Scalable Production of Chiral Ticagrelor Intermediates Using Advanced Immobilized Enzyme Technology
Scalable Production of Chiral Ticagrelor Intermediates Using Advanced Immobilized Enzyme Technology
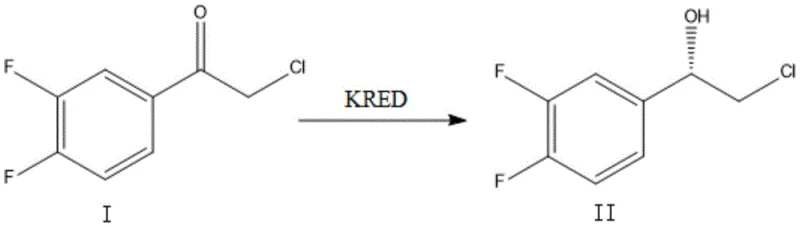
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective routes for synthesizing complex chiral intermediates, particularly for high-value cardiovascular drugs like Ticagrelor. A recent breakthrough detailed in patent CN111575334B offers a transformative solution for producing (S)-2-chloro-1-(3,4-difluorophenyl)ethanol, a critical building block in the synthesis of this potent antiplatelet agent. This patent discloses a novel biocatalytic method utilizing a specifically immobilized ketoreductase (KRED) that overcomes the historical limitations of both chemical catalysis and free-enzyme systems. By leveraging adsorption-based immobilization technology on carriers such as LX-1000ODS, the process achieves exceptional optical purity exceeding 99% ee while ensuring the enzyme can be recycled multiple times. For R&D directors and procurement specialists, this represents a pivotal shift towards greener, more economical manufacturing protocols that do not compromise on the stringent purity standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral chlorohydrins like compound II has relied heavily on chemical asymmetric reduction methods, such as the use of CBS (Corey-Bakshi-Shibata) catalysts with borane reagents. While chemically effective, these traditional routes suffer from significant drawbacks that impact both cost and environmental safety profiles. The use of boron-based reagents introduces toxicological concerns, necessitating rigorous and expensive purification steps to remove trace metal impurities to meet pharmaceutical grade specifications. Furthermore, the optical purity achieved via these chemical routes often hovers around 90-93% ee, requiring additional resolution or recrystallization steps to reach the >99% ee threshold demanded by modern drug regulators. Alternatively, early biocatalytic attempts using free enzymes faced economic hurdles; free ketoreductases are typically single-use, meaning the high cost of the biocatalyst cannot be amortized over multiple batches, rendering the process economically unviable for large-scale industrial application despite its high selectivity.
The Novel Approach
The innovative methodology presented in the patent data fundamentally alters the economic and technical landscape by employing an immobilized enzyme system. Instead of using the enzyme in a soluble, free state, the ketoreductase (specifically SEQ ID NO: 1 derived from Lactobacillus kefiri) is physically adsorbed onto a solid support matrix, specifically the macroporous adsorption resin LX-1000ODS. This immobilization strategy confers remarkable stability to the biocatalyst, allowing it to maintain high activity over extended reaction periods and multiple reuse cycles. The result is a process that combines the exquisite stereoselectivity of biocatalysis with the operational robustness of heterogeneous catalysis. By fixing the enzyme on a solid carrier, the separation of the catalyst from the reaction mixture becomes trivial, eliminating complex downstream processing and enabling a continuous or semi-continuous manufacturing workflow that drastically simplifies the overall production architecture.
Mechanistic Insights into Immobilized Ketoreductase Catalysis
The success of this process lies in the synergistic interaction between the specific protein structure of the ketoreductase and the physicochemical properties of the immobilization carrier. The enzyme, characterized by the amino acid sequence SEQ ID NO: 1, possesses an active site geometry that is highly complementary to the substrate 2-chloro-1-(3,4-difluorophenyl)ethanone. When adsorbed onto the LX-1000ODS carrier, the enzyme undergoes subtle conformational adjustments that appear to lock it into a highly active and selective state. Unlike covalent binding methods which can sometimes distort the active site and reduce activity, physical adsorption on this specific hydrophobic resin preserves the enzyme's native tertiary structure while preventing aggregation. This stabilization effect is crucial for maintaining the rigid stereochemical environment necessary to differentiate between the pro-chiral faces of the ketone substrate, thereby ensuring the exclusive formation of the (S)-enantiomer with near-perfect fidelity.
Furthermore, the reaction mechanism relies on a coupled cofactor regeneration system that is essential for industrial feasibility. The reduction of the ketone requires NADPH as a hydride donor, which would be prohibitively expensive if used stoichiometrically. The process ingeniously couples the primary reduction reaction with the oxidation of isopropanol, a cheap and abundant sacrificial donor. The immobilized ketoreductase oxidizes isopropanol to acetone, regenerating NADPH from NADP+ in situ. This creates a self-sustaining catalytic cycle where only catalytic amounts of the expensive cofactor are needed. The immobilization matrix facilitates this by keeping the enzyme and cofactor in close proximity within the reaction microenvironment, enhancing the local concentration of reactants and driving the equilibrium towards the desired chiral alcohol product with high efficiency and minimal byproduct formation.
How to Synthesize (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Efficiently
Implementing this biocatalytic route requires precise control over the immobilization parameters and reaction conditions to maximize yield and enantiomeric excess. The process begins with the preparation of the crude enzyme solution, typically obtained from the fermentation broth of recombinant E. coli expressing the target ketoreductase. This crude lysate is directly contacted with the pre-treated LX-1000ODS resin under controlled pH and temperature conditions to allow for optimal adsorption. Once the immobilized biocatalyst is prepared, it is introduced into a buffered aqueous system containing the ketone substrate and the isopropanol cosubstrate. The reaction is typically conducted at mild temperatures between 30°C and 45°C and a slightly alkaline pH of 7.5 to 8.5, conditions that are far safer and more energy-efficient than the cryogenic temperatures often required for chemical asymmetric synthesis. Detailed standardized synthetic steps follow below.
- Prepare the immobilized enzyme by adsorbing ketoreductase SEQ ID NO: 1 onto carrier LX-1000ODS in phosphate buffer.
- Mix the substrate 2-chloro-1-(3,4-difluorophenyl)ethanone with isopropanol and coenzyme NADP+ in a buffered reaction system.
- Maintain the reaction at 30-45°C and pH 7.5-8.5 until conversion is complete, then separate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from chemical catalysis or free-enzyme methods to this immobilized biocatalytic platform offers profound strategic advantages. The primary driver for cost reduction is the reusability of the biocatalyst; because the enzyme is fixed on a solid support, it can be recovered by simple filtration and reused for subsequent batches, effectively amortizing the initial enzyme production cost over a much larger volume of product. This eliminates the recurring expense of purchasing fresh enzyme for every run, leading to substantial savings in raw material costs. Additionally, the mild reaction conditions reduce energy consumption associated with heating, cooling, and pressure management, further lowering the operational expenditure (OPEX) of the manufacturing facility. The elimination of toxic boron reagents also reduces the costs associated with hazardous waste disposal and environmental compliance, streamlining the overall cost structure of the supply chain.
- Cost Reduction in Manufacturing: The immobilization technology allows the expensive ketoreductase enzyme to be recycled multiple times without significant loss of activity, effectively spreading the biocatalyst cost across numerous production batches. This contrasts sharply with free enzyme methods where the catalyst is consumed in a single use, creating a direct and significant reduction in the variable cost of goods sold (COGS) for the intermediate. Furthermore, the high conversion rates minimize substrate waste, ensuring that expensive fluorinated starting materials are efficiently converted into valuable product rather than lost to side reactions or incomplete conversion.
- Enhanced Supply Chain Reliability: By utilizing a robust immobilized enzyme system, manufacturers can achieve consistent batch-to-batch quality with minimal variation in yield or optical purity. This reliability reduces the risk of production delays caused by failed batches or the need for re-processing to meet purity specifications. The stability of the immobilized enzyme also simplifies inventory management, as the biocatalyst can be prepared in advance and stored, decoupling enzyme production from the immediate synthesis schedule and providing a buffer against supply disruptions in the upstream fermentation sector.
- Scalability and Environmental Compliance: The process operates in aqueous media at ambient pressure and moderate temperatures, removing the need for specialized high-pressure reactors or extensive solvent recovery systems required for chemical hydrogenation. This inherent safety and simplicity facilitate easier scale-up from pilot plant to commercial tonnage production. Moreover, the biocatalytic nature of the reaction generates benign byproducts like acetone and water, significantly reducing the environmental footprint and simplifying the permitting process for new manufacturing lines in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized enzyme technology for producing chiral pharmaceutical intermediates. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these factors is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into existing manufacturing workflows or for procurement teams assessing the long-term viability of suppliers utilizing this technology.
Q: Why is immobilized enzyme preferred over free enzyme for this synthesis?
A: Immobilization on carriers like LX-1000ODS significantly enhances enzyme stability and allows for repeated reuse, drastically lowering the cost per batch compared to single-use free enzymes while maintaining >99% ee value.
Q: What are the advantages over traditional chemical reduction methods?
A: Unlike CBS catalysis which uses hazardous boron reagents and yields lower optical purity (90-93%), this biocatalytic route achieves >99% ee under mild conditions without heavy metal contamination.
Q: Can this process be scaled for industrial production?
A: Yes, the patent demonstrates high stability over multiple batches with consistent yield and purity, making it highly suitable for large-scale commercial manufacturing of cardiovascular drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development and commercialization of life-saving cardiovascular medications. Our technical team has extensively analyzed emerging biocatalytic technologies, including the immobilized enzyme methods described in recent patents, to ensure our manufacturing capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require material for clinical trials or full-scale market launch, our supply remains uninterrupted. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol meets the exacting standards required for global regulatory submissions.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By leveraging our expertise in process optimization and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to our supply chain can optimize your overall project economics. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being a reliable partner in your pharmaceutical development journey.
