Advanced Synthesis of Ketorolac Tromethamine Intermediate VI for Commercial Scale Production
The pharmaceutical industry continuously seeks robust synthetic pathways for nonsteroidal anti-inflammatory drugs (NSAIDs), particularly for potent analgesics like Ketorolac Tromethamine. A pivotal advancement in this domain is documented in Chinese patent CN113087626A, which introduces a novel intermediate, Compound VI, and a streamlined preparation method that addresses longstanding manufacturing inefficiencies. This technical breakthrough offers a viable alternative to legacy processes by eliminating the reliance on heavy metal oxidants, thereby enhancing both environmental compliance and product quality. The chemical structure of the final active pharmaceutical ingredient, Ketorolac Tromethamine, underscores the complexity of the pyrrolizine ring system that must be constructed with high stereochemical and chemical fidelity. 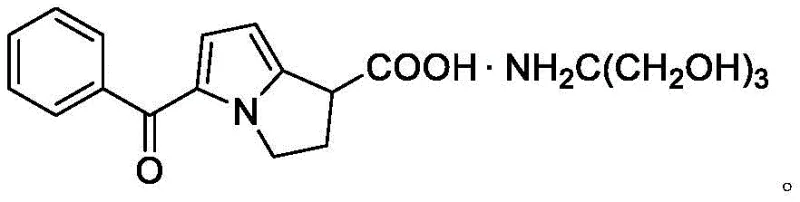 For R&D directors and procurement specialists, understanding the nuances of this new intermediate is critical for securing a reliable supply chain. The patent details a method that not only improves reaction yields but also simplifies the purification workflow, which is often a bottleneck in API manufacturing. By focusing on the synthesis of Compound VI, manufacturers can achieve a more predictable impurity profile, reducing the risk of batch failures during regulatory audits. This report analyzes the technical merits of patent CN113087626A, evaluating its potential to redefine cost structures and supply reliability for global pharmaceutical producers seeking a reliable pharmaceutical intermediates supplier.
For R&D directors and procurement specialists, understanding the nuances of this new intermediate is critical for securing a reliable supply chain. The patent details a method that not only improves reaction yields but also simplifies the purification workflow, which is often a bottleneck in API manufacturing. By focusing on the synthesis of Compound VI, manufacturers can achieve a more predictable impurity profile, reducing the risk of batch failures during regulatory audits. This report analyzes the technical merits of patent CN113087626A, evaluating its potential to redefine cost structures and supply reliability for global pharmaceutical producers seeking a reliable pharmaceutical intermediates supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ketorolac Tromethamine intermediates has relied heavily on oxidative cyclization strategies that introduce significant environmental and operational burdens. As illustrated in prior art literature, such as the work by Wanghua et al., traditional routes often employ excessive amounts of potassium permanganate and manganese acetate to construct the key diester-based intermediate. 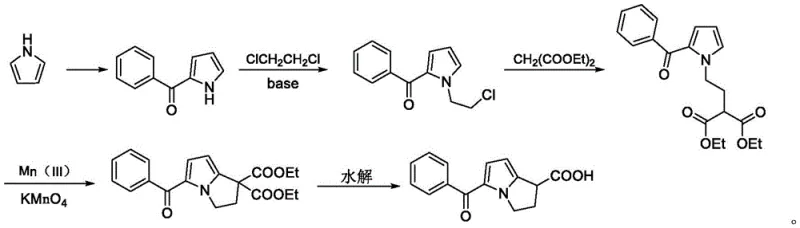 These manganese-containing reagents generate substantial quantities of hazardous process wastewater that is notoriously difficult to treat, posing severe challenges for environmental compliance officers. Furthermore, the oxidation process frequently results in products with dark coloration, necessitating multi-step decolorization procedures involving activated carbon or silica gel. This additional purification not only increases solid waste generation, such as spent carbon and silica, but also leads to significant product loss, thereby driving up the overall cost of goods sold. The complexity of removing trace metal residues to meet stringent pharmacopeial standards further complicates the manufacturing process, making these conventional methods less attractive for modern, green chemistry-focused production facilities.
These manganese-containing reagents generate substantial quantities of hazardous process wastewater that is notoriously difficult to treat, posing severe challenges for environmental compliance officers. Furthermore, the oxidation process frequently results in products with dark coloration, necessitating multi-step decolorization procedures involving activated carbon or silica gel. This additional purification not only increases solid waste generation, such as spent carbon and silica, but also leads to significant product loss, thereby driving up the overall cost of goods sold. The complexity of removing trace metal residues to meet stringent pharmacopeial standards further complicates the manufacturing process, making these conventional methods less attractive for modern, green chemistry-focused production facilities.
The Novel Approach
In stark contrast to the oxidative hurdles of the past, the novel approach detailed in patent CN113087626A utilizes a constructive acylation strategy to assemble the carbon skeleton of Compound VI without the need for harsh oxidants. This method bypasses the formation of manganese sludge entirely, replacing it with a cleaner reaction profile that relies on precise nucleophilic substitution and acylation chemistry. The process begins with readily available starting materials and proceeds through a controlled addition of reagents under mild thermal conditions, significantly reducing the energy footprint of the reaction. By avoiding the use of highly toxic and strongly corrosive substances often associated with older methodologies, this new route enhances operator safety and reduces the requirement for specialized corrosion-resistant equipment. The result is a synthesis pathway that is not only chemically elegant but also operationally simple, making it highly suitable for industrial production where consistency and safety are paramount concerns for any cost reduction in pharmaceutical intermediates manufacturing initiative.
Mechanistic Insights into the Acylation of Diethyl Malonate Derivatives
The core innovation of this patent lies in the efficient construction of the 1,3,5-tricarbonyl system found in Compound VI through a carefully orchestrated acylation reaction. The mechanism involves the generation of a nucleophilic enolate from diethyl 2-(2-chloroethyl)malonate (Compound V) using a strong non-nucleophilic base such as sodium hydride or lithium bis(trimethylsilyl)amide. 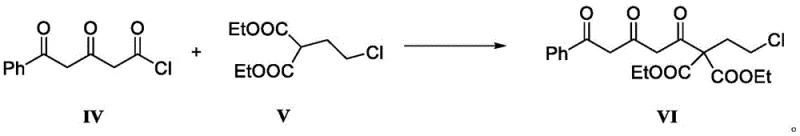 This enolate formation is critically temperature-dependent, requiring maintenance between -20°C and -5°C to prevent self-condensation or decomposition of the sensitive malonate species. Once the enolate is stabilized, the electrophilic acid chloride (Compound IV), derived from 5-phenyl-4,5-dioxopentanoic acid, is introduced dropwise at a controlled temperature of approximately -20°C. This low-temperature addition is essential to manage the exothermic nature of the acylation and to ensure that the reaction proceeds selectively at the alpha-carbon of the malonate without affecting the ester groups or the chloroethyl side chain. The precision required in this step highlights the importance of advanced process control systems in a commercial setting to maintain the narrow thermal window that guarantees high purity.
This enolate formation is critically temperature-dependent, requiring maintenance between -20°C and -5°C to prevent self-condensation or decomposition of the sensitive malonate species. Once the enolate is stabilized, the electrophilic acid chloride (Compound IV), derived from 5-phenyl-4,5-dioxopentanoic acid, is introduced dropwise at a controlled temperature of approximately -20°C. This low-temperature addition is essential to manage the exothermic nature of the acylation and to ensure that the reaction proceeds selectively at the alpha-carbon of the malonate without affecting the ester groups or the chloroethyl side chain. The precision required in this step highlights the importance of advanced process control systems in a commercial setting to maintain the narrow thermal window that guarantees high purity.
Following the acylation, the reaction mixture undergoes a quenching process where the pH is adjusted to a neutral range of 5 to 7 using dilute sulfuric acid. This step is crucial for protonating the intermediate alkoxide and decomposing any excess base, thereby stabilizing the final beta-keto ester product. The subsequent workup involves solvent evaporation under reduced pressure followed by extraction with organic solvents like dichloromethane, which effectively separates the organic product from inorganic salts. The patent data indicates that this mechanistic pathway results in exceptionally low impurity levels, as evidenced by HPLC purity readings consistently above 99.7% across multiple examples. For R&D teams, this high level of chemical fidelity means that downstream cyclization steps to form the pyrrolizine ring will proceed with fewer side reactions, ultimately leading to a higher overall yield of the final API and reducing the burden on analytical quality control laboratories.
How to Synthesize Ketorolac Tromethamine Intermediate Efficiently
The practical implementation of this synthesis requires strict adherence to the molar ratios and solvent systems optimized in the patent examples to ensure reproducibility at scale. The protocol suggests a molar ratio of Compound V to the acid chloride (Compound IV) ranging from 1:1.2 to 1:2.0, with a particular preference for a slight excess of the electrophile to drive the reaction to completion. Solvent selection is equally critical, with tetrahydrofuran (THF) identified as the particularly preferable medium due to its ability to solubilize both the enolate and the acid chloride while maintaining stability at low temperatures. Detailed standardized synthesis steps see the guide below. Adhering to these parameters allows manufacturers to replicate the high yields reported in the patent, such as the 98.6% yield achieved in Example 6, ensuring that the process is not merely theoretical but practically viable for high-purity pharmaceutical intermediates production.
- Preparation of Acid Chloride (Compound IV): React 5-phenyl-4,5-dioxopentanoic acid with thionyl chloride in THF at 20-25°C to form the reactive acyl chloride intermediate.
- Enolate Formation and Acylation: Treat diethyl 2-(2-chloroethyl)malonate (Compound V) with a strong base like NaH at -10°C, then add Compound IV at -20°C to synthesize Compound VI.
- Workup and Purification: Quench the reaction with dilute sulfuric acid to pH 6-7, extract with dichloromethane, and dry to obtain high-purity Compound VI with yields exceeding 98%.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis route offers compelling advantages that extend beyond simple chemical yield improvements. The elimination of heavy metal oxidants like potassium permanganate fundamentally alters the cost structure of the manufacturing process by removing the need for expensive wastewater treatment protocols associated with manganese removal. This reduction in environmental remediation costs translates directly into improved margin potential for the final API, allowing for more competitive pricing in the global market. Furthermore, the simplicity of the operation, which avoids multi-step decolorization and complex purification trains, reduces the consumption of auxiliary materials such as activated carbon and silica gel. These operational efficiencies contribute to substantial cost savings and a leaner manufacturing footprint, aligning perfectly with the goals of any procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for costly heavy metal removal steps and reduces the consumption of purification media, leading to a significantly lower cost of goods sold. By avoiding the use of expensive oxidants and minimizing waste generation, the overall material cost per kilogram of the intermediate is drastically optimized. Additionally, the high reaction yield minimizes raw material waste, ensuring that every gram of starting material contributes effectively to the final product output. This efficiency is critical for maintaining profitability in a competitive generic pharmaceutical market where margin pressure is constant.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable raw materials ensures that the supply chain is resilient against disruptions common with specialized or hazardous reagents. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic corrosion-resistant alloys, facilitates easier technology transfer between manufacturing sites. This flexibility allows for diversified sourcing strategies and reduces the risk of single-point failures in the production network. Consequently, supply chain heads can guarantee more consistent lead times for high-purity pharmaceutical intermediates, ensuring uninterrupted production schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, featuring simple unit operations like extraction and filtration that are easily managed in large reactors. The absence of toxic manganese waste simplifies regulatory compliance and reduces the environmental liability associated with chemical manufacturing. This green chemistry profile not only meets current environmental standards but also future-proofs the manufacturing site against increasingly stringent global regulations. The ability to scale from laboratory to commercial production without significant process re-engineering ensures a faster time-to-market for new generic formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical effects disclosed in patent CN113087626A, providing clarity for stakeholders evaluating this technology. Understanding these details is essential for making informed decisions about process adoption and supplier qualification. This transparency ensures that all technical risks are assessed accurately before committing to large-scale production campaigns.
Q: Why is the new synthesis route for Ketorolac Intermediate VI preferred over traditional manganese-based methods?
A: Traditional methods utilize excessive potassium permanganate and manganese acetate, generating difficult-to-treat wastewater and dark-colored products requiring complex decolorization. The novel route described in CN113087626A avoids heavy metal oxidants entirely, resulting in simpler waste streams and inherently higher product purity without extensive purification steps.
Q: What are the critical reaction conditions for maximizing the yield of Compound VI?
A: Precise temperature control is paramount. The base addition should occur between -20°C and -5°C, while the addition of the acid chloride (Compound IV) is optimally performed at -20°C. Using tetrahydrofuran (THF) as the solvent and maintaining a molar ratio of Compound V to base around 1:2.0 ensures minimal side reactions and yields approaching 98.6%.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is explicitly designed for industrial applicability. It utilizes commercially available raw materials, avoids highly toxic reagents, and features simple operational steps such as standard extraction and filtration. The high reaction speed and reduced impurity profile significantly lower the burden on downstream processing, facilitating robust commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketorolac Tromethamine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of implementing advanced synthetic routes like the one described in CN113087626A to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering stringent purity specifications and operate rigorous QC labs to verify that every batch of Ketorolac Tromethamine Intermediate meets the highest international standards. Our infrastructure is designed to handle complex chemistries safely, providing our partners with a secure and reliable source for their critical raw materials.
We invite you to collaborate with us to leverage this innovative technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency. Let us partner with you to bring high-quality, cost-effective pharmaceutical solutions to market faster.
