Advanced Copper-Catalyzed Synthesis of Ketorolac Tromethamine Intermediates for Commercial Scale-Up
Introduction to Next-Generation Ketorolac Intermediate Manufacturing
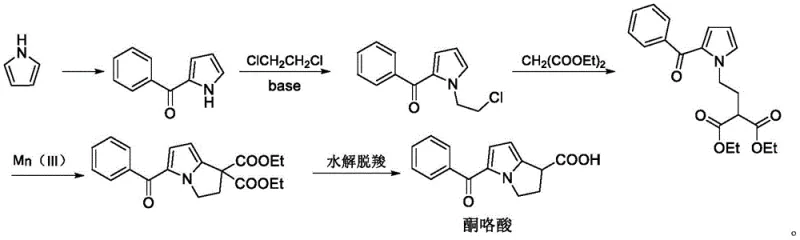
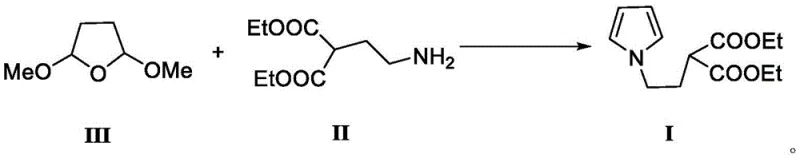
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing non-steroidal anti-inflammatory drugs (NSAIDs), particularly for high-volume analgesics like Ketorolac Tromethamine. A pivotal advancement in this domain is detailed in patent CN113185443A, which discloses a novel preparation method for a critical ketorolac intermediate, specifically 2-(2-(1H-pyrrole-1-yl)ethyl) diethyl malonate. This patent represents a significant departure from legacy synthetic routes by employing a green, copper-catalyzed condensation strategy that utilizes 2,5-dimethoxytetrahydrofuran and 2-(2-aminoethyl)diethyl malonate as key starting materials. For R&D directors and procurement strategists, this technology offers a compelling value proposition: it replaces hazardous reagents with benign copper salts, operates under mild reflux conditions, and delivers exceptional yields approaching 99%. By shifting the paradigm from harsh chemical environments to catalytic precision, this method not only enhances the safety profile of the manufacturing process but also ensures a consistent supply of high-purity intermediates essential for final API quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ketorolac and its precursors has been plagued by significant operational and environmental challenges that hinder efficient commercial scale-up. Traditional routes, such as those described in earlier literature involving pyrrole benzoylation followed by alkylation, rely heavily on phosphorus oxychloride (POCl3) as a cyclization agent. The use of POCl3 introduces severe handling risks due to its corrosive nature and the generation of acidic waste streams that require costly neutralization and disposal protocols. Furthermore, these legacy processes often necessitate the use of expensive bases like potassium carbonate and involve prolonged reaction times that bottleneck production throughput. Another alternative route utilizing strong oxidants like hydrogen peroxide poses substantial safety hazards regarding thermal runaway and explosion risks during mass production. These factors collectively inflate the cost of goods sold (COGS) and complicate supply chain reliability, making traditional methods increasingly untenable for modern, compliance-driven manufacturing facilities.
The Novel Approach
In stark contrast, the methodology outlined in CN113185443A introduces a streamlined, single-step cyclization that elegantly bypasses these historical pain points. By leveraging the reactivity of 2,5-dimethoxytetrahydrofuran as a masked 1,4-dicarbonyl equivalent, the process facilitates a smooth Paal-Knorr-type pyrrole formation with the amino-functionalized malonate. This approach eliminates the need for hazardous chlorinating agents and strong oxidants entirely. The reaction proceeds efficiently in common organic solvents such as tetrahydrofuran (THF) or acetonitrile under standard reflux conditions. The simplicity of the workup procedure—often involving mere filtration and solvent evaporation—drastically reduces processing time and solvent consumption. This technological leap transforms the synthesis from a multi-step, hazard-laden sequence into a robust, convergent process that is ideally suited for the rigorous demands of industrial pharmaceutical manufacturing.
Mechanistic Insights into Copper-Catalyzed Pyrrole Formation
The core innovation of this patent lies in the utilization of copper catalysts to drive the condensation and cyclization reactions with high selectivity and minimal byproduct formation. Mechanistically, the copper species, such as copper sulfate or copper acetate, likely acts as a Lewis acid to activate the acetal groups of the 2,5-dimethoxytetrahydrofuran, facilitating the nucleophilic attack by the primary amine of the diethyl 2-(2-aminoethyl)malonate. This activation lowers the energy barrier for the initial imine formation and subsequent dehydration steps required to close the pyrrole ring. Unlike uncatalyzed thermal methods which may require excessive temperatures leading to decomposition, the copper catalyst allows the reaction to proceed at moderate reflux temperatures (e.g., in THF), preserving the integrity of the sensitive ester functionalities on the malonate side chain. This precise control over reaction kinetics is crucial for maintaining the structural fidelity of the intermediate, ensuring that the final product is free from polymeric impurities or ring-opened byproducts that often plague uncatalyzed variants.
Furthermore, the choice of catalyst and solvent system plays a pivotal role in impurity control, a critical parameter for R&D teams focused on regulatory compliance. The patent data indicates that using copper sulfate in THF yields products with HPLC purity exceeding 99.8%, suggesting that the catalytic cycle is highly specific and does not promote side reactions such as over-alkylation or ester hydrolysis. The presence of a small amount of water in the reaction mixture, as noted in the examples, may assist in the hydrolysis of the methoxy groups without degrading the diethyl ester moieties, striking a delicate balance that maximizes conversion while minimizing degradation. This high level of chemoselectivity means that downstream purification can be simplified to basic crystallization or filtration, rather than requiring resource-intensive chromatographic separations, thereby enhancing the overall process mass intensity (PMI) of the synthesis.
How to Synthesize 2-(2-(1H-pyrrole-1-yl)ethyl) diethyl malonate Efficiently
Implementing this advanced synthesis route requires careful attention to stoichiometry and catalyst loading to replicate the high yields reported in the patent literature. The process begins with the precise charging of 2,5-dimethoxytetrahydrofuran and the amino-malonate derivative into a reactor equipped with efficient agitation and temperature control. The molar ratio is typically adjusted to favor the limiting reagent, often using a slight excess of the furan derivative to drive the equilibrium towards completion. Following the addition of the copper catalyst and the selected solvent, the mixture is heated to reflux for a defined period, typically around 10 hours, to ensure full conversion. The detailed standardized operating procedures, including specific workup techniques like solvent swapping and aqueous washing to remove copper residues, are critical for achieving the reported purity specifications. For a comprehensive step-by-step guide on executing this synthesis safely and effectively, please refer to the technical instructions below.
- Charge 2,5-dimethoxytetrahydrofuran and 2-(2-aminoethyl)diethyl malonate into a reactor with a copper catalyst (e.g., CuSO4) and THF solvent.
- Heat the mixture to reflux temperature and maintain stirring for approximately 10 hours to ensure complete cyclization.
- Cool the reaction, evaporate solvent, dissolve residue in dichloromethane, filter, wash with water, dry over sodium sulfate, and concentrate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed technology translates directly into tangible operational efficiencies and risk mitigation. The elimination of phosphorus oxychloride and hydrogen peroxide removes significant safety liabilities from the production floor, reducing insurance premiums and the need for specialized containment infrastructure. Moreover, the use of commodity-grade copper salts instead of precious metal catalysts or exotic reagents ensures that raw material costs remain stable and predictable, shielding the supply chain from volatile pricing fluctuations associated with rare earth elements. The simplified workup procedure, which avoids complex extraction sequences and extensive neutralization steps, significantly shortens the batch cycle time, allowing manufacturing facilities to increase throughput without capital expansion. This agility is vital for meeting the fluctuating demands of the global NSAID market while maintaining lean inventory levels.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with low-cost copper catalysts and the drastic simplification of the post-reaction workup. By removing the need for corrosion-resistant reactors required for POCl3 handling and eliminating the costs associated with treating acidic waste streams, the overall production cost is substantially lowered. Additionally, the high yield and purity reduce the loss of valuable starting materials and minimize the need for reprocessing off-spec batches, leading to a more efficient utilization of resources and a direct improvement in the gross margin of the intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the dependency on hard-to-source specialty chemicals. Since the key reagents like 2,5-dimethoxytetrahydrofuran and copper sulfate are widely available from multiple global suppliers, the risk of supply disruption due to vendor-specific issues is minimized. The mild reaction conditions also mean that the process can be easily transferred between different manufacturing sites or scaled up in existing multipurpose reactors without requiring significant retrofitting. This flexibility ensures that production schedules can be maintained even in the face of logistical challenges, providing a stable and reliable flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and permitting. The absence of heavy metal waste (other than trace copper which is easily removed) and halogenated byproducts simplifies wastewater treatment and reduces the facility's environmental footprint. The process is inherently safer for scale-up because it avoids exothermic hazards associated with strong oxidants, allowing for larger batch sizes with confidence. This scalability ensures that the technology can support commercial volumes ranging from pilot plant trials to multi-ton annual production runs without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the copper-catalyzed route over traditional POCl3 methods?
A: The copper-catalyzed method avoids highly toxic and corrosive phosphorus oxychloride (POCl3), eliminating the need for specialized corrosion-resistant equipment and complex neutralization waste treatment, thereby significantly lowering operational costs and safety risks.
Q: What purity levels can be achieved with this new synthesis method?
A: According to patent data, this method consistently achieves HPLC purity levels exceeding 99.6%, with optimized examples reaching up to 99.88%, which drastically reduces the burden on downstream purification processes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions, common solvents like THF, and inexpensive copper salts, avoiding dangerous oxidants like hydrogen peroxide, making it inherently safer and more robust for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketorolac Tromethamine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113185443A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are fully equipped to implement this copper-catalyzed route, leveraging our state-of-the-art facilities and rigorous QC labs to ensure that every batch of ketorolac intermediate meets stringent purity specifications. Our commitment to process excellence means we can deliver this high-value intermediate with the consistency and quality required by top-tier pharmaceutical companies worldwide.
We invite you to collaborate with us to optimize your supply chain for ketorolac production. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of scientific rigor and commercial viability.
