synthesis method for Bortezomib, a potent proteasome inhibitor used in the treatment of multiple myeloma. This technology represents a significant departure from traditional methodologies by eliminating the reliance on heavy metal catalysts and complex purification sequences. For R&D directors and procurement specialists, this patent offers a compelling solution to the persistent challenges of residual metal contamination and high production costs. By leveraging a chiral pinanediol protection strategy combined with straightforward condensation reactions, the process achieves exceptional optical purity and chemical integrity. The following analysis dissects the technical merits of this innovation, highlighting its potential to redefine supply chain reliability for high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Bortezomib has been plagued by significant technical bottlenecks that hinder efficient commercial scale-up. Many existing patents, such as CN 101812026 A, rely heavily on palladium-on-carbon (Pd/C) catalytic hydrogenation for deprotection steps. While chemically effective, this approach introduces severe regulatory risks regarding heavy metal residues, which must be maintained below stringent limits of less than 1ppm according to latest pharmacopeia standards. Furthermore, these conventional routes often necessitate the use of hazardous pressurized hydrogen, creating substantial safety liabilities in large-scale reactors. Beyond safety and compliance, the economic burden is exacerbated by the frequent requirement for column chromatography purification, a technique that is notoriously difficult to scale and generates excessive solvent waste. These factors collectively result in inflated production costs and extended lead times, rendering many legacy processes unsuitable for modern, cost-sensitive pharmaceutical manufacturing environments.
The Novel Approach
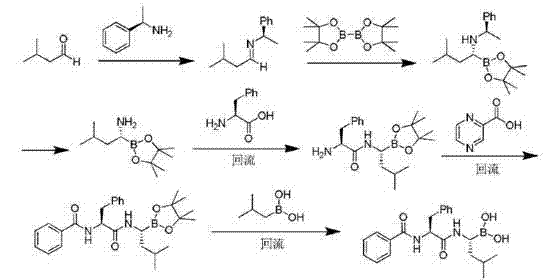
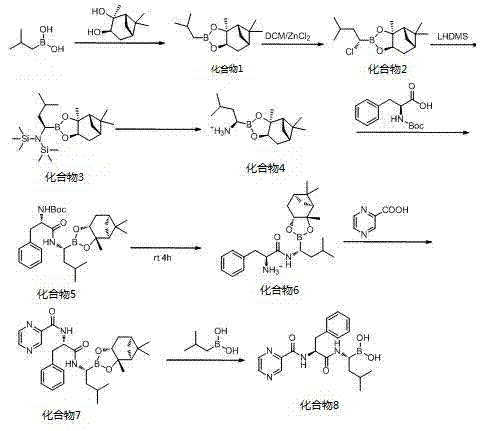
In stark contrast to these legacy methods, the methodology disclosed in CN103897028A presents a streamlined, heavy-metal-free alternative that prioritizes both safety and efficiency. The core innovation lies in the utilization of isobutylboronic acid and (1S,2S,3R,5S)-(+)-2,3-pinanediol as起始 materials to construct the chiral boronic acid ester framework. This route strategically bypasses the need for transition metal catalysts entirely, thereby eliminating the risk of palladium or copper contamination at the source. The process employs mild reaction conditions, including ambient temperature esterification and controlled low-temperature substitutions, which significantly reduce energy consumption and equipment stress. Moreover, the purification strategy has been radically simplified; instead of labor-intensive column chromatography, the final product is isolated through simple crystallization and extraction. This shift not only drastically reduces solvent usage and waste generation but also enhances the overall yield, reportedly achieving up to 82% with chemical purity exceeding 99.5%. Such improvements make this route exceptionally viable for reliable API intermediate supplier operations aiming for green chemistry compliance.
Mechanistic Insights into Boronic Acid Ester Protection and Coupling
The mechanistic elegance of this synthesis rests on the stability and stereoselectivity provided by the pinanediol protecting group. The initial formation of the isobutylboronic acid ester creates a rigid chiral environment that directs subsequent nucleophilic substitutions with high fidelity. The process involves a chlorination step followed by an ammonia substitution, where the steric bulk of the pinanediol moiety prevents racemization, ensuring the retention of the critical (R)-configuration at the boron-bearing carbon. Following the introduction of the amine functionality, the trimethylsilyl protecting group is removed under acidic conditions to reveal the reactive amine intermediate. This intermediate then undergoes peptide coupling with Boc-L-phenylalanine using standard coupling agents like TBTU or EDCI. The final assembly involves the condensation with pyrazine-2-carboxylic acid, followed by the hydrolytic removal of the pinanediol group. This final deprotection step is particularly noteworthy as it proceeds smoothly under mild acidic or hydrolytic conditions, releasing the free boronic acid without degrading the sensitive peptide backbone. The entire sequence is designed to minimize side reactions, resulting in a clean impurity profile that simplifies downstream processing.
From an impurity control perspective, the absence of heavy metals fundamentally alters the purification landscape. In traditional routes, metal scavengers and extensive washing protocols are required to meet regulatory specs, often leading to product loss. Here, the primary impurities are organic byproducts from the coupling steps, which are effectively removed through the optimized crystallization protocols described in the embodiments. The use of specific solvent systems, such as ethyl acetate and isopropyl ether mixtures, allows for the selective precipitation of the target molecule while keeping soluble impurities in the mother liquor. This level of control over the solid-state properties of the intermediate and final product is crucial for ensuring batch-to-batch consistency, a key metric for any commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Bortezomib Efficiently
The synthesis of Bortezomib via this patented route involves a logical sequence of eight distinct operational steps, beginning with the protection of isobutylboronic acid and culminating in the final hydrolytic deprotection. The process is designed to be operationally simple, utilizing common organic solvents and reagents that are readily available in standard chemical inventories. Key to the success of this method is the strict control of temperature during the lithiation and substitution phases to maintain stereochemical integrity. Detailed standard operating procedures for each stage, including specific molar ratios and workup protocols, are essential for replicating the high yields reported in the patent literature. For a comprehensive breakdown of the reaction parameters and isolation techniques, please refer to the standardized synthesis guide below.
- Esterification of isobutylboronic acid with (1S,2S,3R,5S)-(+)-2,3-pinanediol to form the protected boronic acid ester.
- Chlorination and subsequent ammonia substitution to introduce the amino group, followed by deprotection to yield the key amine intermediate.
- Condensation with Boc-L-phenylalanine, deprotection, and final coupling with pyrazine-2-carboxylic acid, concluding with hydrolysis to obtain Bortezomib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic advantages that extend beyond mere technical feasibility. The elimination of expensive and hazardous reagents directly translates to a more resilient and cost-effective supply chain. By removing the dependency on palladium catalysts, manufacturers can avoid the volatility associated with precious metal pricing and the logistical complexities of sourcing high-purity catalytic grades. Furthermore, the simplification of the purification process reduces the consumption of chromatographic silica and vast quantities of organic solvents, leading to significant waste disposal cost savings. These operational efficiencies contribute to a lower overall cost of goods sold (COGS), allowing for more competitive pricing in the global API market without compromising on quality standards.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of heavy metal catalysts and the associated purification burdens. Traditional methods require expensive metal scavengers and rigorous testing to ensure compliance with residual metal limits, adding layers of cost and time to the production cycle. By adopting a heavy-metal-free pathway, manufacturers can eliminate these specific line items entirely. Additionally, the replacement of column chromatography with crystallization significantly lowers solvent consumption and labor hours. Crystallization is inherently more scalable and requires less specialized equipment than chromatography, facilitating a drastic simplification of the manufacturing infrastructure. This structural change in the process flow ensures that cost reduction in pharmaceutical manufacturing is achieved through fundamental process intensification rather than marginal tweaks.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized or hazardous reagents. The proposed method utilizes isobutylboronic acid and pinanediol, which are stable, commercially available commodities with robust supply networks. Unlike pressurized hydrogenation which requires specialized high-pressure reactors and strict safety protocols, this route operates under atmospheric pressure and mild temperatures. This reduces the risk of production stoppages due to equipment maintenance or safety audits. The mild reaction conditions also broaden the range of suitable manufacturing partners, as the process does not demand exotic containment facilities. Consequently, reducing lead time for high-purity API intermediates becomes achievable through a more flexible and accessible manufacturing base, ensuring steady availability for downstream drug formulation.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, the green chemistry attributes of this synthesis become a major asset. The avoidance of heavy metals aligns perfectly with global initiatives to reduce toxic waste in pharmaceutical production. The process generates significantly less hazardous waste compared to metal-catalyzed routes, simplifying effluent treatment and lowering environmental compliance costs. The high yield and purity achieved through simple crystallization mean that less raw material is wasted per unit of product, enhancing the overall atom economy. This scalability ensures that the transition from pilot plant to multi-ton commercial production can be executed smoothly, supporting the growing global demand for oncology treatments without encountering the bottlenecks typical of complex, multi-step syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term viability of the supply source.
Q: How does this synthesis method address heavy metal residue concerns?
A: Unlike conventional methods utilizing palladium-on-carbon hydrogenation, this patent describes a route completely free of heavy metal catalysts, ensuring compliance with strict pharmacopeia limits (<1ppm) without expensive purification steps.
Q: What purification techniques are employed to ensure high purity?
A: The process eliminates the need for column chromatography, relying instead on simple crystallization and extraction techniques to achieve chemical purity levels exceeding 99.5%.
Q: Is this route suitable for large-scale industrial production?
A: Yes, the method utilizes easily available raw materials, mild reaction conditions, and avoids hazardous pressurized hydrogenation, making it highly scalable and environmentally friendly for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bortezomib Supplier
The technical advancements detailed in CN103897028A underscore the immense potential for optimizing the production of critical cancer therapeutics. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated chemistry to life. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Bortezomib meets the highest international standards. We understand that the transition to a new synthetic route requires a partner who can navigate the complexities of process validation and regulatory filing with precision and expertise.
We invite you to collaborate with us to leverage this innovative synthesis method for your supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this heavy-metal-free route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can secure a sustainable and high-quality supply of this vital oncology intermediate.
