Scalable Photochemical Synthesis of 2-Methyl-1,4-Naphthoquinone for Commercial Vitamin K3 Production
The global demand for high-purity vitamin K3 intermediates necessitates a paradigm shift away from environmentally hazardous oxidation processes toward sustainable, green chemistry solutions. Patent CN112358389B introduces a groundbreaking preparation method for the photochemical synthesis of 2-methyl-1,4-naphthoquinone (2-MNQ), a critical precursor in the pharmaceutical and agrochemical sectors. This innovative approach leverages visible light irradiation, specifically utilizing LED technology, to drive the oxidation of 2-methylnaphthalene in the presence of an organic photosensitizer and molecular oxygen. By replacing stoichiometric heavy metal oxidants with clean photon energy, this technology addresses the dual challenges of environmental compliance and process safety that have long plagued the fine chemical industry. For multinational corporations seeking a reliable pharmaceutical intermediates supplier, this patent represents a viable pathway to decarbonize supply chains while maintaining rigorous quality standards.
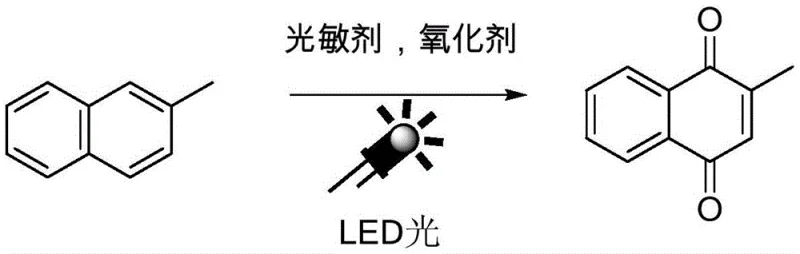
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-methyl-1,4-naphthoquinone has relied heavily on direct oxidation methods employing chromium trioxide (CrO3) in concentrated sulfuric acid media. While chemically effective, this legacy technology generates massive quantities of toxic hexavalent chromium waste, posing severe environmental liabilities and requiring expensive wastewater treatment infrastructure. Furthermore, the harsh acidic conditions often lead to over-oxidation and the formation of difficult-to-remove isomeric impurities, such as 6-methyl-1,4-naphthoquinone, which compromises the purity profile required for downstream vitamin K3 synthesis. Alternative transition metal-catalyzed routes using manganese, palladium, or ruthenium have been explored but often suffer from high catalyst costs, sensitivity to reaction conditions, and the persistent risk of heavy metal contamination in the final active pharmaceutical ingredient. These factors collectively inflate the cost reduction in vitamin K3 manufacturing and create significant bottlenecks for supply chain continuity.
The Novel Approach
In stark contrast, the novel photochemical methodology disclosed in the patent utilizes a metal-free organic photosensitizer system activated by low-energy LED light. This approach fundamentally alters the reaction landscape by employing molecular oxygen or air as the terminal oxidant, producing water as the sole byproduct and adhering strictly to green chemistry principles. The reaction proceeds under mild temperatures ranging from 10 to 50°C, drastically reducing energy consumption compared to thermal oxidation processes. Crucially, the absence of transition metals eliminates the need for costly purification steps to remove trace metal residues, thereby streamlining the post-treatment workflow. The process demonstrates exceptional tolerance to moisture and air, allowing for simplified reactor operations without the need for rigorous inert gas protection, which significantly enhances the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Visible-Light Mediated Photo-Oxidation
The core of this technological advancement lies in the efficient generation of reactive oxygen species through energy transfer mechanisms. Upon irradiation with specific wavelengths of LED light, typically optimized between 450 to 455 nm, the organic photosensitizer absorbs photon energy to reach an excited triplet state. This excited sensitizer then transfers energy to ground-state triplet oxygen, generating highly reactive singlet oxygen. This singlet oxygen species undergoes a [4+2] cycloaddition with the electron-rich 2-methylnaphthalene substrate to form a transient 1,4-endoperoxide intermediate. This key intermediate subsequently undergoes rearrangement and further oxidation to yield the target quinone structure. The selection of specific photosensitizers, such as acridine salts, eosin Y, or rhodamine, is critical as they possess high molar absorptivity in the visible region and extended excited-state lifetimes, ensuring high quantum efficiency for the transformation.
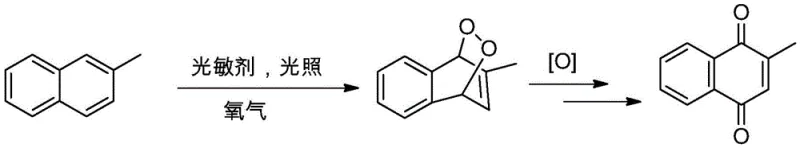
From an impurity control perspective, the selectivity of this photochemical pathway is superior to traditional radical-based oxidations. The mechanism avoids the non-selective hydrogen abstraction often seen with thermal initiators, which frequently leads to ring degradation or positional isomers. Experimental data indicates that products synthesized via this route exhibit HPLC purities exceeding 95%, with negligible detection of the 6-MNQ isomer that plagues chromium-based methods. The reaction kinetics are governed by the photon flux and the concentration of dissolved oxygen, allowing for precise tuning of the reaction rate by adjusting LED intensity and gas flow rates. This level of control ensures consistent batch-to-batch reproducibility, a critical parameter for regulatory compliance in the production of high-purity OLED material or pharmaceutical precursors.
How to Synthesize 2-Methyl-1,4-Naphthoquinone Efficiently
To implement this synthesis effectively, operators must carefully balance the molar ratio of the substrate to the photosensitizer, typically maintaining a ratio between 1:0.01 and 1:0.04 to maximize light absorption without excessive reagent cost. The choice of solvent is also pivotal; polar protic solvents like ethanol or methanol are preferred for their ability to dissolve both the organic substrate and the ionic photosensitizers while facilitating the stabilization of polar intermediates. The detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with 2-methylnaphthalene, an organic photosensitizer (such as acridine salt or eosin Y), and a suitable solvent like ethanol.
- Irradiate the mixture with LED light (preferably 450-455 nm wavelength) while stirring under an oxygen or air atmosphere at 20-30°C.
- Upon completion (20-28 hours), concentrate the reaction mixture, induce crystallization with water, and recrystallize to obtain high-purity 2-MNQ.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photochemical technology offers transformative advantages that extend beyond simple yield improvements. The elimination of hazardous chromium reagents removes a major regulatory burden and reduces the total cost of ownership associated with waste disposal and environmental remediation. Furthermore, the reliance on air or oxygen as the oxidant means that raw material costs are minimized, as these gases are ubiquitous and inexpensive compared to specialized chemical oxidants. The robustness of the reaction conditions, particularly its insensitivity to moisture, reduces the risk of batch failures due to minor deviations in operating parameters, thereby enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The most significant economic driver is the complete removal of heavy metal catalysts and stoichiometric oxidants from the process bill of materials. By avoiding the use of chromium or precious metals like palladium, the direct material costs are substantially lowered. Additionally, the simplified workup procedure, which involves merely concentrating the reaction mixture and crystallizing the product, eliminates the need for complex chromatographic purification or metal scavenging resin treatments. This streamlined downstream processing translates directly into reduced labor hours and lower utility consumption, driving significant operational expenditure savings without compromising product quality.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as ethanol, acridine salts, and atmospheric oxygen ensures that the supply chain is not vulnerable to the geopolitical or logistical disruptions often associated with specialized reagents. The process operates at near-ambient temperatures and pressures, reducing the mechanical stress on reactor equipment and minimizing maintenance downtime. This operational stability ensures consistent delivery schedules and reduces the lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market fluctuations in vitamin K3 demand.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been challenging due to light penetration limits, but the advent of high-power LED arrays and flow chemistry techniques has mitigated these issues. The modular nature of LED lighting allows for linear scale-up by increasing the irradiated surface area rather than just reactor volume. Moreover, the generation of water as the only byproduct aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against potential carbon taxes or waste discharge restrictions. This sustainability profile enhances the brand value of the final product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photochemical synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear framework for evaluating the technology's fit within existing manufacturing infrastructures.
Q: How does this photochemical method improve purity compared to traditional chromium oxidation?
A: Traditional methods using CrO3/H2SO4 often generate isomeric impurities like 6-MNQ and leave toxic heavy metal residues. The photochemical route described in patent CN112358389B achieves 95% purity with negligible 6-MNQ content, eliminating the need for complex heavy metal scavenging steps.
Q: What are the specific light source requirements for scaling this reaction?
A: The process utilizes standard visible light LEDs, specifically optimized for wavelengths between 450-455 nm (blue light). This allows for the use of commercially available, energy-efficient LED arrays rather than expensive UV lamps, facilitating easier heat management during scale-up.
Q: Is the process sensitive to moisture or air exposure?
A: Unlike many sensitive organometallic catalytic systems, this photochemical oxidation is remarkably robust. It operates effectively under ambient air conditions and tolerates the presence of moisture, significantly simplifying the operational requirements for industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-1,4-Naphthoquinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting green synthesis technologies to meet the evolving needs of the global pharmaceutical and agrochemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-methyl-1,4-naphthoquinone adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced photochemical technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our innovative manufacturing capabilities can drive value and sustainability for your organization.
