Advanced Manufacturing of High-Purity Oxaliplatin: Technical Breakthroughs and Commercial Scalability
Introduction to Next-Generation Oxaliplatin Manufacturing
The global demand for third-generation platinum-based antineoplastic agents continues to surge, driven by their efficacy in treating colorectal cancer. However, the manufacturing of these complex coordination compounds faces stringent regulatory hurdles, particularly regarding heavy metal residues and organic impurities. Patent CN101723988A introduces a transformative methodology for preparing oxaliplatin with exceptionally low impurity profiles, specifically targeting the critical limits of silver (Ag) and free oxalic acid. This technical advancement addresses the European Pharmacopoeia EP5.0 standards, which mandate silver content ≤5ppm and free oxalic acid ≤0.1%, by implementing a refined purification sequence that integrates scavenging steps directly into the synthesis workflow. By leveraging a precise stoichiometric balance between the platinum precursor, silver nitrate, and potassium oxalate, this process ensures that the final active pharmaceutical ingredient (API) meets the rigorous purity specifications required for injectable oncology drugs.
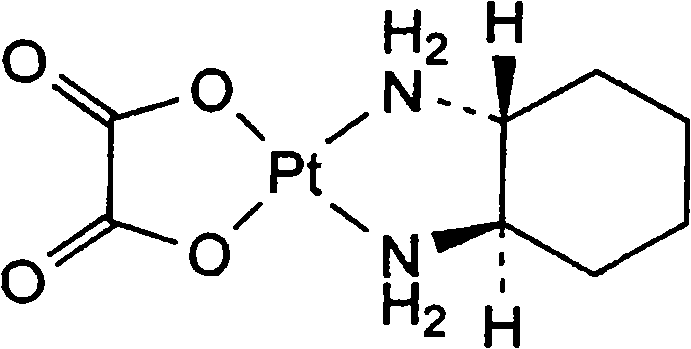
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for oxaliplatin often struggle with the complete removal of silver ions used in the initial activation of the dichloro-platinum precursor. In many legacy processes, residual silver remains trapped within the crystal lattice or as colloidal suspensions, necessitating multiple, yield-reducing recrystallization steps to meet safety standards. Furthermore, conventional methods frequently expose the intermediate aqua-complexes to elevated temperatures or prolonged reaction times, which accelerates the hydrolytic degradation of the oxalate ligand. This degradation leads to elevated levels of free oxalic acid, a toxic impurity that complicates downstream formulation and stability. The reliance on inefficient filtration media in older protocols also fails to capture fine particulate matter, resulting in products that require extensive reprocessing, thereby inflating production costs and extending lead times for pharmaceutical manufacturers.
The Novel Approach
The methodology disclosed in CN101723988A overcomes these historical bottlenecks through a sophisticated, multi-stage purification strategy embedded within the reaction sequence. Instead of relying solely on post-synthesis cleaning, this approach introduces a targeted quenching step using a 10% potassium iodide (KI) solution immediately after the silver-mediated activation. This step quantitatively precipitates excess silver ions as insoluble silver iodide (AgI), effectively scrubbing the reaction matrix of heavy metal contaminants before the final coordination occurs. Additionally, the integration of diatomite filtration at controlled temperatures (30-60°C) ensures the removal of fine particulates and colloidal impurities without inducing thermal degradation. This proactive impurity management allows the subsequent reaction with potassium oxalate monohydrate to proceed in a highly purified environment, yielding a crude product that requires minimal refinement to achieve >99.8% purity.
Mechanistic Insights into Silver-Mediated Ligand Exchange and Scavenging
The core chemical transformation relies on the high affinity of silver ions for chloride ligands to generate a reactive diaqua-platinum intermediate. In this mechanism, silver nitrate reacts with cis-dichloro(trans-(-)-1,2-cyclohexanediamine)platinum, precipitating silver chloride and leaving behind a highly electrophilic platinum center coordinated with water molecules. This aqua-complex is unstable and prone to decomposition if not immediately stabilized. The novelty of this patent lies in the precise temporal control of the subsequent ligand exchange. By maintaining the reaction temperature between 30-70°C during activation and then cooling to 15-45°C for oxalate addition, the process kinetically favors the formation of the stable oxalato-platinum bond while suppressing hydrolysis side reactions. The use of deionized water as the primary solvent medium further minimizes the introduction of extraneous ions that could compete for coordination sites on the platinum center.
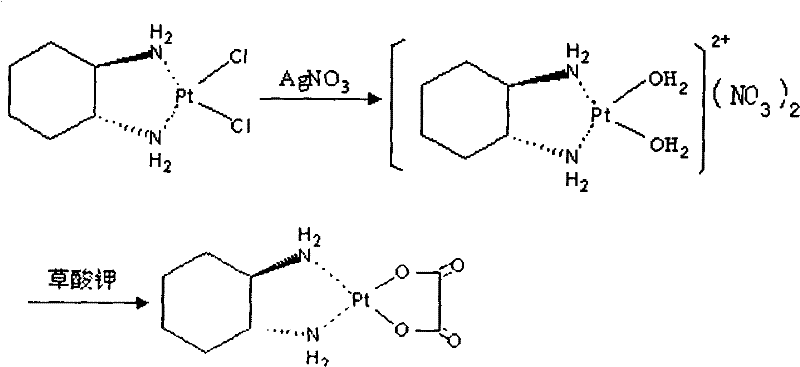
Impurity control is mechanistically achieved through the strategic application of potassium iodide and diatomite. The addition of KI serves as a chemical trap; any unreacted silver nitrate remaining in the filtrate reacts instantly with iodide ions to form a yellow precipitate of AgI, which is easily removed by filtration. This step is critical because residual silver can catalyze the decomposition of the final product during storage. Following this, the addition of diatomite acts as a physical adsorbent, capturing trace organic byproducts and ensuring the clarity of the solution prior to the final crystallization. The final crystallization step, conducted in a 45% ethanol-water mixture at sub-zero temperatures (0 to -20°C), exploits the differential solubility of oxaliplatin versus its impurities, effectively excluding free oxalic acid and remaining salts from the growing crystal lattice, thus locking in the high purity profile observed in the experimental embodiments.
How to Synthesize Oxaliplatin Efficiently
The synthesis of ultra-low impurity oxaliplatin requires strict adherence to the sequential addition of reagents and precise thermal management to ensure optimal yield and purity. The process begins with the dispersion of the platinum precursor in deionized water, followed by the controlled addition of silver nitrate to effect chloride abstraction. Once the silver chloride precipitate is removed, the filtrate undergoes a critical scavenging treatment with potassium iodide to eliminate residual silver, followed by a polishing filtration with diatomite. The purified intermediate is then reacted with potassium oxalate monohydrate under mild thermal conditions to form the final coordination complex. For a detailed breakdown of the specific operational parameters, stoichiometric ratios, and crystallization protocols necessary to replicate this high-purity outcome, please refer to the standardized synthesis guide below.
- Disperse cis-dichloro(trans-(-)-1,2-cyclohexanediamine)platinum in deionized water and react with silver nitrate at 30-70°C to form the diaqua intermediate.
- Treat the filtrate with 10% potassium iodide solution to precipitate excess silver ions, followed by diatomite filtration to ensure clarity.
- React the purified intermediate with potassium oxalate monohydrate at 15-45°C, followed by recrystallization in ethanol-water mixture at sub-zero temperatures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical compliance. The integration of the silver scavenging step directly into the main reaction line eliminates the need for separate, costly purification units or the use of expensive specialized resin columns often required for heavy metal removal in traditional processes. This streamlining of the workflow significantly reduces the overall processing time and labor intensity associated with batch production. Furthermore, the robustness of the crystallization protocol, which tolerates a broad temperature range while maintaining high purity, enhances the reliability of the manufacturing schedule, reducing the risk of batch failures that can disrupt supply continuity. By minimizing the generation of hazardous waste associated with multiple recrystallization solvents, the process also aligns with increasingly stringent environmental regulations, lowering the total cost of ownership for waste disposal and compliance auditing.
- Cost Reduction in Manufacturing: The elimination of complex post-synthesis metal scavenging treatments drastically simplifies the production workflow. By converting residual silver into an easily filterable solid (AgI) early in the process, the method avoids the consumption of expensive polymeric scavengers or activated carbon typically used in downstream purification. This reduction in auxiliary material consumption, combined with the high molar yield reported in the patent embodiments, translates directly into improved gross margins for the manufacturer. Additionally, the use of common, commodity-grade reagents such as silver nitrate and potassium oxalate ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty catalysts.
- Enhanced Supply Chain Reliability: The process demonstrates exceptional consistency across different scales, as evidenced by the reproducible results in the patent examples. The reliance on standard unit operations—filtration, stirring, and crystallization—means that the technology can be transferred to existing multipurpose facilities without requiring significant capital investment in new hardware. This flexibility allows suppliers to rapidly ramp up production capacity in response to market demand surges for anticancer therapies. Moreover, the stability of the intermediate species under the specified conditions reduces the pressure on logistics and timing between reaction steps, providing operators with a wider processing window and reducing the likelihood of time-sensitive batch losses.
- Scalability and Environmental Compliance: From an environmental perspective, the process is designed to minimize the release of heavy metals into the wastewater stream. The quantitative precipitation of silver as AgI allows for the recovery and potential recycling of silver values, turning a waste liability into a recoverable asset. The crystallization step utilizes an ethanol-water system, which is easier to recover and recycle compared to halogenated solvents often found in older organometallic syntheses. This green chemistry approach not only reduces the environmental footprint of the facility but also simplifies the permitting process for expansion, ensuring long-term operational viability in regions with strict ecological oversight.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this low-impurity oxaliplatin synthesis route. These insights are derived directly from the experimental data and process descriptions found in the source patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for R&D teams planning technology transfer and for quality assurance teams establishing control strategies for the new process.
Q: How does this method control silver impurities compared to standard protocols?
A: The process utilizes a specific stoichiometric addition of silver nitrate followed by a targeted quenching step using potassium iodide (KI). This converts residual soluble silver into insoluble silver iodide (AgI), which is removed via filtration, achieving silver levels far below the 5ppm limit.
Q: What are the critical temperature controls for preventing oxalic acid degradation?
A: The patent specifies maintaining the oxalate coordination reaction between 15-45°C. Higher temperatures during the aqueous phase can promote hydrolysis of the oxalate ligand, leading to free oxalic acid impurities, which this protocol strictly avoids through moderate thermal conditions.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrialized production. It employs common reagents like silver nitrate and potassium oxalate, avoids exotic catalysts, and utilizes standard unit operations like filtration and crystallization, facilitating easy scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaliplatin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the production of high-value oncology APIs like oxaliplatin demands not just chemical expertise, but a deep commitment to quality and scalability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We understand that for injectable drugs, stringency is non-negotiable; therefore, our facilities are equipped with rigorous QC labs capable of detecting impurities at the ppm level, guaranteeing that every batch meets stringent purity specifications and complies with international pharmacopoeial standards. Our infrastructure is designed to handle complex organometallic chemistry safely, with dedicated containment systems for potent compounds and advanced waste treatment capabilities.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your supply chain. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional regulatory needs. We encourage you to reach out today to obtain specific COA data from our recent pilot batches and to discuss detailed route feasibility assessments. Let us help you secure a stable, high-quality supply of oxaliplatin that drives down costs while elevating the safety profile of your final drug product.
