Scaling High-Purity 3,4,5-Trifluorophenol: A Technical Breakthrough in Fluorinated Aromatic Synthesis
Scaling High-Purity 3,4,5-Trifluorophenol: A Technical Breakthrough in Fluorinated Aromatic Synthesis
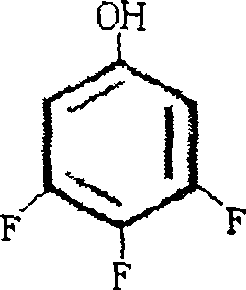
The global demand for highly fluorinated aromatic compounds has surged, driven by their critical applications in liquid crystal displays and advanced pharmaceutical intermediates. Among these, 3,4,5-trifluorophenol (CAS 99627-05-1) stands out as a pivotal building block, yet its efficient production has historically been constrained by complex synthetic routes. Patent CN100488930C introduces a transformative production technology that shifts the paradigm from cumbersome Grignard methodologies to a streamlined, copper-catalyzed amination sequence. This innovation not only addresses the purity concerns of R&D directors but also offers a robust framework for cost reduction in pharmaceutical intermediates manufacturing. By leveraging 1-halo-3,4,5-trifluorobenzene as a starting material, the process achieves yields significantly higher than previous industry standards, positioning it as a vital asset for any reliable agrochemical intermediate supplier seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4,5-trifluorophenol relied heavily on Grignard reagent chemistry, a pathway fraught with operational inefficiencies and safety hazards. The conventional route involves converting 3,4,5-trifluorobromobenzene into a Grignard reagent, followed by reaction with a borate ester and subsequent oxidation. This multi-step sequence demands strictly anhydrous conditions, necessitating expensive solvent drying protocols and specialized inert atmosphere equipment. Furthermore, the Grignard reaction is notoriously sensitive to moisture and temperature fluctuations, often leading to inconsistent batch quality and significant safety risks associated with exothermic events. From a supply chain perspective, the overall yield of this legacy method rarely exceeds 50%, resulting in excessive raw material consumption and a heavy burden on waste treatment facilities due to the large volumes of organic solvents required for workup and purification.
The Novel Approach
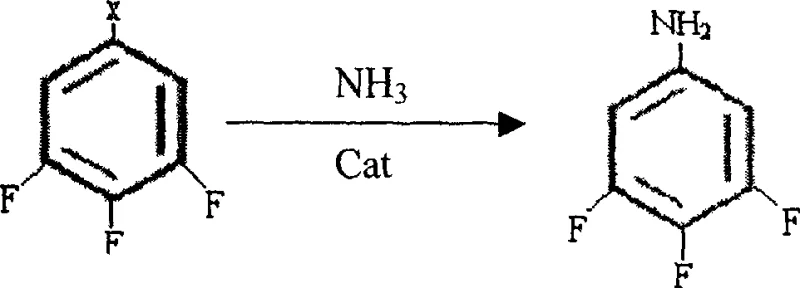
In stark contrast, the novel approach detailed in the patent utilizes a direct nucleophilic aromatic substitution strategy that dramatically simplifies the process flow. The core innovation lies in the high-pressure reaction of 1-halo-3,4,5-trifluorobenzene with aqueous ammonia, catalyzed by sub-stoichiometric amounts of copper species. This single transformation effectively installs the nitrogen functionality required for the subsequent conversion to the phenol, bypassing the need for moisture-sensitive organometallic reagents entirely. The reaction operates efficiently at temperatures between 150°C and 160°C, conditions that are readily achievable in standard industrial autoclaves without requiring exotic materials of construction. By eliminating the Grignard step, the process inherently reduces the solvent load and simplifies the post-reaction workup to a straightforward filtration and crystallization, thereby enhancing the commercial scale-up of complex fluorinated aromatics.
Mechanistic Insights into Copper-Catalyzed Amination and Diazotization
The success of this synthetic route hinges on the precise mechanistic control of the copper-catalyzed amination step. In this transformation, the copper catalyst, whether introduced as cuprous oxide, cuprous sulfate, or other copper salts, facilitates the displacement of the halogen atom on the electron-deficient aromatic ring by the ammonia nucleophile. The presence of three fluorine atoms activates the ring towards nucleophilic attack, but the copper catalyst is essential for lowering the activation energy and ensuring high conversion rates within a reasonable timeframe. Optimization data indicates that maintaining the ammonia concentration above 20%, preferably at 28%, drives the equilibrium forward, while a molar ratio of ammonia to substrate between 5:1 and 15:1 ensures complete consumption of the starting halide. This careful balance prevents the formation of poly-aminated byproducts and ensures that the residual halide content drops below 1%, a critical purity metric for downstream processing.
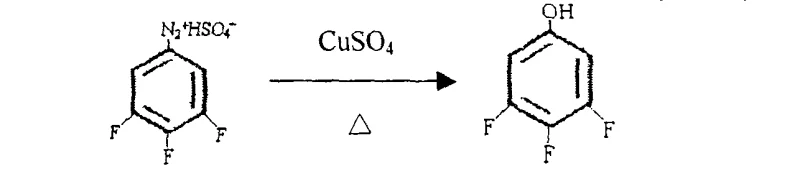
Following the amination, the conversion of 3,4,5-trifluoroaniline to the target phenol proceeds via a classic diazotization-hydrolysis sequence, yet with specific optimizations for this fluorinated substrate. The diazotization is conducted in concentrated sulfuric acid at low temperatures, typically between -5°C and 10°C, to stabilize the reactive diazonium intermediate. Immediate hydrolysis of this intermediate is then effected by heating in the presence of copper sulfate and additional sulfuric acid. The copper ions in the hydrolysis step act as a catalyst to facilitate the replacement of the diazo group with a hydroxyl group. Crucially, the reaction temperature during hydrolysis is maintained between 120°C and 130°C; exceeding this range can lead to rapid decomposition and tar formation, while lower temperatures result in incomplete conversion. This controlled thermal profile allows for the continuous distillation of the product, shifting the equilibrium and minimizing side reactions.
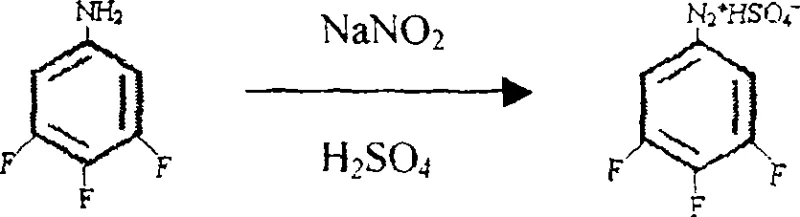
How to Synthesize 3,4,5-Trifluorophenol Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure reproducibility and safety. The process begins with the charging of the copper catalyst and the halogenated starting material into a pressure vessel, followed by the controlled addition of concentrated ammonium hydroxide. Once the amination is complete and the aniline intermediate is isolated, it is dissolved in sulfuric acid for the diazotization phase, where temperature control is paramount to prevent runaway reactions. Finally, the hydrolysis step integrates the addition of copper sulfate and careful thermal ramping to distill the product directly from the reaction mixture. For laboratory or pilot plant teams looking to replicate these results, the detailed standardized synthesis steps are provided in the guide below to ensure operational consistency.
- React 1-halo-3,4,5-trifluorobenzene with aqueous ammonia at 150-160°C under high pressure using a copper catalyst to form 3,4,5-trifluoroaniline.
- Perform diazotization of the resulting aniline using sodium nitrite and sulfuric acid at low temperatures (-5 to 10°C).
- Hydrolyze the diazonium salt intermediate using copper sulfate and sulfuric acid at 120-130°C to yield the final phenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed route offers compelling economic and logistical advantages that extend beyond simple yield metrics. The elimination of the Grignard reagent preparation removes the need for expensive, high-purity anhydrous solvents and the associated infrastructure for moisture control, leading to significant capital expenditure savings. Furthermore, the simplified workup procedure, which relies on filtration and distillation rather than complex chromatographic separations, drastically reduces the consumption of silica gel and elution solvents. This streamlining of the manufacturing process translates directly into a more competitive cost structure, allowing for better margin protection in volatile markets. Additionally, the use of aqueous ammonia and common copper salts as reagents ensures a stable and readily available supply chain, mitigating the risk of raw material shortages that often plague specialty organometallic reagents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substantial increase in overall yield, which climbs from a historical baseline of roughly 50% to over 74%. This efficiency gain means that for every ton of final product produced, nearly half the amount of starting raw material is required compared to legacy methods. Moreover, the reduction in solvent usage and the avoidance of cryogenic conditions for Grignard formation lower the utility costs associated with cooling and solvent recovery. The qualitative impact is a leaner manufacturing footprint with reduced waste disposal fees, as the process generates fewer hazardous byproducts and spent solvent streams that require specialized treatment.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as sulfuric acid, sodium nitrite, and copper sulfate, the process decouples production from the volatile supply chains of specialized organometallic reagents. The starting material, 1-halo-3,4,5-trifluorobenzene, is a stable solid or liquid that can be stored and transported without the stringent safety protocols required for pyrophoric Grignard reagents. This stability enhances the resilience of the supply chain, allowing for larger batch sizes and longer campaign runs without the risk of reagent degradation. Consequently, manufacturers can offer more reliable lead times and maintain consistent inventory levels, which is critical for downstream customers in the pharmaceutical and electronics sectors who operate on just-in-time delivery schedules.
- Scalability and Environmental Compliance: The process is inherently scalable because it utilizes unit operations—high-pressure amination, acid-base extraction, and distillation—that are standard in fine chemical plants. There is no need for specialized reactors capable of handling extreme cryogenic temperatures or highly reactive organometallics, which simplifies the technology transfer from pilot to commercial scale. From an environmental standpoint, the reduction in organic solvent volume and the absence of magnesium waste streams simplify effluent treatment. The process aligns well with green chemistry principles by improving atom economy and reducing the E-factor (mass of waste per mass of product), making it easier for facilities to meet increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this production technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on yield expectations, catalyst selection, and process safety. Understanding these nuances is essential for engineering teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does the yield of this new process compare to traditional Grignard methods?
A: Traditional Grignard-based routes typically suffer from yields of 50% or lower due to harsh anhydrous requirements and side reactions. The patented copper-catalyzed amination route achieves yields between 74% and 77%, representing a substantial improvement in material efficiency.
Q: What catalysts are preferred for the initial amination step?
A: The process utilizes cuprous salts, copper salts, or copper oxides. Specifically, cuprous oxide and cuprous sulfate have demonstrated optimal performance, facilitating the nucleophilic substitution of the halogen atom by the amino group under high-pressure conditions.
Q: Why is the hydrolysis temperature critical in the final step?
A: Maintaining the hydrolysis temperature between 120°C and 130°C is crucial. Temperatures exceeding 170°C in earlier steps or improper control during hydrolysis can accelerate side reactions and tar formation, whereas the optimized range ensures steady distillate collection and maximizes product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trifluorophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced intermediates like 3,4,5-trifluorophenol requires more than just a patent; it demands deep process engineering expertise and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this copper-catalyzed route are fully realized in a GMP-compliant environment. Our facilities are equipped with high-pressure reactors and advanced distillation columns capable of handling the specific thermal and pressure profiles required for this synthesis, guaranteeing stringent purity specifications that meet the exacting standards of the global pharmaceutical and liquid crystal industries.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this high-yield process. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your manufacturing operations.
