Revolutionizing Oxaliplatin Production: A Deep Dive into Efficient One-Pot Synthesis Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology agents, and the synthesis of oxaliplatin remains a focal point of process chemistry innovation. Patent CN100540556C introduces a transformative approach to producing this third-generation platinum-based antineoplastic agent, addressing long-standing inefficiencies in legacy protocols. By utilizing a direct one-pot reaction strategy involving cis-dihalo(trans-(-)-1,2-cyclohexanediamine)platinum(II), silver nitrate, and soluble oxalates, this technology streamlines the coordination chemistry required to install the bidentate oxalate ligand. The method operates under mild thermal conditions ranging from 20°C to 100°C and leverages the differential solubility of silver halides to drive the equilibrium forward without necessitating hazardous intermediates like silver oxalate. For R&D directors and process engineers, this represents a significant leap towards operational simplicity and enhanced safety profiles in the production of high-value anticancer intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of oxaliplatin has been plagued by multi-step sequences that introduce unnecessary complexity and potential points of failure. Early methodologies, such as those described in US Patent 4169846A, relied on a sequential process where dichlorocyclohexanediammine platinum(II) was first reacted with silver nitrate, followed by a distinct filtration step to remove silver chloride precipitate, and finally treated with oxalic acid. This fragmentation of the process increases labor intensity, solvent consumption, and the risk of product loss during transfer and isolation stages. Furthermore, alternative routes utilizing pre-formed silver oxalate, as seen in European Patent EP0625523, present severe safety liabilities; silver oxalate is notoriously unstable, prone to decomposition upon exposure to light, and carries a risk of explosion when subjected to thermal stress or mechanical shock. These factors collectively hinder the ability to achieve reliable cost reduction in anticancer drug manufacturing and complicate the supply chain for essential chemotherapy agents.
The Novel Approach
The methodology disclosed in CN100540556C fundamentally reengineers this workflow by consolidating the ligand exchange and precipitation events into a single vessel operation. By introducing silver nitrate and an alkali metal oxalate directly to the platinum diamine precursor in an aqueous medium, the system generates the reactive silver oxalate species in situ, which immediately reacts with the platinum center. This eliminates the need to isolate or handle unstable silver salts externally. The process is conducted under an inert nitrogen atmosphere and dark conditions to preserve product integrity, typically yielding white to off-white crystalline oxaliplatin with exceptional purity levels exceeding 98%. This consolidation not only simplifies the equipment footprint required for production but also drastically reduces the cycle time, making it highly attractive for procurement managers seeking to optimize inventory turnover and production throughput.
Mechanistic Insights into Silver-Mediated Ligand Exchange
The core chemical transformation relies on the high affinity of silver ions for halide ligands, which drives the displacement of chloride or iodide from the platinum coordination sphere. In this mechanism, the silver cation acts as a halide scavenger, precipitating as insoluble silver halide (AgCl or AgI), thereby shifting the equilibrium towards the formation of the aqua-platinum intermediate which subsequently coordinates with the oxalate anion. The choice of the starting halogen is critical; while both chloro and iodo precursors are viable, the iodo analog offers distinct kinetic and thermodynamic advantages due to the extremely low solubility product constant (Ksp) of silver iodide. This ensures that silver ions are quantitatively removed from the solution phase, minimizing the risk of silver contamination in the final API, a critical quality attribute for injectable drugs. The reaction proceeds efficiently in deionized water, leveraging the polarity of the solvent to facilitate ion dissociation and collision frequency between the reactants.
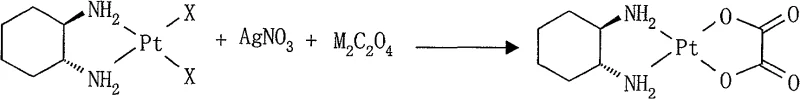
Impurity control is intrinsically built into this mechanistic design through the precise stoichiometric management of the silver and oxalate reagents. The patent data indicates that maintaining a slight deficit of silver nitrate relative to the theoretical requirement prevents the formation of colored impurities often associated with excess silver species or colloidal silver formation. Conversely, a slight excess of the oxalate source ensures that the platinum center is fully saturated with the bidentate ligand, preventing the formation of mono-oxalato species or hydrolysis byproducts. The reaction temperature window of 25°C to 60°C is optimized to balance reaction kinetics with thermal stability, ensuring that the delicate platinum-amine bonds remain intact while providing sufficient energy for the ligand substitution to proceed to completion within a practical timeframe of 4 to 10 hours.
How to Synthesize Oxaliplatin Efficiently
Implementing this synthesis route requires strict adherence to the specified environmental controls and reagent ratios to guarantee the high purity and yield demonstrated in the patent examples. The process begins with the dissolution of the platinum precursor in anaerobic water, followed by the simultaneous or sequential addition of the silver and oxalate sources under rigorous exclusion of light. Detailed standard operating procedures regarding mixing rates, filtration techniques for the fine silver halide precipitate, and crystallization parameters are essential for reproducibility. For a comprehensive breakdown of the exact experimental conditions and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining cis-dihalo(trans-(-)-1,2-cyclohexanediamine)platinum(II), silver nitrate, and a monovalent metal oxalate (such as potassium or sodium oxalate) in anaerobic deionized water.
- Maintain the reaction under nitrogen protection and dark conditions, stirring at a temperature between 20°C and 100°C, preferably 25°C to 60°C, for 2 to 24 hours.
- Upon completion, filter off the silver halide precipitate, concentrate the filtrate to induce crystallization, and wash the resulting oxaliplatin crystals with water and organic solvents before drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this one-pot synthesis technology offers substantial strategic benefits for supply chain resilience and cost structure optimization. By removing the dependency on hazardous and difficult-to-source reagents like silver oxalate, manufacturers can secure a more stable supply of raw materials that are commodity chemicals available from multiple global vendors. The simplification of the process flow directly translates to reduced utility consumption, lower labor costs per kilogram of output, and minimized waste generation, all of which contribute to a leaner manufacturing model. Furthermore, the robustness of the reaction conditions allows for easier scale-up from pilot batches to multi-ton commercial production without the need for specialized explosion-proof infrastructure, thereby lowering capital expenditure barriers for new production lines.
- Cost Reduction in Manufacturing: The elimination of discrete isolation steps for intermediates significantly lowers solvent usage and processing time. By avoiding the synthesis and handling of silver oxalate, the process removes a costly and dangerous unit operation, leading to direct savings in raw material procurement and safety compliance costs. The ability to use cheaper chloride precursors, albeit with slightly different purification needs compared to iodide, provides flexibility in sourcing strategies to further drive down the cost of goods sold (COGS) without compromising the final quality specifications required for pharmaceutical grade material.
- Enhanced Supply Chain Reliability: Utilizing stable, shelf-stable reagents such as silver nitrate and potassium oxalate mitigates the risk of supply disruptions associated with custom-synthesized or unstable intermediates. The shortened reaction timeline and simplified workup reduce the overall manufacturing lead time, enabling faster response to market demand fluctuations for oxaliplatin. This agility is crucial for maintaining continuous supply to downstream formulators and ensuring that critical cancer treatments remain available to patients without interruption due to production bottlenecks.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium aligns well with green chemistry principles, reducing the reliance on volatile organic compounds (VOCs) during the reaction phase. The primary waste stream consists of silver halide salts, which have established recycling pathways for silver recovery, turning a waste product into a value-recovery opportunity. The process is inherently safer, reducing the regulatory burden associated with handling explosive materials, and facilitates smoother environmental impact assessments for facility expansions or new plant constructions.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this oxaliplatin synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on critical process parameters and quality control measures.
Q: Why is the iodine-substituted platinum precursor preferred over the chlorine analog?
A: The iodine-substituted precursor is preferred because silver iodide has significantly lower solubility than silver chloride. This facilitates more complete precipitation and removal of silver ions from the reaction mixture, ensuring higher product purity and reducing the need for complex downstream purification steps.
Q: What are the critical molar ratios for achieving optimal yield and color?
A: While the theoretical ratio is 1:2:1, optimal results are achieved when the silver nitrate dosage is slightly less than theoretical (e.g., ratio of 1:1.8~2.0 relative to platinum) to prevent darkening of the product, while the oxalate dosage should be slightly in excess (e.g., ratio of 1:1.20~1.60 relative to silver) to maximize yield.
Q: How does this method improve safety compared to traditional silver oxalate routes?
A: Traditional methods often require the use of silver oxalate, which is light-sensitive, unstable, and poses explosion risks upon heating or impact. This patented method generates the active species in situ using stable silver nitrate and alkali oxalates, eliminating the hazards associated with handling and storing bulk silver oxalate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaliplatin Supplier
The technological advancements detailed in patent CN100540556C underscore the potential for highly efficient, safe, and scalable production of oxaliplatin intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality material regardless of volume. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying trace metal levels and enantiomeric excess, guaranteeing that every batch meets the demanding standards of the global pharmaceutical market.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain requirements. By leveraging our expertise in platinum chemistry and process optimization, we can help you transition to this superior manufacturing route. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
