Advanced Biocatalytic Synthesis of Ticagrelor Key Intermediate for Commercial Scale-up
Introduction to Novel Biocatalytic Pathways for Ticagrelor Intermediates
The pharmaceutical industry continuously seeks robust and scalable methods for synthesizing chiral intermediates, particularly for blockbuster drugs like Ticagrelor. Patent CN112063531B introduces a groundbreaking approach utilizing a newly isolated strain, Geotrichum candidum ZJPH1907, for the asymmetric reduction of 2-chloro-1-(3,4-difluorophenyl)ethanone. This technology addresses critical bottlenecks in traditional manufacturing by enabling high-selectivity catalysis in a slightly acidic environment, a condition that inherently minimizes microbial contamination risks during large-scale fermentation. The strategic implementation of this whole-cell biocatalyst allows for the efficient production of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol with exceptional stereocontrol, positioning it as a superior alternative to conventional chemical synthesis routes that often rely on toxic reagents and complex purification steps.
For R&D directors and process chemists, the significance of this patent lies in its ability to streamline the supply chain for cardiovascular drug precursors. The disclosed method achieves a yield of 83.8% and an e.e. value of 97.1% at a substrate concentration of 7.6g/L within just 2 hours under optimized conditions. This performance metric is not merely a laboratory curiosity but represents a viable industrial protocol that balances reaction speed with product quality. By leveraging the intrinsic cofactor regeneration systems of the whole cell, the process eliminates the need for exogenous addition of expensive nicotinamide cofactors, thereby simplifying the reaction matrix and reducing the overall cost of goods sold (COGS) for the final active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of (S)-2-chloro-1-(3,4-difluorophenyl)ethanol typically relies on asymmetric reduction using chiral boron reagents, such as (S)-2-methyl-CBS-oxazaborolidine coupled with borane complexes. While these methods can achieve high enantiomeric excess, they suffer from significant drawbacks including the use of hazardous, moisture-sensitive reagents that require stringent anhydrous conditions and low temperatures, often around -20°C. Furthermore, the downstream processing involves complex workups to remove boron residues, which can be detrimental to catalyst life in subsequent steps and pose environmental disposal challenges. Alternative enzymatic approaches using isolated carbonyl reductases have been reported, but they frequently necessitate the continuous addition of costly cofactors like NADPH or NADH, along with auxiliary enzyme systems for regeneration, drastically inflating the operational expenditure and complicating the scale-up potential for multi-ton production campaigns.
The Novel Approach
In stark contrast, the novel approach detailed in CN112063531B utilizes a whole-cell biocatalyst that functions effectively in a biphasic or aqueous system at mild temperatures ranging from 20°C to 40°C. The use of Geotrichum candidum ZJPH1907 allows the reaction to proceed in a slightly acidic buffer, specifically optimized at pH 4.2, which creates a selective pressure against common bacterial contaminants without inhibiting the fungal biocatalyst. This innovation removes the dependency on external cofactor supplementation because the living cells possess endogenous metabolic pathways to regenerate the necessary reducing equivalents from simple co-substrates like glucose. Consequently, the process simplifies the reaction setup to a mixture of wet cells, buffer, substrate, and a carbon source, significantly reducing the number of unit operations required for purification and making the technology highly attractive for cost-sensitive generic drug manufacturing.
Mechanistic Insights into Whole-Cell Asymmetric Reduction
The core of this technology relies on the stereoselective activity of intracellular carbonyl reductases present within the Geotrichum candidum ZJPH1907 biomass. When the substrate 2-chloro-1-(3,4-difluorophenyl)ethanone penetrates the cell membrane, it encounters the chiral pocket of the reductase enzyme, which facilitates the hydride transfer from the reduced cofactor (NADPH or NADH) to the si-face of the ketone carbonyl group. This specific orientation ensures the formation of the (S)-enantiomer with high fidelity. The presence of a hydrophobic natural deep eutectic solvent, such as a coumarin-thymol mixture, in the reaction medium further enhances the solubility of the fluorinated substrate, increasing the mass transfer rate and preventing substrate inhibition at higher concentrations. This mechanistic understanding is crucial for process engineers aiming to maximize space-time yields in large-scale bioreactors.
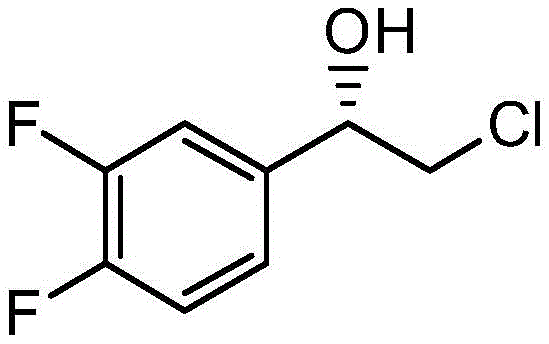
Furthermore, the impurity profile is tightly controlled by the specificity of the biological catalyst, which rarely produces the unwanted (R)-enantiomer or over-reduced byproducts common in chemical hydrogenation. The maintenance of the acidic environment (pH 3.5 to 8.0, optimally 4.2) plays a dual role: it stabilizes the enzyme conformation within the cell and simultaneously acts as a bio-preservative. The addition of surfactants like Tween-80 improves the dispersion of the hydrophobic substrate in the aqueous phase, ensuring uniform exposure to the biocatalyst. This comprehensive mechanistic control results in a clean reaction profile where the primary impurity is unreacted starting material, which is easily separated during the extraction phase, thereby ensuring the final product meets the rigorous purity standards required for pharmaceutical intermediates.
How to Synthesize (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Efficiently
To implement this synthesis route effectively, manufacturers must adhere to the specific fermentation and transformation parameters outlined in the patent data to ensure consistent batch-to-batch reproducibility. The process begins with the cultivation of the ZJPH1907 strain in a nutrient-rich medium containing maltose and beef extract to maximize cell density and enzyme expression. Following harvest, the wet cells are resuspended in a citrate buffer system where the pH is critically adjusted to 4.2 to leverage the acid-tolerance of the strain. Detailed standardized synthesis steps regarding specific inoculation rates, agitation speeds, and extraction protocols are provided in the guide below to assist technical teams in replicating these high-efficiency results.
- Cultivate Geotrichum candidum ZJPH1907 in optimized fermentation media containing maltose, glucose, and beef extract at 30°C to generate wet cell biomass.
- Prepare the biotransformation system by resuspending wet cells in citrate-sodium citrate buffer at pH 4.2, adding glucose as a co-substrate and Tween-80 as a cosolvent.
- Introduce 2-chloro-1-(3,4-difluorophenyl)ethanone substrate and maintain reaction at 30°C with agitation, followed by extraction and purification to isolate the chiral alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this biocatalytic route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive chiral boron reagents and the avoidance of cryogenic reaction conditions translate into substantial cost savings in raw material procurement and energy consumption. Unlike chemical processes that require specialized corrosion-resistant equipment for handling harsh acids or bases, this biological process operates in standard stainless steel fermentation tanks, reducing capital expenditure requirements for new production lines. The simplified workflow also means fewer intermediate storage steps and a reduced footprint for waste treatment facilities, as the aqueous waste streams are generally more biodegradable than those containing heavy metals or organic solvents typical of traditional synthesis.
- Cost Reduction in Manufacturing: The economic advantage of this method is primarily driven by the removal of costly stoichiometric reducing agents and the proprietary chiral ligands associated with chemical synthesis. By utilizing glucose as a cheap co-substrate for cofactor regeneration, the variable cost per kilogram of the intermediate is drastically lowered. Additionally, the high conversion efficiency reduces the amount of starting material lost to side reactions, improving the overall atom economy of the process. This efficiency gain allows manufacturers to offer more competitive pricing for the final API while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain continuity is significantly bolstered by the use of a robust microbial strain that can be stored long-term and scaled up rapidly from shake flasks to industrial fermenters. The reliance on readily available bulk chemicals like glucose and citric acid buffers mitigates the risk of supply disruptions often associated with specialized fine chemical reagents. Furthermore, the ability to operate under non-sterile or semi-sterile conditions due to the acidic pH reduces the downtime required for cleaning and sterilization between batches, thereby increasing the overall equipment effectiveness (OEE) and ensuring timely delivery of critical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the homogeneous nature of the whole-cell suspension and the absence of gas-liquid mass transfer limitations found in hydrogenation reactions. The environmental profile is markedly improved, aligning with modern green chemistry principles by reducing the generation of hazardous waste and lowering the carbon footprint of the manufacturing process. Regulatory compliance is facilitated by the use of a well-characterized biological strain and food-grade additives, simplifying the documentation required for regulatory filings and audits by health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process robustness and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using Geotrichum candidum ZJPH1907 over chemical reduction methods?
A: Unlike chemical methods requiring expensive boron reagents and strict anhydrous conditions, this biocatalytic process operates in aqueous media with self-regenerating cofactors, significantly reducing raw material costs and environmental waste.
Q: How does the acidic reaction environment benefit industrial production?
A: Operating at pH 4.2 naturally suppresses the growth of contaminating bacteria, reducing the risk of batch failure and lowering the requirement for sterile conditions compared to neutral pH fermentations.
Q: What is the expected optical purity of the product using this method?
A: Under optimized conditions with substrate concentrations around 7.6g/L, the process achieves an enantiomeric excess (e.e.) value exceeding 97%, meeting stringent pharmaceutical specifications for chiral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-chloro-1-(3,4-difluorophenyl)ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-quality chiral intermediates for the global cardiovascular drug market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced chiral chromatography to verify enantiomeric excess and chemical purity. Our infrastructure is designed to handle complex biocatalytic processes, guaranteeing consistency and reliability for our long-term partners.
We invite you to collaborate with us to leverage this innovative technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's economic viability and accelerate your time to market.
