Advanced One-Step Catalytic Synthesis of Quinazolinone Derivatives for Commercial Pharmaceutical Manufacturing
Introduction to Patent CN111718301B: A Breakthrough in Quinazolinone Synthesis
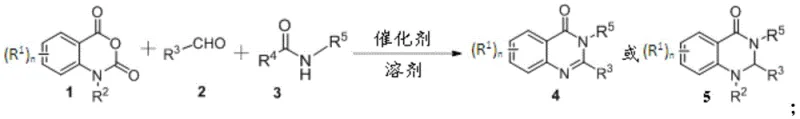
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct privileged scaffolds, and the quinazolinone core remains a cornerstone structure due to its profound biological activities ranging from anticancer to antithrombotic properties. Patent CN111718301B, published in late 2021, introduces a transformative synthetic methodology that addresses long-standing inefficiencies in producing these valuable derivatives. This innovation leverages a one-step multicomponent reaction strategy, utilizing isatoic anhydride or its derivatives, aldehyde compounds, and inert amides as the fundamental building blocks. By streamlining what was traditionally a multi-step, resource-intensive process into a single operational unit, this technology offers a compelling value proposition for manufacturers aiming to optimize their production lines. The method is characterized by its exceptional atom economy and the use of non-toxic, commercially abundant raw materials, which collectively lower the barrier to entry for high-quality intermediate production. Furthermore, the reaction demonstrates remarkable functional group compatibility, allowing for the synthesis of a diverse library of quinazolinone derivatives without the need for extensive protecting group strategies. This patent represents a significant leap forward in process chemistry, providing a robust foundation for the scalable manufacturing of complex heterocyclic compounds essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone backbone has relied on methodologies that are often fraught with operational complexities and environmental drawbacks. Traditional routes frequently involve the ring-closing condensation of anthranilamide with aldehydes, a process that can suffer from poor atom economy and the generation of significant waste streams. More modern approaches have attempted to utilize carbonylation cyclization reactions or oxidative cyclization of anthranilamide derivatives, yet these methods often necessitate the use of expensive transition metal catalysts, harsh oxidizing agents, or coupling reagents that are difficult to remove from the final product. Additionally, many prior art methods require the pre-synthesis of specific reactants, adding extra steps that increase both the time and cost of production. The reliance on heavy metals poses a particular challenge for pharmaceutical applications, where stringent limits on residual metals mandate costly purification procedures. Furthermore, the sensitivity of some traditional methods to moisture or oxygen requires specialized equipment and inert atmospheres, further complicating the scale-up process and increasing capital expenditure for manufacturing facilities. These cumulative disadvantages create a pressing need for a more streamlined, environmentally benign, and cost-effective synthetic alternative.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the technology disclosed in CN111718301B offers a streamlined, one-pot solution that fundamentally reshapes the production landscape for quinazolinone derivatives. This novel approach utilizes a direct three-component coupling of isatoic anhydride, aldehydes, and inert amides, effectively bypassing the need for pre-functionalized intermediates. The reaction proceeds under relatively mild conditions, typically requiring only an acidic catalyst and a suitable solvent, and notably, it can be conducted under an air atmosphere, eliminating the need for expensive inert gas setups. The use of carrier-supported acidic catalysts, such as p-toluenesulfonic acid on activated carbon, not only enhances the reaction efficiency but also facilitates easy catalyst recovery and recycling, thereby minimizing waste and reducing raw material costs. This method exhibits excellent tolerance for a wide range of functional groups, including halogens, alkyl chains, and heteroaromatic rings, enabling the rapid generation of diverse chemical libraries for structure-activity relationship studies. By consolidating multiple synthetic transformations into a single step, this approach drastically reduces the overall process time and energy consumption, making it an ideal candidate for green chemistry initiatives and large-scale industrial adoption.
Mechanistic Insights into Acid-Catalyzed Multicomponent Cyclization
The mechanistic pathway of this synthesis is driven by the activation of electrophilic centers through Brønsted acid catalysis, which orchestrates the sequential assembly of the quinazolinone ring system. Initially, the acidic catalyst activates the carbonyl group of the aldehyde or the isatoic anhydride, enhancing its susceptibility to nucleophilic attack by the amide nitrogen. This initial interaction triggers a cascade of bond-forming events, including the cleavage of the amide bond within the isatoic anhydride structure, which is a critical step highlighted in the patent documentation. The reaction mechanism likely involves the formation of an imine intermediate followed by an intramolecular cyclization that closes the six-membered heterocyclic ring. The presence of the inert amide serves a dual purpose, acting both as a reactant that incorporates the nitrogen substituent and potentially as a solvent or stabilizer for the transition states involved. The robustness of this mechanism is evidenced by its ability to proceed efficiently across a broad temperature range, from ambient conditions up to 130°C, accommodating substrates with varying electronic properties. This flexibility suggests a low activation energy barrier for the rate-determining steps, allowing the reaction to reach completion within reasonable timeframes of 1 to 24 hours. Understanding this mechanistic nuance is vital for process chemists aiming to fine-tune reaction parameters for specific substrate classes, ensuring maximum yield and purity in a commercial setting.
Impurity control in this synthetic route is inherently managed by the high selectivity of the acid-catalyzed pathway and the simplicity of the reaction matrix. Unlike oxidative methods that can generate a plethora of over-oxidized byproducts or radical-induced side reactions, this ionic mechanism tends to produce cleaner reaction profiles. The use of supported catalysts further aids in impurity management by preventing the leaching of catalytic species into the product stream, which is a common source of contamination in homogeneous catalysis. The patent data indicates that simple workup procedures, such as solvent removal followed by column chromatography, are sufficient to isolate the target quinazolinone derivatives in high purity, often exceeding yields of 80% for model substrates. For instance, Example 1 demonstrates an 89% yield for the unsubstituted phenyl derivative, while Example 4 achieves an 87% yield for a pyridyl-substituted analog, showcasing the method's consistency. The absence of heavy metals means that downstream purification does not require specialized scavenging resins, simplifying the manufacturing workflow. This inherent cleanliness of the reaction profile translates directly to reduced processing costs and higher throughput, as less time and material are wasted on extensive purification protocols required to meet regulatory specifications for pharmaceutical intermediates.
How to Synthesize Quinazolinone Derivatives Efficiently
To implement this synthesis effectively, operators should follow the standardized protocol outlined in the patent examples, which emphasizes precise stoichiometric control and temperature management. The general procedure involves charging a reaction vessel with isatoic anhydride, the chosen aldehyde, and the inert amide in a molar ratio that favors complete conversion, typically around 1:1.5 for the aldehyde component. The addition of the supported acidic catalyst, such as activated carbon-loaded p-toluenesulfonic acid, is critical, with loading amounts optimized between 0.02 to 0.08 equivalents relative to the starting anhydride. The reaction mixture is then heated to the specified temperature, often between 100°C and 120°C, and stirred under air for the designated duration. Detailed standard operating procedures for scaling this reaction from gram to kilogram scale are provided in the technical guide below, ensuring reproducibility and safety.
- Combine isatoic anhydride, aldehyde compound, and inert amide in a solvent such as N-methylformamide with an acidic catalyst like activated carbon-supported p-toluenesulfonic acid.
- Heat the reaction mixture to temperatures between 25°C and 130°C under air atmosphere for 1 to 24 hours to facilitate cyclization.
- Remove the solvent via rotary evaporation and purify the crude product using silica gel column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; isatoic anhydride, benzaldehydes, and formamides are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. This abundance ensures price stability and consistent availability, which are critical factors for maintaining uninterrupted production schedules. Furthermore, the elimination of expensive transition metal catalysts and hazardous oxidizing agents significantly lowers the direct material costs associated with each batch. The ability to run the reaction under air atmosphere removes the need for specialized inert gas infrastructure, reducing both capital investment and operational overheads related to gas consumption and monitoring. These factors combine to create a manufacturing process that is not only cheaper to operate but also more resilient to market fluctuations and supply disruptions.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the convergence of low-cost inputs and high-efficiency processing. By utilizing inexpensive, bulk-available starting materials like isatoic anhydride and simple aldehydes, the baseline cost of goods sold is inherently minimized. The use of recyclable, supported acid catalysts eliminates the recurring expense of purchasing fresh catalyst for every batch and avoids the costly disposal fees associated with hazardous chemical waste. Moreover, the one-step nature of the synthesis reduces labor costs and utility consumption, as fewer unit operations such as filtration, drying, and intermediate isolation are required. The high atom economy ensures that a greater proportion of the raw material mass is converted into the final product, minimizing waste generation and maximizing yield per kilogram of input. Collectively, these efficiencies result in a substantially lower cost base for quinazolinone production, allowing companies to improve their profit margins or offer more competitive pricing to downstream customers without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness and simplicity of this synthetic method. Since the reaction tolerates a wide variety of functional groups and does not require ultra-pure or exotic reagents, sourcing flexibility is greatly enhanced. Manufacturers can switch between different grades of solvents or suppliers of aldehydes without needing to re-validate the entire process, providing a buffer against regional shortages or logistics delays. The operational simplicity, characterized by air-stable conditions and straightforward workup procedures, reduces the likelihood of batch failures due to operator error or equipment malfunction. This reliability ensures a steady flow of finished goods, enabling supply chain planners to maintain leaner inventory levels while still meeting delivery commitments. The reduced lead time for production, stemming from the elimination of multi-step sequences, allows for faster response to sudden spikes in demand, making the supply chain more agile and responsive to market dynamics.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard reactor equipment and benign reaction conditions. The absence of highly exothermic steps or unstable intermediates makes the reaction safe to run in large vessels, mitigating the risks often associated with process intensification. From an environmental perspective, the method aligns well with green chemistry principles by avoiding toxic heavy metals and minimizing solvent usage through high concentration reactions. The use of supported catalysts that can be filtered and reused reduces the volume of solid waste generated, simplifying waste management and lowering disposal costs. This environmental friendliness not only helps companies meet increasingly stringent regulatory requirements but also enhances their corporate social responsibility profile. The combination of safety, scalability, and sustainability makes this technology a future-proof choice for long-term manufacturing strategies in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinazolinone synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN111718301B, providing a reliable basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement teams assessing the total cost of ownership. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application, ensuring that all stakeholders have a clear understanding of the process capabilities and limitations.
Q: What are the primary advantages of this quinazolinone synthesis method over traditional routes?
A: This method utilizes a one-step multicomponent reaction involving isatoic anhydride, aldehydes, and inert amides, eliminating the need for multi-step preparation of reactants. It employs non-toxic, cheap, and easily available raw materials with high atom economy and excellent functional group compatibility, significantly simplifying the production process compared to traditional oxidative cyclization or carbonylation methods.
Q: Can the catalyst be recycled in this industrial process?
A: Yes, the patent specifically highlights the use of carrier-supported acidic catalysts, such as p-toluenesulfonic acid loaded on activated carbon, diatomite, or polyaniline. These supported catalysts are easy to recover and recycle, ensuring that the final quinazolinone derivative has no metal residue, which is critical for pharmaceutical applications.
Q: What represents the optimal reaction conditions for scaling up this synthesis?
A: The reaction operates effectively under air atmosphere without the need for inert gas protection, with temperatures ranging from 25°C to 130°C and reaction times between 1 to 24 hours. The use of common solvents like N-methylformamide or DMF and the tolerance for various substituents on the isatoic anhydride and aldehyde make it highly suitable for large-scale commercial preparation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Derivatives Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthetic technology to deliver high-quality quinazolinone derivatives to the global market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest international standards. Our commitment to quality is matched by our dedication to sustainability, as we actively integrate green chemistry principles like those found in this patent into our manufacturing operations to minimize environmental impact. By partnering with us, clients gain access to a reliable supply of complex intermediates produced via cutting-edge, cost-effective methodologies that enhance the overall value of their drug development programs.
We invite potential partners to engage with our technical procurement team to discuss how this specific synthetic route can be tailored to your unique project requirements. Whether you need a Customized Cost-Saving Analysis for your existing supply chain or require specific COA data to validate our material against your internal standards, we are ready to provide comprehensive support. Our experts can also conduct detailed route feasibility assessments to determine the optimal production strategy for your target quinazolinone derivatives, ensuring that you achieve the best balance of cost, speed, and quality. Contact us today to request a quote and discover how NINGBO INNO PHARMCHEM can become your trusted partner in bringing innovative pharmaceutical solutions to market.
