Scalable Manufacturing of High-Purity Tetramaleimide Precursors for Advanced Composites
The landscape of high-performance polymer manufacturing is constantly evolving, driven by the demand for materials that can withstand extreme thermal and mechanical stress in aerospace and microelectronics. Patent CN101250152A introduces a robust and highly efficient preparation method for 1,3-bis(2,4-bismaleimidophenoxy)benzene, a critical tetramaleimide compound serving as a foundational building block for next-generation thermosetting polyimides. This specific molecular architecture, characterized by its four reactive maleimide double bonds and stable ether linkages, offers exceptional potential for creating advanced composite matrices and high-temperature epoxy modifiers. The disclosed technology represents a significant leap forward in process engineering, moving away from complex, multi-stage syntheses towards a streamlined, two-step protocol that maximizes yield while minimizing operational complexity. For R&D directors and procurement strategists alike, understanding the nuances of this synthesis is key to securing a reliable supply chain for high-value electronic and structural materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multifunctional maleimide monomers has been plagued by significant technical hurdles that hindered their widespread commercial adoption. Traditional routes often relied on harsh reaction conditions, requiring high-pressure equipment and aggressive reagents that introduced safety risks and increased capital expenditure for reactor infrastructure. Furthermore, conventional methods frequently suffered from poor control over the cyclization step, leading to incomplete imidization and the formation of stubborn amic acid impurities that were difficult to separate. These impurities not only compromised the thermal stability of the final polyimide resin but also affected the curing kinetics, resulting in inconsistent batch-to-batch performance. The reliance on stoichiometric amounts of expensive dehydrating agents without effective recovery systems further inflated production costs, making these high-performance monomers economically unviable for all but the most niche applications.
The Novel Approach
The methodology presented in CN101250152A fundamentally restructures the synthesis pathway to address these legacy inefficiencies through a mild, atmospheric pressure process. By separating the reaction into a distinct room-temperature amidation phase followed by a controlled cyclodehydration step, the process ensures complete conversion of the starting diamine before the critical ring-closing event occurs. This novel approach eliminates the need for specialized high-pressure vessels, allowing the reaction to proceed in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. The flexibility to choose between azeotropic dehydration using common solvents like toluene or chemical dehydration using acetic anhydride provides manufacturers with strategic options to optimize for either energy efficiency or reaction speed. This adaptability is crucial for scaling production to meet the growing demands of the aerospace and printed circuit board industries without compromising on product quality.
Mechanistic Insights into Stepwise Amidation and Cyclodehydration
The core of this synthetic strategy lies in the precise kinetic control of the nucleophilic attack between the primary amine groups of 1,3-bis(2,4-diaminophenoxy)benzene and the electrophilic carbonyl carbons of maleic anhydride. In the initial stage, conducted at room temperature in polar aprotic solvents such as N,N-dimethylacetamide (DMAc) or N-methyl-2-pyrrolidone (NMP), the reaction proceeds rapidly to form the tetra-amic acid intermediate. Maintaining the temperature at ambient levels during this exothermic addition is critical; it prevents the premature thermal cyclization that could lead to cross-linking or gelation within the reactor. The use of a slight molar excess of maleic anhydride (ratio 1.0:4.0-4.2) drives the equilibrium towards the complete formation of the amic acid, ensuring that no free amine groups remain to act as defects in the final polymer network. This meticulous control over the first step lays the groundwork for a high-purity final product by establishing a clean intermediate profile before the more rigorous dehydration conditions are applied.
Following the formation of the amic acid, the mechanism shifts to the dehydration cyclization, which can be achieved via two distinct pathways depending on the desired operational parameters. In the azeotropic method, the addition of a water-immiscible solvent like toluene or xylene allows for the continuous removal of the water byproduct through reflux, driving the equilibrium towards the imide formation according to Le Chatelier's principle. Alternatively, the chemical dehydration pathway employs acetic anhydride coupled with a tertiary amine catalyst, such as triethylamine or pyridine, to activate the carboxylic acid groups for intramolecular nucleophilic attack. This chemical route operates effectively at moderate temperatures between 50°C and 100°C, significantly lower than traditional thermal imidization temperatures, thereby preserving the integrity of the unsaturated maleimide double bonds. Both mechanisms result in the evolution of four molecules of water per molecule of product, transforming the flexible amic acid chains into rigid, thermally stable five-membered imide rings.
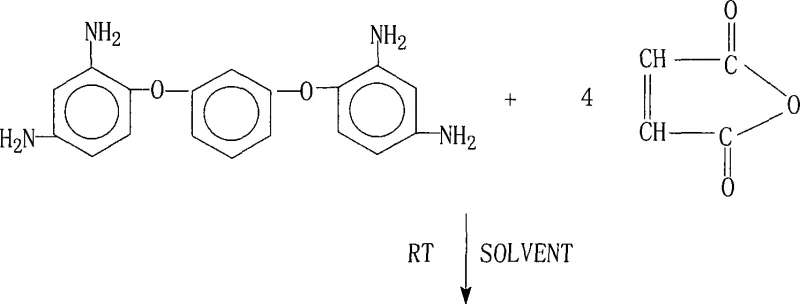
How to Synthesize 1,3-Bis(2,4-Bismaleimidophenoxy)benzene Efficiently
The practical execution of this synthesis requires careful attention to solvent selection and isolation techniques to maximize recovery and purity. The process begins with the dissolution of the diamine in a high-boiling polar solvent, followed by the controlled addition of maleic anhydride to manage the exotherm. Once the intermediate solution is confirmed, the choice of cyclization method dictates the subsequent workup. For the azeotropic route, the reaction mixture is heated to reflux until water separation ceases, whereas the chemical route requires precise dosing of the anhydride and catalyst mixture. Following the reaction, the product is isolated via cooling and filtration, with specific washing protocols—such as rinsing with cold DMAc or soaking in acetone—employed to remove residual solvents and unreacted starting materials.
- React 1,3-bis(2,4-diaminophenoxy)benzene with maleic anhydride in a polar aprotic solvent at room temperature to form the tetra-amic acid intermediate.
- Perform cyclodehydration using either azeotropic distillation with toluene/xylene or chemical dehydration with acetic anhydride and a tertiary amine catalyst.
- Isolate the final product through cooling, filtration, washing with cold solvent or acetone, and vacuum drying to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the process outlined in this patent offers substantial benefits that directly impact the bottom line and supply chain resilience for manufacturers of advanced materials. The ability to conduct the entire synthesis under atmospheric pressure removes the need for costly high-pressure autoclaves, significantly lowering the barrier to entry for production and reducing capital depreciation costs. Moreover, the explicit emphasis on solvent recovery and recycling addresses one of the largest variable costs in fine chemical manufacturing. By reclaiming solvents like NMP and toluene for reuse in subsequent batches, the process drastically reduces raw material consumption and waste disposal fees, contributing to a more sustainable and cost-effective operation. This efficiency is particularly valuable when scaling up to metric ton quantities, where even marginal savings in solvent usage translate into significant financial advantages.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements and the implementation of solvent recycling loops create a leaner manufacturing model. By avoiding the need for specialized containment systems and reducing the volume of fresh solvent required per kilogram of product, the overall cost of goods sold is significantly optimized. Additionally, the use of readily available commodity chemicals like maleic anhydride and acetic anhydride ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often associated with exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by minimizing the risk of batch failures due to equipment limitations or reagent scarcity. The flexibility to switch between azeotropic and chemical dehydration methods allows production planners to adapt to fluctuations in utility costs or solvent availability without halting operations. Furthermore, the high yields reported in the patent examples indicate a consistent and reliable output, ensuring that downstream customers in the aerospace and electronics sectors receive their critical materials on schedule without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as filtration, distillation, and drying that are easily replicated in large-scale facilities. The absence of corrosive byproducts and the ability to recover organic solvents align with increasingly stringent environmental regulations, reducing the regulatory burden on the manufacturer. This compliance not only mitigates the risk of fines but also enhances the brand reputation of the supplier as a responsible partner in the global supply chain for high-performance polymers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetramaleimide precursor. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is essential for technical teams evaluating the integration of this material into their existing formulation workflows.
Q: What are the advantages of the two-step cyclization method described in CN101250152A?
A: The patent outlines two distinct pathways for the critical imidization step: azeotropic dehydration and chemical dehydration. This flexibility allows manufacturers to choose based on equipment availability and energy costs. The azeotropic method utilizes common solvents like toluene for water removal, while the chemical method uses acetic anhydride and tertiary amines at moderate temperatures (50°C-100°C), avoiding the need for extreme thermal stress that could degrade sensitive functional groups.
Q: How does this process ensure high purity for electronic grade applications?
A: High purity is achieved through a controlled room temperature amidation phase that prevents premature polymerization or side reactions. Furthermore, the isolation protocol involves specific washing steps—such as rinsing with cold N,N-dimethylacetamide or soaking in acetone—which effectively remove unreacted maleic anhydride and oligomeric impurities, ensuring the final tetramaleimide meets stringent specifications for FPC adhesives and microelectronic insulation.
Q: Is the solvent system compatible with large-scale industrial recovery?
A: Yes, the process is explicitly designed for industrial scalability with a focus on sustainability. The patent highlights the recovery and recycling of organic solvents such as N-methyl-2-pyrrolidone (NMP) and toluene after the reaction concentration step. This closed-loop solvent management not only drastically reduces raw material consumption but also minimizes environmental impact, aligning with modern green chemistry standards for bulk chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(2,4-Bismaleimidophenoxy)benzene Supplier
As the demand for high-temperature resistant materials continues to surge in sectors ranging from aviation to flexible printed circuits, securing a dependable source of high-purity monomers is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 1,3-bis(2,4-bismaleimidophenoxy)benzene meets the exacting standards required for aerospace composites and electronic insulation applications. We understand that in the world of advanced materials, consistency is not just a metric but a necessity for mission-critical performance.
We invite procurement leaders and R&D innovators to collaborate with us to optimize their material sourcing strategies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability. Let us partner with you to drive the next generation of high-performance polymer solutions.
