Advanced Solid Superacid Catalysis for the Commercial Production of 4,4'-Dihydroxybenzophenone
Introduction to Patented Synthetic Innovation
The global demand for high-performance organic intermediates, particularly in the pharmaceutical and photosensitive material sectors, necessitates manufacturing processes that balance efficiency with environmental stewardship. Patent CN101270038B introduces a groundbreaking methodology for the synthesis of 4,4'-dihydroxybenzophenone, a critical building block for UV absorbers, polymer stabilizers, and active pharmaceutical ingredients. This technology fundamentally shifts the paradigm by utilizing carbon tetrachloride—a substance historically classified as an Ozone Depleting Substance (ODS)—as a primary carbon source, thereby transforming a regulated environmental liability into a valuable chemical feedstock. By employing robust solid superacid catalysts such as sulfated metal oxides (e.g., SO42-/Fe2O3), the process achieves exceptional selectivity and yield while operating under relatively mild thermal conditions ranging from 80°C to 200°C. This innovation not only addresses the urgent industrial need to phase out ODS emissions in compliance with international treaties but also offers a streamlined, one-pot synthetic route that significantly reduces downstream processing complexity compared to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 4,4'-dihydroxybenzophenone has relied heavily on the condensation of p-hydroxybenzoic acid with phenol, typically catalyzed by polyphosphoric acid (PPA). While this route is chemically feasible, it suffers from severe operational and environmental drawbacks that hinder modern large-scale manufacturing. The use of PPA creates highly corrosive reaction environments that demand expensive, specialized reactor materials, leading to increased capital expenditure and maintenance costs. Furthermore, the disposal of waste phosphoric acid presents a significant environmental burden, requiring complex neutralization and treatment protocols to meet regulatory standards. Alternative multi-step syntheses involving acetylation, chlorination, and Fries rearrangement have been proposed to circumvent some issues, but these pathways introduce excessive unit operations, resulting in cumulative yield losses and inflated production costs due to the consumption of stoichiometric reagents and solvents at each stage.
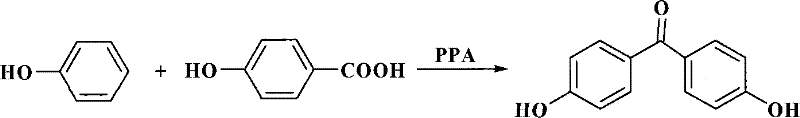
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the novel approach detailed in CN101270038B leverages a direct Friedel-Crafts type reaction between phenol and carbon tetrachloride. This strategy simplifies the molecular construction of the diaryl ketone skeleton by utilizing the tetravalent carbon of CCl4 to bridge two phenolic rings in a single reaction vessel. The introduction of solid superacid catalysts replaces the hazardous liquid acids, offering a heterogeneous catalytic system that is non-corrosive to standard stainless steel equipment. This shift allows for a dramatic reduction in reactor maintenance and extends the lifespan of plant infrastructure. Moreover, the reaction proceeds with high atom economy relative to the carbon source, and the catalyst can be mechanically separated from the reaction mixture via simple filtration, washed, calcined, and reintroduced into subsequent batches, creating a closed-loop catalytic cycle that minimizes waste generation and raw material consumption.
Mechanistic Insights into Solid Superacid-Catalyzed Diaryl Ketone Formation
The core of this technological advancement lies in the unique electronic properties of the solid superacid catalyst, specifically sulfated metal oxides like SO42-/Fe2O3 or SO42-/Al2O3. These materials possess acid strengths far exceeding those of traditional solid acids, enabling them to activate the relatively inert carbon-chlorine bonds of carbon tetrachloride at moderate temperatures. The mechanism likely involves the generation of a highly electrophilic dichlorocarbocation or a coordinated complex on the catalyst surface, which subsequently attacks the electron-rich aromatic ring of the phenol substrate. Following the initial alkylation or acylation event, a second phenolic molecule attacks the intermediate, eventually leading to the formation of a dichloromethyl-bridged species which undergoes hydrolysis during the workup phase to yield the target carbonyl functionality. This pathway avoids the formation of stable ester intermediates common in other routes, directly forging the ketone linkage with high regioselectivity for the para-position due to the steric and electronic directing effects of the hydroxyl group.
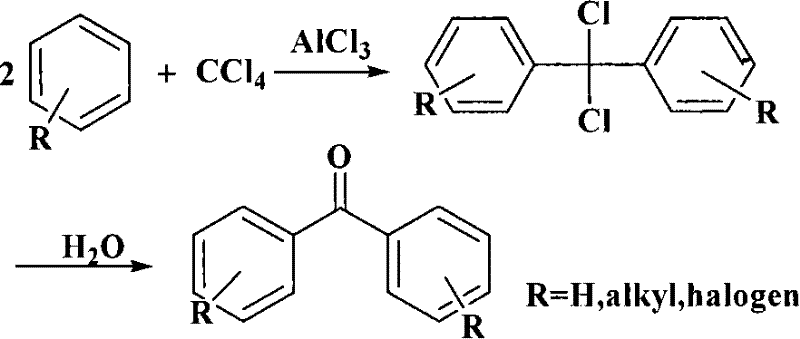
From an impurity control perspective, the heterogeneous nature of the catalyst plays a pivotal role in ensuring product quality. Unlike homogeneous catalysts that remain dissolved in the product stream and require energy-intensive distillation or extraction for removal, the solid superacid remains distinct from the liquid phase. This physical separation prevents the leaching of metal ions into the final product, a critical factor for pharmaceutical intermediates where heavy metal residues are strictly regulated. The reaction conditions, typically maintained between 100°C and 180°C for 4 to 12 hours, are optimized to maximize conversion while minimizing side reactions such as poly-alkylation or charring. The subsequent hydrolysis step with ice water not only converts the chloro-intermediates to the ketone but also quenches any residual acidity, ensuring a stable crude product ready for purification via alkaline dissolution and acid precipitation.
How to Synthesize 4,4'-Dihydroxybenzophenone Efficiently
Implementing this synthesis requires precise control over reaction parameters to replicate the high yields and purity reported in the patent embodiments. The process begins with the preparation or procurement of the specific solid superacid catalyst, followed by the careful charging of phenol and carbon tetrachloride into the reactor under an inert atmosphere to prevent oxidation. The reaction mixture is then heated to the optimal temperature range, typically around 160°C, and stirred vigorously to ensure efficient mass transfer between the liquid reactants and the solid catalyst surface. Upon completion, the protocol dictates a controlled cooling and hydrolysis sequence to safely decompose reactive intermediates before proceeding to the isolation of the crude solid. For a comprehensive understanding of the exact operational parameters, stoichiometry, and purification techniques required for GMP-compliant manufacturing, please refer to the standardized synthesis guide below.
- Prepare the solid superacid catalyst (e.g., SO4^2-/Fe2O3) by precipitating metal hydroxides, impregnating with sulfuric acid, and calcining at 500-550°C.
- React carbon tetrachloride and phenol (molar ratio 1: 5 to 1:10) with 5-15% catalyst loading at 160°C for 10 hours under stirring.
- Cool the mixture, hydrolyze with ice water, filter to recover the catalyst, extract the organic layer, and recrystallize the crude product from ethanol to achieve >99.7% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid superacid-catalyzed route offers transformative economic and logistical benefits that extend far beyond simple yield improvements. The primary value driver is the utilization of carbon tetrachloride, a commodity chemical that is often available at depressed prices due to its restricted status as an ODS, thereby drastically lowering the raw material cost basis compared to purchasing specialized carboxylic acid precursors. Furthermore, the elimination of corrosive liquid acids removes the need for expensive glass-lined reactors or Hastelloy equipment, allowing production to be scaled in standard stainless steel vessels which are more readily available and cheaper to maintain. The ability to recycle the catalyst multiple times without regeneration losses translates into a substantial reduction in consumable costs per kilogram of finished product, enhancing overall margin potential.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the convergence of low-cost feedstocks and reduced operational overhead. By substituting expensive p-hydroxybenzoic acid with inexpensive phenol and carbon tetrachloride, the direct material cost is significantly compressed. Additionally, the simplified workup procedure, which relies on filtration and crystallization rather than complex chromatographic separations or multi-stage distillations, reduces energy consumption and solvent usage. The absence of heavy metal catalysts also negates the need for costly scavenging resins or activated carbon treatments to meet strict residual metal specifications, further streamlining the cost structure.
- Enhanced Supply Chain Reliability: Supply continuity is strengthened by the reliance on bulk commodity chemicals that are produced globally in massive volumes, reducing the risk of shortages associated with niche fine chemical intermediates. The robustness of the solid catalyst system ensures consistent batch-to-batch performance, minimizing the variability that often leads to production delays or off-spec material. Moreover, the simplified process flow shortens the overall cycle time from raw material intake to finished goods, allowing for more agile response to market demand fluctuations and reducing the working capital tied up in work-in-progress inventory.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is inherently safer and more straightforward due to the absence of exothermic risks associated with liquid acid handling and the ease of solid-liquid separation. From a regulatory standpoint, converting a banned ODS into a useful product aligns perfectly with global sustainability goals and environmental regulations, potentially qualifying the manufacturing site for green chemistry incentives. The reduction in acidic wastewater and the elimination of spent acid disposal issues simplify environmental permitting and lower the long-term liability associated with waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning process validation and for procurement teams negotiating supply agreements based on this specific technological platform.
Q: What are the primary advantages of using solid superacid catalysts over traditional Lewis acids?
A: Solid superacid catalysts eliminate equipment corrosion, allow for easy separation via filtration, and can be regenerated and reused multiple times without significant loss of activity, unlike traditional liquid acids which generate hazardous waste.
Q: How does this process address environmental concerns regarding carbon tetrachloride?
A: This method utilizes carbon tetrachloride, an Ozone Depleting Substance (ODS) restricted by the Montreal Protocol, as a valuable raw material rather than a waste product, effectively recycling it into high-value fine chemicals.
Q: What purity levels can be achieved with this synthetic route?
A: Through optimized reaction conditions and a rigorous purification process involving alkaline dissolution and acid precipitation followed by recrystallization, the process consistently yields 4,4'-dihydroxybenzophenone with purity exceeding 99.7%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dihydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, eco-friendly synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the solid superacid method are fully realized in a GMP-compliant environment. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 4,4'-dihydroxybenzophenone meets the exacting standards required for pharmaceutical and high-performance polymer applications.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our mastery of this patented process can drive value and reliability for your organization.
