Industrial Scale-Up of Apixaban Intermediate APM07 via Safe Phase Transfer Catalysis
Introduction to Advanced Apixaban Intermediate Manufacturing
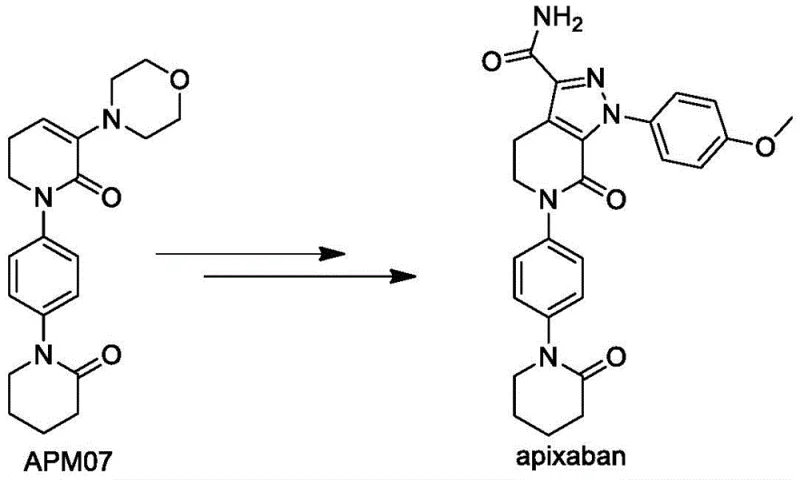
The global demand for direct oral anticoagulants continues to surge, placing immense pressure on the supply chains of critical pharmaceutical intermediates. Among these, the synthesis of Apixaban intermediate APM07 represents a significant technological bottleneck due to the complexity of constructing the fused heterocyclic core while maintaining high purity standards. Patent CN113264870A, published in August 2021, introduces a groundbreaking preparation method specifically designed for industrial production that addresses these challenges through innovative process engineering. This technology leverages a robust phase transfer catalytic system to facilitate efficient amidation and cyclization reactions, effectively bypassing the limitations of traditional routes that rely on hazardous reagents and expensive starting materials. By shifting the synthetic paradigm towards safer inorganic bases and streamlined one-pot operations, this method offers a viable pathway for reliable pharmaceutical intermediates supplier networks to enhance their production capabilities. The strategic implementation of this protocol not only ensures consistent quality but also aligns with modern green chemistry principles by reducing waste generation and energy consumption throughout the manufacturing lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Apixaban intermediates has been plagued by reliance on costly and potentially dangerous chemical transformations that hinder large-scale adoption. Prior art, such as the methodologies described in WO03049681 and US2003181466, typically utilizes p-iodoaniline as a primary raw material, which is significantly more expensive than nitro-based alternatives and introduces iodine impurities that are difficult to remove. Furthermore, these conventional routes often necessitate the use of sodium hydride (NaH), a pyrophoric reagent that poses severe safety risks during handling and requires specialized equipment for containment and quenching. The coupling reactions in these older processes frequently employ expensive copper catalysts and cesium carbonate, driving up the overall cost of goods sold and complicating the purification landscape. Additionally, the multi-step isolation procedures required between each transformation lead to substantial yield losses and increased solvent consumption, making cost reduction in pharmaceutical intermediates manufacturing nearly impossible under these legacy frameworks. The harsh reaction conditions and violent exotherms associated with these methods further limit their scalability, creating supply chain vulnerabilities for downstream API producers who require consistent, high-volume delivery.
The Novel Approach
In stark contrast, the novel approach detailed in CN113264870A revolutionizes the synthesis landscape by adopting p-nitroaniline as a cost-effective starting material, thereby eliminating the dependency on premium iodine-containing reagents. This innovative route employs a phase transfer catalytic amidation reaction in a biphasic organic-aqueous system, allowing for precise control over reaction kinetics under mild inorganic alkaline conditions. By utilizing sodium hydroxide instead of sodium hydride for the cyclization steps, the process inherently enhances operational safety and simplifies the engineering controls required for industrial reactors. The methodology integrates a seamless one-pot cyclization strategy that converts the initial amidation product directly into the cyclic intermediate without the need for intermediate isolation, drastically reducing processing time and solvent waste. Moreover, the subsequent chlorination and elimination steps are optimized to proceed in the same organic phase, minimizing material transfer and exposure to atmospheric moisture. This holistic redesign of the synthetic pathway ensures that the production of high-purity pharmaceutical intermediates is not only economically feasible but also environmentally sustainable and operationally robust for commercial scale-up of complex molecules.
Mechanistic Insights into Phase Transfer Catalytic Amidation and Cyclization
The core of this technological advancement lies in the sophisticated application of phase transfer catalysis (PTC) to drive the initial amidation between p-nitroaniline and 5-chlorovaleryl chloride. In this biphasic system, a quaternary ammonium salt, specifically tetrabutylammonium bromide, acts as the molecular shuttle, transporting the deprotonated aniline species from the aqueous phase into the organic phase where the acyl chloride resides. This mechanism significantly accelerates the reaction rate at low temperatures, typically maintained between -5°C and 10°C, preventing thermal degradation and suppressing side reactions such as hydrolysis of the acid chloride. The use of weak inorganic bases like sodium carbonate in this initial stage ensures gentle deprotonation, preserving the integrity of the nitro group while facilitating nucleophilic attack on the carbonyl carbon. Following the formation of the linear amide APM01, the addition of a strong base like sodium hydroxide triggers an intramolecular nucleophilic substitution, closing the ring to form the dihydropyridinone core of APM02. This tandem sequence exemplifies how careful manipulation of pH and phase dynamics can achieve high conversion rates without the need for exotic catalysts or extreme thermal inputs.
Impurity control is meticulously managed through the strategic sequencing of the chlorination and elimination steps involving phosphorus pentachloride and morpholine. The activation of the alpha-position of the ketone in APM02 via dichlorination with PCl5 generates a highly reactive intermediate APM03, which is immediately subjected to nucleophilic attack by excess morpholine. This condensation-elimination cascade is critical for installing the morpholine moiety while simultaneously establishing the double bond character required for the final aromatic system. The process includes a simple acid washing and liquid separation protocol that effectively removes inorganic byproducts and unreacted amines, ensuring that the resulting APM04 solid possesses high chemical purity prior to the reduction step. The subsequent reduction of the nitro group to an amino group using sodium sulfide is conducted in a water-soluble organic solvent system, which facilitates easy precipitation of the product APM05 upon solvent removal. This rigorous attention to mechanistic detail at every stage guarantees that the final intermediate APM07 meets the stringent purity specifications required for downstream API synthesis, minimizing the risk of genotoxic impurities or heavy metal contamination.
How to Synthesize Apixaban Intermediate APM07 Efficiently
The execution of this synthesis protocol requires precise adherence to the defined stoichiometric ratios and temperature profiles to maximize yield and safety. The process begins with the preparation of a two-phase reaction mixture containing p-nitroaniline, an organic solvent such as dichloromethane, and an aqueous solution of sodium carbonate and tetrabutylammonium bromide. Upon cooling this mixture to sub-zero temperatures, 5-chlorovaleryl chloride is added dropwise to control the exotherm, followed by the introduction of sodium hydroxide to effect cyclization. The resulting organic layer is dried and treated with phosphorus pentachloride under reflux, then quenched and reacted with morpholine to isolate APM04. Finally, the nitro group is reduced, and the final ring closure is performed to yield the target molecule. For the complete technical operating procedures and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Perform phase transfer catalytic amidation of p-nitroaniline with 5-chlorovaleryl chloride in a two-phase system to obtain APM01, followed by in-situ cyclization with sodium hydroxide to yield APM02.
- Activate the APM02 organic solution with phosphorus pentachloride to form APM03, then react with excess morpholine to induce condensation-elimination and crystallize APM04.
- Reduce APM04 to APM05 using sodium sulfide, followed by a final amidation-cyclization one-pot reaction with 5-chlorovaleryl chloride to isolate the key intermediate APM07.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative benefits that directly address the pain points of procurement managers and supply chain directors in the fine chemical sector. By fundamentally altering the raw material input profile, the process decouples production costs from the volatile pricing of iodine-based reagents and precious metal catalysts. The elimination of hazardous reagents like sodium hydride not only reduces insurance and safety compliance costs but also simplifies the logistics of raw material storage and handling. Furthermore, the streamlined workup procedures, which avoid multiple concentration and recrystallization steps, lead to significant reductions in utility consumption and waste disposal fees. These operational efficiencies translate into a more resilient supply chain capable of meeting tight delivery schedules without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The substitution of expensive p-iodoaniline with readily available p-nitroaniline serves as the primary driver for raw material cost optimization, removing the financial burden associated with halogenated aromatic precursors. Additionally, the removal of copper catalysts and cesium carbonate eliminates the need for expensive metal scavenging processes and complex wastewater treatment protocols required to meet heavy metal limits. The one-pot nature of the amidation-cyclization sequence reduces solvent turnover and labor hours, contributing to a leaner manufacturing cost structure. Consequently, this approach enables substantial cost savings in pharmaceutical intermediates manufacturing without sacrificing the chemical integrity of the final product.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as sodium hydroxide, sodium carbonate, and morpholine ensures that the supply chain is not vulnerable to the shortages often experienced with specialized reagents. The robustness of the phase transfer catalytic system allows for flexible production scheduling, as the reaction tolerates minor variations in conditions better than sensitive organometallic couplings. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on schedule. The simplified purification train also means that batch release times are shortened, accelerating the overall velocity of the supply chain from reactor to shipment.
- Scalability and Environmental Compliance: The avoidance of pyrophoric reagents and the use of aqueous workups make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduction in solvent usage and the elimination of heavy metal catalysts significantly lower the environmental footprint, facilitating easier permitting and compliance with increasingly strict environmental regulations. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet market surges. Moreover, the generation of less hazardous waste streams simplifies disposal logistics and reduces the long-term liability associated with chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and industry best practices. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new method for their production lines. The answers provided reflect the specific advantages and operational parameters defined in the intellectual property documentation.
Q: Why is the new synthesis route for APM07 considered safer than conventional methods?
A: The novel process eliminates the use of highly dangerous reagents such as sodium hydride (NaH) and expensive iodine-containing organic compounds found in prior art. Instead, it utilizes safer inorganic bases like sodium hydroxide and sodium carbonate under mild phase transfer catalytic conditions, significantly reducing operational hazards.
Q: How does this method improve production efficiency for Apixaban intermediates?
A: By employing one-pot amidation-cyclization strategies and avoiding intermediate purification steps between the formation of APM02 and APM03, the process drastically reduces unit operations. This streamlined approach minimizes solvent usage, shortens cycle times, and improves overall yield compared to multi-step isolation methods.
Q: What represents the key cost-saving advantage in this manufacturing protocol?
A: The substitution of expensive p-iodoaniline with cost-effective p-nitroaniline as the starting material, combined with the removal of precious metal catalysts like copper complexes, results in substantial raw material cost reductions. Additionally, the simplified workup procedures lower utility and waste treatment expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apixaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technological potential of patent CN113264870A and is fully prepared to implement this safer, more efficient protocol for the production of APM07. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for API synthesis.
We invite you to collaborate with us to leverage these process innovations for your specific project requirements. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this new route for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, cost-effective supply of high-quality Apixaban intermediates that will strengthen your position in the anticoagulant market.
