Advanced Synthesis of 1,3-Bis(2,4-Bismaleimidophenoxy)benzene for High-Performance Polyimide Manufacturing
The development of advanced high-temperature resistant materials is critical for the aerospace and electronics industries, driving the demand for superior polyimide precursors. Patent CN101250152A introduces a robust and efficient preparation method for 1,3-bis(2,4-bismaleimidophenoxy)benzene, a specialized tetramaleimide compound essential for creating next-generation thermosetting resins. This innovation addresses the growing need for materials that can withstand extreme thermal environments while maintaining structural integrity in applications ranging from aircraft components to printed circuit boards. By optimizing the reaction pathway, this technology offers a streamlined approach to synthesizing complex imide structures that were previously difficult to produce with consistent quality.
For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable polyimide precursor supplier. The patent details a method that not only achieves high yields but also simplifies the operational workflow, making it highly attractive for cost reduction in advanced composite manufacturing. The ability to produce this key intermediate with high purity and minimal environmental footprint positions it as a cornerstone for developing high-performance epoxy-tetrama resin systems. As industries push the boundaries of material science, having access to such optimized synthetic routes becomes a strategic advantage in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing tetramaleimide compounds often suffer from significant drawbacks that hinder their widespread industrial adoption. Many conventional routes require harsh reaction conditions, including high pressures and temperatures, which necessitate expensive specialized equipment and rigorous safety protocols. Furthermore, older techniques frequently struggle with incomplete cyclization, leading to products contaminated with amic acid intermediates that compromise the thermal stability of the final polymer. The use of corrosive reagents in some legacy processes also poses challenges for equipment longevity and waste management, increasing the total cost of ownership for manufacturing facilities. These inefficiencies create bottlenecks in the commercial scale-up of complex polymer additives, limiting the availability of high-quality materials for critical applications.
The Novel Approach
The method disclosed in the patent represents a paradigm shift by utilizing a mild, two-stage reaction sequence that maximizes efficiency and product quality. The initial step involves the formation of the tetra-amic acid intermediate at room temperature, which eliminates the risk of premature polymerization or side reactions often seen in heated environments. The subsequent cyclization step offers flexibility, allowing manufacturers to choose between azeotropic dehydration or chemical dehydration using acetic anhydride and tertiary amines. This adaptability ensures that the process can be tailored to specific facility capabilities while maintaining high conversion rates. By operating under atmospheric pressure and avoiding corrosive substances, this novel approach significantly lowers the barrier to entry for producing high-purity tetramaleimide, facilitating broader adoption in the electronics and aerospace sectors.
Mechanistic Insights into Imidization Cyclization
The core of this synthesis lies in the precise control of the imidization mechanism, which transforms the reactive amic acid groups into stable imide rings. The process begins with the nucleophilic attack of the amine groups on 1,3-bis(2,4-diaminophenoxy)benzene onto the carbonyl carbons of maleic anhydride. This exothermic reaction is carefully managed in polar aprotic solvents such as N,N-dimethylacetamide or N-methyl-2-pyrrolidone to ensure complete solubility and homogeneity. Maintaining the reaction at room temperature during this phase is crucial, as it prevents the formation of insoluble oligomers that could trap impurities within the crystal lattice. The resulting tetra-amic acid solution serves as a clean slate for the subsequent cyclization, setting the stage for high structural fidelity in the final product.
Following the intermediate formation, the dehydration cyclization step is executed with meticulous attention to water removal, which drives the equilibrium towards the desired imide product. Whether employing azeotropic distillation with solvents like toluene or xylene, or utilizing a chemical dehydrating system with acetic anhydride and pyridine, the goal is to efficiently eliminate the water molecules generated during ring closure. This step is critical for achieving the high thermal stability required for aerospace applications, as any residual water or unreacted acid groups can act as weak points in the polymer network. The patent's emphasis on solvent recovery and recycling further underscores the mechanistic efficiency, ensuring that the process remains economically viable and environmentally sustainable.
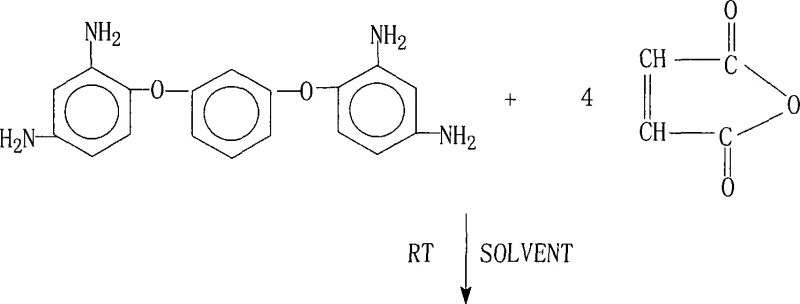
How to Synthesize 1,3-Bis(2,4-Bismaleimidophenoxy)benzene Efficiently
To achieve optimal results in the production of this high-value intermediate, strict adherence to the patented protocol is essential for maintaining batch-to-batch consistency. The process requires precise molar ratios, specifically maintaining a slight excess of maleic anhydride to drive the initial amidation to completion without leaving unreacted amine groups. Operators must ensure that the solvent system is anhydrous and that the temperature is strictly controlled during the addition of reagents to prevent localized overheating. For the cyclization phase, the choice between thermal and chemical dehydration should be based on the available infrastructure, with careful monitoring of reaction time to ensure full conversion. Detailed standardized synthesis steps are provided below to guide technical teams in implementing this robust methodology.
- React 1,3-bis(2,4-diaminophenoxy)benzene with maleic anhydride in a polar aprotic solvent at room temperature to form the tetra-amic acid intermediate.
- Perform dehydration cyclization using either azeotropic distillation with aromatic solvents or chemical dehydration with acetic anhydride and a tertiary amine catalyst.
- Isolate the final product through cooling, filtration, washing with appropriate solvents, and drying under vacuum to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers compelling benefits that directly address the pain points of modern chemical procurement and supply chain management. The elimination of high-pressure equipment requirements drastically reduces capital expenditure for new production lines, allowing for faster deployment of manufacturing capacity. Additionally, the ability to recover and recycle organic solvents such as DMAc and NMP creates a closed-loop system that minimizes raw material consumption and waste disposal costs. This efficiency translates into substantial cost savings over the lifecycle of the product, making it a financially sound choice for long-term sourcing strategies. The simplicity of the operation also reduces the reliance on highly specialized labor, further enhancing the economic viability of the process.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by removing the need for expensive high-pressure reactors and corrosive-resistant linings, which are typically major cost drivers in imide synthesis. The ambient pressure operation allows for the use of standard glass-lined or stainless steel reactors, lowering the initial investment threshold for production facilities. Furthermore, the high yield reported in the patent examples indicates minimal raw material wastage, ensuring that the cost per kilogram of the final product remains competitive. By integrating solvent recovery systems, manufacturers can further reduce variable costs associated with purchasing fresh solvents for every batch.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 1,3-bis(2,4-diaminophenoxy)benzene and maleic anhydride, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. This abundance of feedstock ensures that production schedules can be maintained even during periods of market volatility or logistical disruptions. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in utility supplies, such as steam pressure or cooling water temperature, guaranteeing consistent output. This reliability is crucial for downstream customers in the aerospace and electronics sectors who depend on uninterrupted material flows.
- Scalability and Environmental Compliance: The design of this synthesis inherently supports scalability, as the reaction kinetics and heat transfer characteristics remain favorable when moving from pilot to full-scale production. The absence of corrosive byproducts simplifies waste treatment protocols, making it easier for facilities to comply with increasingly stringent environmental regulations. Solvent recycling not only lowers costs but also significantly reduces the facility's carbon footprint by minimizing volatile organic compound emissions. This alignment with green chemistry principles enhances the brand value of the manufacturer and meets the sustainability criteria often required by multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetramaleimide compound. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply chains. The responses cover aspects of purity, scalability, and process safety to provide a comprehensive overview.
Q: What are the key advantages of this synthesis method over traditional imidization routes?
A: This method operates under atmospheric pressure without requiring high-pressure equipment, utilizes readily available raw materials, and allows for solvent recovery, significantly reducing environmental impact and operational costs compared to harsher traditional methods.
Q: How does the process ensure high purity for electronic grade applications?
A: The process employs specific polar aprotic solvents like NMP or DMAc which ensure complete dissolution and homogeneous reaction, followed by rigorous washing and recrystallization steps that effectively remove unreacted anhydride and oligomeric impurities.
Q: Is this synthesis route scalable for industrial production of polyimide precursors?
A: Yes, the reaction conditions are mild and the equipment requirements are standard, allowing for easy scale-up from laboratory to multi-ton production without the need for specialized high-pressure reactors or complex safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(2,4-Bismaleimidophenoxy)benzene Supplier
As the demand for high-performance polyimide materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 1,3-bis(2,4-bismaleimidophenoxy)benzene meets the exacting standards required for aerospace and electronic applications. We are committed to delivering quality that empowers your innovation.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your material costs and performance metrics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can become a cornerstone of your supply chain strategy.
