Advanced Enzymatic Synthesis of Brivaracetam Intermediates via Engineered OYE1 Mutants
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of high-value antiepileptic drugs, and the recent disclosure in patent CN113444702B represents a significant leap forward in the biocatalytic synthesis of Brivaracetam intermediates. This patent details the development of novel enone reductase mutants, specifically derived from the Old Yellow Enzyme 1 (OYE1) family, which demonstrate exceptional performance in the asymmetric hydrogenation of 4-propyl-2(5H)-furanone. Unlike traditional chemical methods that often struggle with stereoselectivity and environmental impact, this biological approach leverages protein engineering to create a highly specific catalyst, the OYE1-P296A mutant, capable of driving reactions to near-completion with outstanding enantiomeric excess. For R&D directors and process chemists, this technology offers a robust alternative to existing synthetic routes, promising to streamline the manufacturing of this critical third-generation antiepileptic agent while adhering to stricter green chemistry principles.
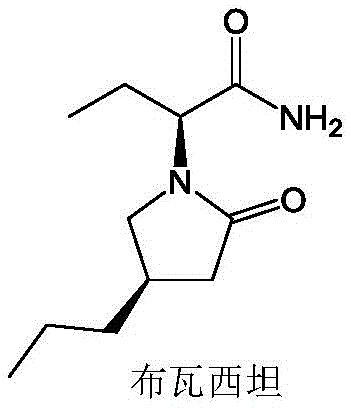
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Brivaracetam and its key chiral intermediates has relied heavily on chemical asymmetric synthesis or chiral resolution techniques, both of which present significant drawbacks for large-scale manufacturing. Chemical asymmetric hydrogenation often necessitates the use of expensive transition metal catalysts, such as rhodium or ruthenium complexes, which not only inflate raw material costs but also introduce the risk of heavy metal contamination that requires rigorous and costly removal steps to meet pharmaceutical standards. Furthermore, chiral resolution methods are inherently inefficient, theoretically limiting the maximum yield to 50% unless dynamic kinetic resolution is employed, which adds further complexity and cost to the process. Existing enzymatic reports prior to this invention often lacked critical data on substrate conversion rates or exhibited insufficient catalytic activity, rendering them impractical for industrial application where high throughput and minimal waste are paramount requirements for economic viability.
The Novel Approach
The innovative strategy outlined in the patent overcomes these hurdles by employing a semi-rational design approach to engineer the OYE1 enzyme, specifically targeting the substrate binding pocket to enhance catalytic efficiency. By mutating the proline residue at position 296 to alanine (P296A), the inventors successfully expanded the active site volume, facilitating easier access for the bulky 4-propyl-2(5H)-furanone substrate. This structural modification results in a dramatic improvement in reaction kinetics, allowing the system to achieve substrate conversion rates exceeding 99% and product enantiomeric excess values greater than 99%. This novel biocatalytic route operates under mild aqueous conditions using a recyclable coenzyme system, effectively eliminating the need for hazardous organic solvents and precious metals, thereby offering a cleaner, safer, and more economically attractive pathway for the reliable pharmaceutical intermediate supplier market.
Mechanistic Insights into OYE1-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the precise manipulation of the enzyme's active site architecture to optimize hydride transfer from the reduced flavin mononucleotide (FMNH2) cofactor to the substrate's carbon-carbon double bond. The wild-type OYE1 enzyme, while capable of reducing activated alkenes, often suffers from steric hindrance when accommodating substrates with larger alkyl chains like the propyl group found in the Brivaracetam precursor. Through homology modeling and molecular docking simulations, the researchers identified that the proline at position 296 creates a steric bottleneck; replacing this rigid cyclic amino acid with the smaller, flexible alanine residue alleviates this constraint. This mutation not only improves the binding affinity (Km) but also enhances the turnover number (kcat), ensuring that the enzyme can process the substrate rapidly and with high fidelity, locking the stereochemistry into the desired (R)-configuration essential for the drug's biological activity.
Impurity control is another critical aspect where this enzymatic method excels, primarily due to the inherent stereospecificity of the biocatalyst which minimizes the formation of unwanted diastereomers or regioisomers common in chemical catalysis. The high enantiomeric excess (>99% ee) achieved directly from the biotransformation step significantly reduces the burden on downstream purification processes, such as crystallization or chromatography, which are often the most costly and time-consuming stages in API manufacturing. Additionally, the integration of an efficient coenzyme recycling system, utilizing alcohol dehydrogenase (lbADH) and isopropanol, ensures that the expensive NADPH cofactor is continuously regenerated in situ. This closed-loop system prevents the accumulation of oxidized byproducts and maintains the redox balance necessary for sustained catalytic activity, thereby ensuring a consistent and high-purity product profile throughout the reaction lifecycle.
How to Synthesize Brivaracetam Intermediate Efficiently
Implementing this advanced biocatalytic process requires a systematic approach to strain construction, fermentation, and biotransformation to fully realize the potential of the OYE1-P296A mutant. The process begins with the cloning of the optimized gene sequence into a robust expression vector, followed by transformation into a high-density fermentation host like E. coli BL21(DE3). Once the engineered strains for both the reductase and the coenzyme recycling enzyme are established, they are cultivated under controlled conditions to maximize biomass and enzyme expression levels. The subsequent catalytic step involves mixing the crude enzyme preparations with the substrate and the recycling system in a buffered aqueous medium, where parameters such as pH, temperature, and substrate loading are carefully optimized to maintain enzyme stability while driving the reaction to completion.
- Construct recombinant E. coli BL21(DE3) strains expressing the OYE1-P296A mutant and the coenzyme recycling enzyme lbADH.
- Ferment the engineered strains to obtain wet cells or crude enzyme preparations, optimizing cell density for catalytic efficiency.
- Perform asymmetric hydrogenation of 4-propyl-2(5H)-furanone using the crude enzyme system with isopropanol for NADPH regeneration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this enzymatic technology translates into tangible strategic benefits that extend beyond simple laboratory metrics. The shift from chemical to biological catalysis fundamentally alters the cost structure of the manufacturing process by removing dependency on volatile markets for precious metals and specialized chiral ligands. Instead, the process relies on fermentable sugars and standard amino acids, commodities that are widely available and subject to much lower price volatility, ensuring a more predictable and stable cost base for long-term supply contracts. Furthermore, the high efficiency of the OYE1-P296A mutant means that less enzyme is required per kilogram of product, reducing the overall fermentation load and associated utility costs, which contributes to significant cost reduction in pharmaceutical intermediate manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal scavenging steps leads to a drastic simplification of the production workflow. By achieving near-quantitative conversion and high stereoselectivity in a single step, the process minimizes the loss of valuable starting materials and reduces the volume of solvent waste generated, resulting in substantial cost savings related to raw material consumption and waste disposal fees.
- Enhanced Supply Chain Reliability: Utilizing a biological system based on E. coli fermentation leverages well-established and globally available infrastructure, reducing the risk of supply disruptions associated with specialized chemical reagents. The robustness of the OYE1-P296A mutant allows for flexible manufacturing schedules and the ability to scale production up or down rapidly in response to market demand, ensuring a continuous and reliable supply of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the use of biodegradable enzymes align perfectly with increasingly stringent environmental regulations, simplifying the permitting process for new manufacturing facilities. The high space-time yield of the process facilitates easy scale-up from pilot to commercial scales, allowing manufacturers to meet growing global demand for Brivaracetam while maintaining a reduced carbon footprint and adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route, providing clarity for stakeholders evaluating this technology for their supply chains. These insights are derived directly from the experimental data and process descriptions found in the patent literature, ensuring that the information provided is grounded in verified scientific evidence rather than theoretical speculation. Understanding these details is crucial for making informed decisions about process adoption and partnership opportunities in the competitive landscape of antiepileptic drug production.
Q: What is the primary advantage of the OYE1-P296A mutant over wild-type enzymes?
A: The OYE1-P296A mutant exhibits significantly improved catalytic activity and substrate binding pocket volume, achieving over 99% conversion and ee values, whereas wild-type or other mutants often show lower activity or incomplete conversion.
Q: How does this enzymatic route impact cost compared to chemical synthesis?
A: This route eliminates the need for expensive chiral metal catalysts and complex resolution steps. The use of a robust coenzyme recycling system further reduces the consumption of costly cofactors, leading to substantial operational cost savings.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes standard E. coli fermentation and operates under mild aqueous conditions. The high conversion rate minimizes downstream purification requirements, making it highly scalable for commercial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Brivaracetam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the OYE1-P296A enzymatic technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to handle complex biocatalytic processes, ensuring that every batch meets stringent purity specifications and regulatory requirements. We understand that transitioning to a new synthetic route involves careful validation, and our team of expert process chemists is dedicated to optimizing every parameter to maximize yield and minimize cost, providing our partners with a secure and efficient source of critical pharmaceutical building blocks.
We invite you to engage with our technical procurement team to discuss how this advanced enzymatic route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the superior quality and reliability of our Brivaracetam intermediate offerings with confidence.
