Advanced One-Pot Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
Introduction to Advanced Quinazolinone Synthesis
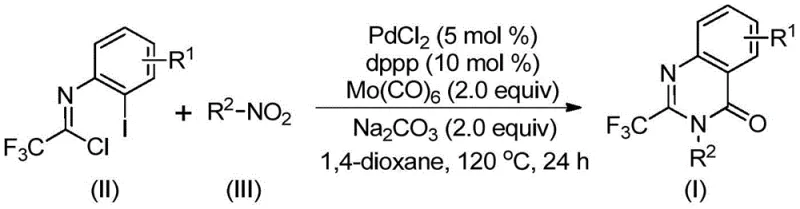
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting broad biological activity. Patent CN112480015B discloses a groundbreaking multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones, a privileged structure found in numerous therapeutic agents ranging from antifungals to anticancer drugs. The introduction of the trifluoromethyl group is strategically critical, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, thereby optimizing pharmacokinetic profiles for drug candidates. This technological breakthrough addresses the longstanding challenges in heterocyclic synthesis by leveraging a palladium-catalyzed carbonylation cascade that operates under relatively mild conditions compared to traditional high-pressure methodologies. For R&D directors and process chemists, this represents a paradigm shift towards more atom-economical and operationally simple protocols that can accelerate the discovery and development of novel pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has relied heavily on methodologies that impose severe logistical and safety burdens on manufacturing facilities. Traditional synthetic pathways often necessitate the use of high-pressure carbon monoxide gas, which requires specialized autoclaves and rigorous safety protocols to prevent leakage and exposure, thereby inflating capital expenditure and operational complexity. Furthermore, many established routes depend on expensive, pre-activated substrates such as 2-bromoformylaniline or acid anhydrides, which not only drive up the cost of goods but also generate substantial stoichiometric waste during the activation steps. Other methods involving iron or ruthenium catalysts frequently suffer from narrow substrate scope and harsh reaction conditions that limit functional group tolerance, making them unsuitable for the late-stage functionalization of complex drug molecules. These inherent inefficiencies create bottlenecks in the supply chain, leading to longer lead times and reduced agility in responding to market demands for diverse chemical libraries.
The Novel Approach
In stark contrast, the methodology described in the patent utilizes a sophisticated yet practical strategy that circumvents these traditional hurdles by employing cheap and readily available nitro compounds as the nitrogen source. By integrating a palladium catalyst system with molybdenum hexacarbonyl as a solid carbon monoxide surrogate, the process effectively generates the necessary carbonyl species in situ, eliminating the need for dangerous high-pressure gas infrastructure. This one-pot cascade reaction seamlessly combines the reduction of the nitro group, intermolecular coupling, and cyclization into a single operational step, drastically simplifying the workflow and minimizing solvent usage. The use of trifluoroethylimidoyl chloride as a key building block ensures the direct incorporation of the valuable trifluoromethyl motif without requiring additional fluorination steps. This streamlined approach not only enhances reaction efficiency but also broadens the applicability of the synthesis to a wide array of substituted derivatives, offering a robust platform for the rapid generation of structural diversity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern transition metal catalysis, involving a intricate sequence of redox and organometallic events that drive the formation of the heterocyclic ring. The reaction is initiated by the reduction of the nitro compound to the corresponding amine species, facilitated by the carbon monoxide released from the thermal decomposition of molybdenum hexacarbonyl. Subsequently, a base-promoted intermolecular coupling occurs between the generated amine and the trifluoroethylimidoyl chloride, forming a trifluoroacetamidine intermediate that serves as the precursor for cyclization. The palladium catalyst then inserts into the carbon-iodine bond of the aromatic ring, generating a reactive divalent palladium species that undergoes migratory insertion with the in situ generated carbon monoxide to form an acyl-palladium intermediate. This acyl species is then poised for intramolecular nucleophilic attack by the nitrogen atom, promoted by the base, which closes the seven-membered palladacycle before undergoing reductive elimination to release the final 2-trifluoromethyl substituted quinazolinone product. Understanding this catalytic cycle is crucial for process optimization, as it highlights the dual role of Mo(CO)6 as both a reductant and a carbonyl source.
From an impurity control perspective, the choice of ligands and bases plays a pivotal role in ensuring high purity and selectivity throughout the reaction course. The use of 1,3-bis(diphenylphosphino)propane (dppp) as the ligand provides the optimal steric and electronic environment to stabilize the palladium center, preventing premature catalyst deactivation and suppressing the formation of homocoupling byproducts. Sodium carbonate acts as a mild yet effective base that facilitates the deprotonation steps required for cyclization without promoting hydrolysis of the sensitive imidoyl chloride starting material. The compatibility of this system with various functional groups, including halogens and alkyl chains, demonstrates a high degree of chemoselectivity, which is essential for maintaining a clean impurity profile in pharmaceutical intermediates. The ability to tolerate diverse substituents on both the nitro compound and the imidoyl chloride allows for the modular synthesis of a wide range of analogs, enabling medicinal chemists to explore structure-activity relationships with minimal synthetic effort.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The execution of this synthesis protocol is designed to be straightforward and accessible, requiring standard laboratory equipment and commercially available reagents that simplify the technology transfer from bench to plant. The procedure involves charging a reaction vessel with the palladium catalyst, ligand, base, carbonyl source, and the two primary organic substrates in an aprotic solvent such as dioxane, followed by heating to 120°C for a defined period. This operational simplicity reduces the training burden on technical staff and minimizes the risk of operator error, which is a critical factor in maintaining consistent quality across different production batches. For detailed procedural specifics regarding stoichiometry, workup, and purification parameters, please refer to the standardized synthesis guide provided below.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 to 30 hours to allow for carbonylation and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic methodology offers tangible strategic benefits that directly impact the bottom line and operational resilience. The shift towards using nitro compounds as starting materials leverages a vast global supply chain of commodity chemicals, ensuring stable pricing and reliable availability even during periods of market volatility. By eliminating the dependency on high-pressure carbon monoxide infrastructure, facilities can avoid the significant capital investments and regulatory compliance costs associated with handling toxic gases, thereby lowering the barrier to entry for manufacturing this high-value scaffold. Furthermore, the one-pot nature of the reaction reduces the number of unit operations, solvent swaps, and isolation steps, which translates to lower utility consumption and reduced waste generation, aligning with increasingly stringent environmental regulations. These factors collectively contribute to a more sustainable and cost-efficient manufacturing model that enhances competitiveness in the global pharmaceutical intermediate market.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the utilization of low-cost nitro compounds and the avoidance of expensive pre-functionalized precursors, which significantly lowers the raw material cost per kilogram of the final product. The replacement of gaseous carbon monoxide with solid molybdenum hexacarbonyl removes the need for specialized high-pressure reactors, allowing production to proceed in standard glass-lined or stainless steel vessels, thus optimizing asset utilization. Additionally, the high reaction efficiency and yield reported in the patent examples suggest that less starting material is wasted, further improving the overall mass balance and reducing the cost of waste disposal. These cumulative savings create a compelling value proposition for large-scale production, enabling suppliers to offer competitive pricing without compromising on quality margins.
- Enhanced Supply Chain Reliability: Sourcing reliability is dramatically improved as the key reagents, including palladium chloride, dppp, and various nitroarenes, are widely produced by multiple chemical manufacturers globally, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a broad range of functional groups, means that supply chain disruptions for specific specialized substrates can often be managed by switching to alternative analogs without redesigning the entire synthetic route. This flexibility ensures continuity of supply for downstream customers who rely on consistent delivery schedules for their own drug development pipelines. Moreover, the scalability of the process from gram to potentially tonnage levels ensures that the supply chain can expand seamlessly to meet increasing demand as a drug candidate progresses through clinical trials.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is favorably positioned due to the atom-economic nature of the carbonylation cascade and the use of recyclable solvents like dioxane. The absence of heavy metal waste streams associated with stoichiometric oxidants or reductants simplifies the effluent treatment process, reducing the load on wastewater treatment facilities and lowering compliance costs. The process has been demonstrated to be scalable, indicating that engineering challenges related to heat transfer and mixing can be effectively managed in larger reactors. This scalability, combined with the reduced hazard profile compared to high-pressure gas methods, makes the technology highly attractive for contract development and manufacturing organizations (CDMOs) seeking to expand their portfolio of safe and green chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic technology, providing clarity for stakeholders evaluating its potential integration into their supply chains. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific project requirements and for anticipating any potential technical hurdles during scale-up.
Q: What are the primary advantages of using nitro compounds in this synthesis?
A: Nitro compounds serve as inexpensive and readily available starting materials that replace costly pre-activated substrates, significantly reducing raw material costs while maintaining high reaction efficiency.
Q: How does this method improve safety compared to traditional carbonylation?
A: By utilizing solid Mo(CO)6 as a carbon monoxide substitute instead of high-pressure CO gas, the process eliminates the need for specialized high-pressure equipment and mitigates significant safety hazards associated with toxic gas handling.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level with simple operation and post-treatment, indicating strong potential for commercial scale-up in pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure required to translate this innovative patent technology into commercial reality for our global clientele. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 2-trifluoromethyl quinazolinone intermediates we produce. We understand the critical importance of quality consistency in the pharmaceutical industry and are committed to delivering products that meet the highest international standards.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development programs.
