Advanced Solid Superacid Catalysis for Commercial Scale-up of 4,4'-Dihydroxybenzophenone Manufacturing
Introduction to Green Synthesis of 4,4'-Dihydroxybenzophenone
The global demand for high-performance organic intermediates, particularly in the photosensitive and pharmaceutical sectors, has necessitated a paradigm shift towards greener, more efficient synthetic methodologies. Patent CN101270038B introduces a groundbreaking approach for the production of 4,4'-dihydroxybenzophenone, a critical building block for UV absorbers and polymer stabilizers, by leveraging solid superacid catalysis. This technology fundamentally reimagines the utilization of carbon tetrachloride, transforming a restricted ozone-depleting substance into a valuable resource through a robust Friedel-Crafts acylation mechanism. By replacing traditional corrosive liquid acids with heterogeneous solid superacids such as SO4/Fe2O3 or SO4/Al2O3, the process achieves exceptional selectivity and catalyst recyclability while operating within a temperature range of 80 to 200°C. For R&D directors and supply chain leaders, this innovation represents a significant opportunity to enhance process safety, reduce hazardous waste generation, and secure a sustainable supply chain for complex aromatic ketones essential in modern material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4,4'-dihydroxybenzophenone has been plagued by significant technical and environmental hurdles that compromise both economic efficiency and operational safety. The traditional route, often depicted in early literature, relies heavily on p-hydroxybenzoic acid and phenol condensed in the presence of polyphosphoric acid (PPA), a method that generates vast quantities of acidic wastewater that is notoriously difficult to treat and recycle. As illustrated in the reaction scheme below, this legacy process not only suffers from low atom economy but also imposes severe corrosion risks on reactor vessels, leading to frequent maintenance downtime and increased capital expenditure for specialized alloy equipment. Furthermore, alternative multi-step pathways involving acetyl protection groups require tedious deprotection steps using ammoniacal solutions, resulting in overall yields that rarely exceed 68% and generating substantial solvent waste. The reliance on stoichiometric amounts of traditional Lewis acids like aluminum chloride further exacerbates the "three wastes" problem, creating a sludge byproduct that requires costly disposal protocols and complicates the isolation of the target molecule.
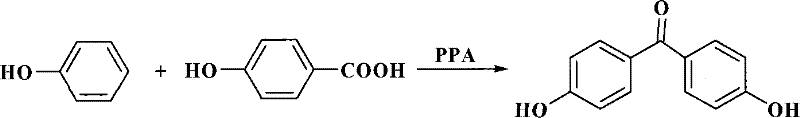
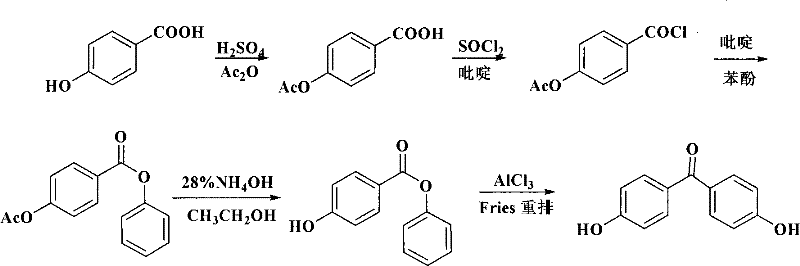
The Novel Approach
In stark contrast to these inefficient legacy methods, the novel methodology described in CN101270038B utilizes a direct acylation strategy between phenol and carbon tetrachloride mediated by a reusable solid superacid catalyst. This approach elegantly bypasses the need for protecting groups and eliminates the generation of spent liquid acid waste, as the heterogeneous catalyst can be simply filtered off and regenerated through calcination. The general mechanism, similar to the classic Friedel-Crafts reaction shown below but optimized with solid acids, allows for precise control over the reaction kinetics, minimizing the formation of ortho-isomers and poly-acylated byproducts. By operating at elevated temperatures between 100 and 180°C, the system ensures complete conversion of the limiting reagent while maintaining the structural integrity of the catalyst over multiple cycles. This streamlined single-pot process significantly reduces the number of unit operations required, thereby lowering energy consumption and simplifying the downstream purification workflow to a series of extractions and recrystallizations that yield a product with purity greater than 99.7%.
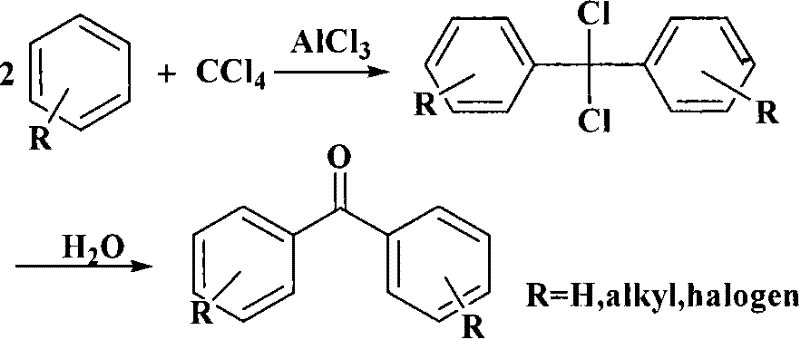
Mechanistic Insights into Solid Superacid-Catalyzed Acylation
The core innovation of this technology lies in the unique physicochemical properties of the solid superacid catalysts, specifically sulfated metal oxides like SO4/Fe2O3, which exhibit both strong Brønsted and Lewis acidity on their surfaces. Unlike homogeneous catalysts that dissolve into the reaction medium, these solid acids provide active sites that facilitate the generation of the acylium ion intermediate from carbon tetrachloride and phenol without leaching into the product stream. The high surface area and thermal stability of the catalyst allow it to withstand the rigorous reaction conditions of 160°C for extended periods, typically 6 to 10 hours, without significant deactivation. This heterogeneity is crucial for impurity control, as it prevents the catalyst from participating in side reactions that often lead to tar formation or resinification, common issues when using liquid sulfuric acid or PPA. Furthermore, the specific pore structure of the calcined metal oxide can exert a shape-selective effect, favoring the para-substitution required for 4,4'-dihydroxybenzophenone over the ortho-isomer, thus simplifying the purification burden on the manufacturing team.
From a quality control perspective, the mechanism ensures a clean impurity profile by avoiding the introduction of metal ions that are difficult to remove from the final API intermediate. The hydrolysis step, conducted by adding ice water post-reaction, effectively quenches any remaining reactive intermediates and facilitates the phase separation of the organic product from the aqueous layer. Because the catalyst remains solid throughout this quenching process, it can be recovered via simple suction filtration, washed, and returned to the reactor after a high-temperature roasting treatment to restore its acidic sites. This closed-loop catalytic cycle not only enhances the sustainability metrics of the process but also guarantees batch-to-batch consistency, a critical parameter for pharmaceutical customers who require strict adherence to impurity specifications. The ability to tune the catalyst preparation, such as adjusting the pH during the precipitation of the metal hydroxide precursor, allows manufacturers to optimize the acid strength for specific substrate reactivities, ensuring robust performance across different scales of production.
How to Synthesize 4,4'-Dihydroxybenzophenone Efficiently
The practical implementation of this synthesis involves a carefully controlled sequence of mixing, heating, and purification steps designed to maximize yield while ensuring operator safety. The process begins with the charging of phenol and carbon tetrachloride into a reactor equipped with efficient stirring and temperature control, followed by the addition of the pre-calcined solid superacid catalyst at a loading of 5 to 15 weight percent. Detailed standard operating procedures for the reaction conditions, workup, and purification are outlined in the structured guide below, which reflects the optimal parameters identified in the patent embodiments to achieve yields exceeding 60%.
- Mix carbon tetrachloride and phenol in a molar ratio of 1: 5 to 1:10 with 5-15% solid superacid catalyst (e.g., SO4/Fe2O3).
- Heat the reaction mixture to 100-180°C and stir for 4-12 hours to complete the acylation.
- Cool to room temperature, add ice water for hydrolysis, filter to recover catalyst, and purify the organic layer via extraction and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this solid superacid-catalyzed route offers transformative benefits that directly impact the bottom line and operational resilience. By shifting from a multi-step, waste-intensive process to a streamlined one-pot reaction, manufacturers can drastically simplify their production schedules and reduce the inventory of hazardous reagents required on-site. The elimination of corrosive liquid acids like polyphosphoric acid means that standard stainless steel reactors can be utilized instead of expensive glass-lined or Hastelloy vessels, leading to substantial capital cost savings and reduced maintenance overheads. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity creates a circular economy within the plant, minimizing the recurring cost of catalyst procurement and waste disposal fees associated with neutralizing spent acids. This efficiency translates into a more competitive pricing structure for the final 4,4'-dihydroxybenzophenone, allowing suppliers to offer better value to downstream customers in the UV stabilizer and pharmaceutical markets.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the utilization of carbon tetrachloride, a low-cost feedstock that is often available as a byproduct or surplus material due to regulatory restrictions on its use as a solvent. By converting this inexpensive raw material into a high-value intermediate, the process inherently lowers the raw material cost basis compared to routes starting with expensive protected benzoic acid derivatives. Additionally, the simplified workup procedure, which avoids complex distillation columns or chromatographic separations, significantly reduces utility consumption in the form of steam and electricity. The qualitative reduction in waste treatment costs is equally significant, as the absence of large volumes of acidic wastewater removes the need for extensive neutralization and effluent treatment infrastructure, further driving down the operational expenditure per kilogram of product produced.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the solid superacid catalyst, which is stable under ambient storage conditions and does not require the specialized handling or cold chain logistics associated with sensitive liquid reagents. The simplicity of the reaction setup allows for rapid scale-up from pilot plant to commercial production without the engineering bottlenecks often encountered when transferring exothermic liquid acid reactions to larger vessels. Moreover, the high selectivity of the catalyst ensures consistent product quality, reducing the risk of batch failures or off-spec material that could disrupt delivery schedules to key accounts. This reliability makes the supplier a more dependable partner for long-term contracts, as the risk of production stoppages due to equipment corrosion or waste disposal limits is effectively mitigated by the green chemistry principles embedded in the process design.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the environmental profile of a manufacturing process becomes a critical factor in vendor selection and site licensing. This technology aligns perfectly with green chemistry principles by preventing waste generation at the source rather than treating it after formation, thereby facilitating easier compliance with local environmental protection agency standards. The solid nature of the catalyst eliminates the risk of acid spills and leaks, enhancing workplace safety and reducing insurance liabilities. From a scalability perspective, the heterogeneous nature of the reaction allows for continuous flow processing possibilities in the future, offering a pathway to even higher throughput and efficiency. The ability to market the product as "sustainably manufactured" using recycled ODS materials adds a layer of brand value that resonates with environmentally conscious multinational corporations seeking to reduce their Scope 3 carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid superacid catalytic system for 4,4'-dihydroxybenzophenone production. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations for potential adopters.
Q: What are the advantages of using solid superacid catalysts over traditional Lewis acids like AlCl3?
A: Solid superacid catalysts (e.g., SO4/Fe2O3) offer superior stability, ease of separation via filtration, and recyclability without calcination loss, eliminating the corrosive waste streams associated with traditional liquid acids like polyphosphoric acid or aluminum chloride.
Q: How does this process address environmental concerns regarding carbon tetrachloride?
A: This method utilizes carbon tetrachloride, an Ozone Depleting Substance (ODS) restricted by the Montreal Protocol, as a valuable raw material rather than a waste product, effectively converting a regulated pollutant into high-value pharmaceutical intermediates.
Q: What purity levels can be achieved with this synthetic route?
A: Through optimized reaction conditions and a rigorous purification process involving alkaline dissolution and acid precipitation followed by recrystallization, the final 4,4'-dihydroxybenzophenone product achieves a purity exceeding 99.7%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dihydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the solid superacid method are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace isomers and heavy metals, guaranteeing that every batch of 4,4'-dihydroxybenzophenone meets the exacting standards required for pharmaceutical and electronic grade applications. We are committed to leveraging this innovative patent technology to deliver a product that not only performs exceptionally but also aligns with our clients' sustainability goals.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized synthesis route can enhance your supply chain efficiency and product quality. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your volume requirements. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to green chemistry and operational excellence can drive value for your organization.
