Advanced Synthesis of Thiophene-Based Herbicide Intermediates for Commercial Scale-Up
The chemical landscape for agrochemical intermediates is constantly evolving, driven by the need for more efficient and scalable synthetic routes. Patent CN85106474A introduces a significant advancement in the production of N-(thiophen-3-yl)-chloroacetamide, a critical building block for herbicide formulations. This technology addresses long-standing challenges in thiophene chemistry by utilizing a novel tetrahydrothiophene-3-subunit imine compound as a key precursor. The methodology described offers a robust pathway that begins with accessible raw materials and proceeds through a series of optimized transformations, including cyclization and dehydrogenation. For R&D directors and procurement specialists, understanding this patent is crucial as it represents a shift towards more cost-effective and reliable manufacturing processes for complex sulfur-containing heterocycles. The ability to synthesize these structures without relying on scarce reagents positions this technology as a vital asset for securing long-term supply chains in the agrochemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing thiophene-based herbicide intermediates often suffer from significant drawbacks related to raw material availability and process complexity. Many existing routes rely on starting materials that are difficult to source commercially or require multi-step preparations that drastically increase the overall cost of goods. Furthermore, conventional dehydrogenation techniques frequently necessitate harsh reaction conditions or expensive noble metal catalysts that are difficult to recover and reuse efficiently. These factors contribute to inconsistent batch quality and extended lead times, creating bottlenecks for supply chain managers who require steady volumes of high-purity intermediates. The reliance on complex purification steps to remove side products further diminishes the overall yield, making large-scale production economically challenging for many manufacturers.
The Novel Approach
The approach detailed in the patent overcomes these hurdles by introducing a streamlined sequence that begins with the addition reaction of thiolactic acid and methacrylic acid derivatives. This foundational step utilizes commodity chemicals that are readily available in the global market, thereby stabilizing the supply chain against raw material fluctuations. The subsequent cyclization to form the tetrahydrothiophene ring is facilitated by common metal catalysts such as iron powder or acetates, which are far more economical than specialized reagents. This novel pathway not only simplifies the operational workflow but also enhances the overall atom economy of the process. By enabling the direct conversion of intermediates without rigorous isolation steps, the method significantly reduces processing time and solvent consumption, aligning with modern green chemistry principles while maintaining high commercial viability.
Mechanistic Insights into Catalytic Dehydrogenation and Cyclization
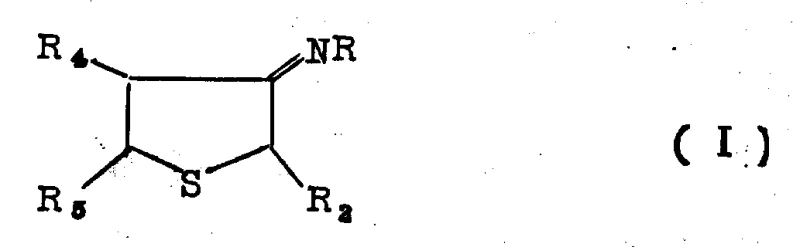
The core of this synthetic innovation lies in the efficient transformation of the tetrahydrothiophene-3-subunit imine into the corresponding aminothiophene derivative. This dehydrogenation step is critical for establishing the aromatic character of the thiophene ring, which is essential for the biological activity of the final herbicide. The patent discloses that this transformation can be achieved through catalytic means using precious metals like platinum or palladium distributed on carriers such as charcoal. Alternatively, chemical oxidation using agents like thionyl chloride or sulfur provides a versatile option that can be tuned based on specific production requirements. The mechanism involves the removal of hydrogen atoms from the saturated ring system, a process that is carefully controlled to prevent over-oxidation or chlorination side reactions. This precision ensures that the resulting aminothiophene retains the necessary structural integrity for subsequent functionalization.
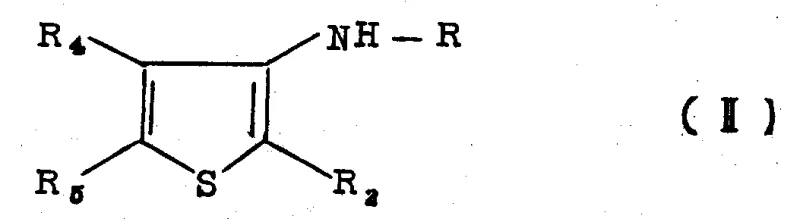
Impurity control is another vital aspect of this mechanism, particularly given the sensitivity of sulfur-containing compounds to thermal degradation. The process utilizes specific reaction conditions, such as maintaining temperatures above 180°C for catalytic dehydrogenation or utilizing inert gas atmospheres to prevent unwanted oxidation. When using thionyl chloride, the reaction can proceed at milder temperatures, reducing the thermal load on the system and minimizing the formation of tarry by-products. The ability to form the amine as a hydrochloride salt directly in the reaction mixture allows for easy purification through simple washing steps, effectively removing inorganic impurities and unreacted starting materials. This level of control over the reaction environment ensures that the final product meets the stringent purity specifications required for agrochemical applications, reducing the burden on downstream quality control laboratories.
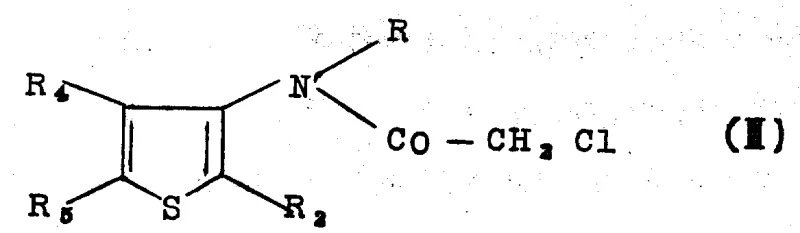
How to Synthesize N-(Thiophen-3-yl)-chloroacetamide Efficiently
The synthesis of this high-value herbicide intermediate follows a logical progression from simple acid derivatives to the complex chloroacetamide target. The process begins with the condensation of thiolactic acid and methacrylic acid, followed by cyclization to form the ketone precursor. This ketone is then converted into the imine derivative, which serves as the substrate for the critical dehydrogenation step. Once the aminothiophene is generated, it undergoes N-chloroacetylation to yield the final product. The patent emphasizes that these steps can be telescoped, meaning that intermediates like the imine or the amine hydrochloride do not necessarily need to be isolated in pure form before proceeding to the next stage. This operational flexibility is key to maximizing throughput and minimizing waste in a commercial setting. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up.
- Condense thiolactic acid and methacrylic acid derivatives to form the hexanedioic acid precursor.
- Perform cyclization using iron powder or metal acetates to generate the tetrahydrothiophene-3-ketone.
- Convert the ketone to an imine, followed by catalytic dehydrogenation to yield the aminothiophene intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond simple chemical yield. The primary advantage lies in the stabilization of raw material costs, as the process relies on thiolactic acid and methacrylic acid, which are produced in large volumes for other industries. This reduces the risk of supply disruptions that are common with specialty reagents. Additionally, the elimination of complex separation steps for unstable intermediates translates into shorter manufacturing cycles and reduced inventory holding costs. The flexibility in catalyst choice allows production facilities to optimize for either speed or cost, depending on current market demands. These factors combine to create a more resilient supply chain capable of meeting the fluctuating demands of the global agrochemical market without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by utilizing inexpensive metal catalysts like iron powder instead of relying solely on high-cost noble metals for every step. Furthermore, the ability to perform reactions without isolating intermediate salts reduces solvent usage and energy consumption associated with drying and purification. This streamlined approach minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint. By avoiding the need for specialized equipment to handle highly unstable compounds, capital expenditure for new production lines is also kept in check, ensuring a faster return on investment for manufacturing upgrades.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved because the foundational building blocks are commodity chemicals with established global supply networks. This reduces dependency on single-source suppliers for exotic reagents, mitigating the risk of geopolitical or logistical disruptions. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without significant re-validation efforts. This consistency ensures that procurement teams can secure long-term contracts with confidence, knowing that the technical feasibility of the supply is backed by a proven and flexible chemical process.
- Scalability and Environmental Compliance: The method is inherently designed for scale-up, with reaction conditions that can be safely translated from laboratory glassware to industrial reactors. The use of common solvents like toluene and hexane simplifies solvent recovery and recycling systems, aligning with strict environmental regulations. Moreover, the reduction in side reactions means less hazardous waste is generated per kilogram of product, easing the burden on waste treatment facilities. This compliance with environmental standards is crucial for maintaining operational licenses and meeting the sustainability goals of modern agrochemical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the operational parameters and potential benefits for stakeholders considering this technology for their supply chain. Understanding these details is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary starting materials for this thiophene synthesis?
A: The process utilizes readily available thiolactic acid and methacrylic acid, avoiding the need for obscure or expensive specialty reagents often required in traditional thiophene manufacturing.
Q: How is the dehydrogenation step optimized for industrial application?
A: The patent discloses multiple dehydrogenation methods, including catalytic options using platinum on carbon or chemical oxidation using thionyl chloride, allowing flexibility based on available infrastructure and cost targets.
Q: Does this route require isolation of unstable intermediates?
A: The process allows for telescoping steps, such as converting the imine directly to the amine hydrochloride without isolation, which streamlines the workflow and reduces material loss.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(Thiophen-3-yl)-chloroacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in maintaining a competitive edge in the agrochemical industry. Our CDMO expertise allows us to adapt complex pathways like the one described in CN85106474A to meet the rigorous demands of commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of N-(thiophen-3-yl)-chloroacetamide meets the highest industry standards. By leveraging our technical capabilities, you can secure a stable supply of high-quality intermediates that drive the efficacy of your final herbicide products.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical engineering. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for this and other critical intermediates. Let us partner with you to transform innovative patent technologies into reliable commercial realities, ensuring your products reach the market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
