Advanced Manufacturing Process for Quinolonyl Lactam Antimicrobial Intermediates and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex antimicrobial agents, particularly those combining distinct pharmacophores like quinolones and lactams. Patent CN1159809A discloses a groundbreaking process for making quinolonyl lactam antimicrobials that addresses longstanding synthetic inefficiencies. Traditional methods often struggle with the solubility of quinolone precursors and the stability of the lactam ring during coupling, leading to suboptimal yields. This innovation introduces a strategic coupling of a compound of Formula (III) with a lactam-containing compound, followed by a crucial cyclization step utilizing an organosilicon compound. 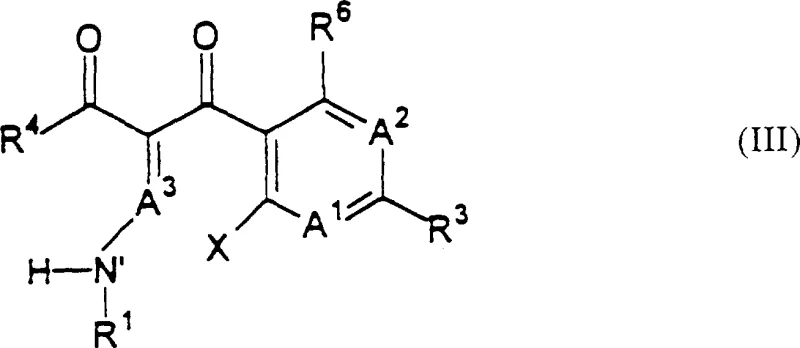 . This specific structural framework allows for the formation of a linking moiety that connects the quinolone and lactam sections without compromising the integrity of either sensitive component. The ability to synthesize these dual-action antimicrobials efficiently is critical for developing next-generation treatments against resistant bacterial strains, positioning this technology as a cornerstone for modern antibiotic development pipelines.
. This specific structural framework allows for the formation of a linking moiety that connects the quinolone and lactam sections without compromising the integrity of either sensitive component. The ability to synthesize these dual-action antimicrobials efficiently is critical for developing next-generation treatments against resistant bacterial strains, positioning this technology as a cornerstone for modern antibiotic development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the ligation of quinolone and lactam moieties has been plagued by significant chemical challenges that hinder commercial viability. Prior art methods frequently rely on harsh reagents and polar solvents, such as water, which induce degradation of the final product and cause insolubility of the components, particularly the quinolone or related heterocyclic parts. These adverse conditions result in low overall yields and necessitate extensive purification protocols that drive up manufacturing costs and extend production timelines. Furthermore, the sensitivity of the beta-lactam ring to nucleophilic attack or hydrolysis under standard coupling conditions often leads to the formation of unwanted impurities, complicating the regulatory approval process for new drug applications. The inability to maintain mild reaction environments in conventional synthesis restricts the scope of compatible substituents, limiting the chemical diversity available for medicinal chemists to optimize biological activity and pharmacokinetic profiles in new antimicrobial candidates.
The Novel Approach
The process outlined in the patent data represents a paradigm shift by employing organosilicon compounds to facilitate the cyclization step under surprisingly efficient and mild reaction conditions. By avoiding the harsh reagents and polar solvents described in prior art, this method ensures that sensitive functional groups in both the reaction substrate and the final product remain intact throughout the synthesis. The improved solubility of the quinolone precursor in the chosen solvent systems allows for homogeneous reaction conditions, which drastically enhances the conversion rates and simplifies downstream processing. This novel approach provides more synthetic flexibility for the preparation of such molecules, enabling the incorporation of diverse linking moieties such as carbamates, secondary amines, or tertiary amines without fear of decomposition. Consequently, manufacturers can achieve higher purity specifications with fewer isolation steps, directly translating to a more streamlined and cost-effective production workflow for high-value pharmaceutical intermediates.
Mechanistic Insights into Organosilicon-Mediated Cyclization
The core of this technological advancement lies in the mechanistic role of the organosilicon compound during the ring-closure phase. In the coupling step, a compound of Formula (III) reacts with a lactam-containing compound to form an intermediate, which is then subjected to cyclization. The organosilicon reagent acts as a activating agent that promotes the intramolecular reaction required to close the quinolone ring system while preserving the adjacent lactam structure. 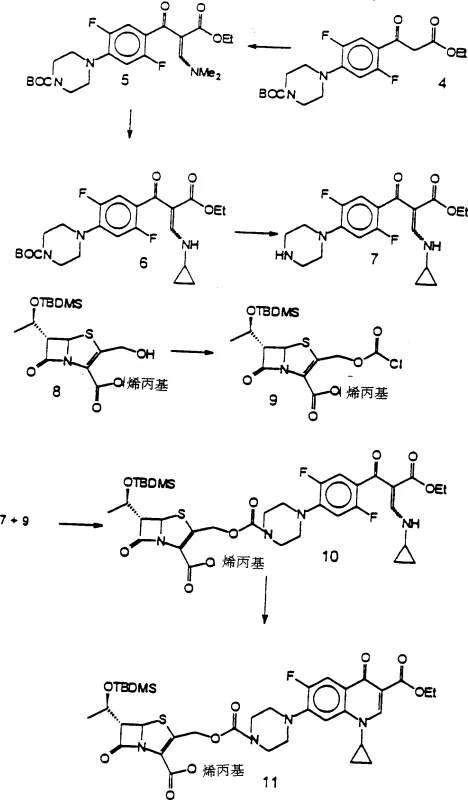 . This mechanism avoids the need for strong bases typically used in quinolone synthesis, which are often incompatible with beta-lactam antibiotics. The reaction conditions are carefully controlled, often at temperatures less than 0°C during coupling and slightly elevated temperatures during cyclization, to maximize selectivity. This precise control over the reaction environment minimizes side reactions such as epimerization at chiral centers, which is a critical quality attribute for antimicrobial efficacy and safety profiles in clinical applications.
. This mechanism avoids the need for strong bases typically used in quinolone synthesis, which are often incompatible with beta-lactam antibiotics. The reaction conditions are carefully controlled, often at temperatures less than 0°C during coupling and slightly elevated temperatures during cyclization, to maximize selectivity. This precise control over the reaction environment minimizes side reactions such as epimerization at chiral centers, which is a critical quality attribute for antimicrobial efficacy and safety profiles in clinical applications.
Furthermore, the versatility of this mechanism extends to the wide range of lactam moieties that can be successfully integrated into the final structure. The process supports the incorporation of penems, carbapenems, cephems, and carbacephems, each bringing unique antibacterial spectrums to the final quinolonyl lactam antimicrobial. 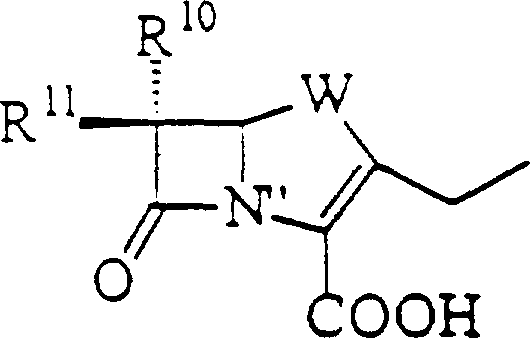 . The compatibility with these varied structures is due to the mildness of the organosilicon-mediated conditions, which do not trigger the ring-opening or degradation pathways common in more aggressive synthetic routes. This broad substrate tolerance allows for the rapid generation of analog libraries for structure-activity relationship studies, accelerating the drug discovery process. For R&D teams, understanding this mechanistic robustness is essential for designing derivatives that overcome specific resistance mechanisms while maintaining manufacturability at a commercial scale.
. The compatibility with these varied structures is due to the mildness of the organosilicon-mediated conditions, which do not trigger the ring-opening or degradation pathways common in more aggressive synthetic routes. This broad substrate tolerance allows for the rapid generation of analog libraries for structure-activity relationship studies, accelerating the drug discovery process. For R&D teams, understanding this mechanistic robustness is essential for designing derivatives that overcome specific resistance mechanisms while maintaining manufacturability at a commercial scale.
How to Synthesize Quinolonyl Lactam Antimicrobials Efficiently
Implementing this synthesis route requires careful attention to the sequence of protection, coupling, and deprotection steps to ensure optimal yield and purity. The process begins with the preparation of the quinolone precursor and the lactam component, often requiring protective groups on carboxyl, hydroxyl, and amino functionalities to prevent unwanted side reactions during the coupling phase. Once the intermediates are prepared, they are coupled in a suitable solvent system such as dichloromethane or tetrahydrofuran, followed by the critical addition of the organosilicon compound to induce cyclization. . The final stages involve rigorous deprotection and purification to isolate the pharmaceutically acceptable salt or biohydrolyzable ester. Adhering to these standardized protocols ensures that the complex molecular architecture is assembled correctly, providing a reliable foundation for scaling up production to meet global demand for advanced antimicrobial therapies.
- Couple a compound of Formula (III) with a lactam-containing compound to form an intermediate compound under controlled temperature conditions.
- Cyclize the intermediate by reaction with an organosilicon compound to yield the final quinolonyl lactam antimicrobial structure.
- Perform deprotection steps after cyclization to remove protecting groups and isolate the pharmaceutically acceptable active product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers substantial strategic benefits beyond mere technical feasibility. The elimination of harsh reagents and the reduction in purification complexity directly contribute to significant cost reduction in pharmaceutical intermediates manufacturing. By minimizing the number of unit operations and avoiding expensive heavy metal catalysts or difficult-to-remove impurities, the overall cost of goods sold is optimized without compromising quality standards. This efficiency gain allows for more competitive pricing models when sourcing these critical building blocks, enabling pharmaceutical companies to allocate resources towards further clinical development or market expansion initiatives. Additionally, the robustness of the process reduces the risk of batch failures, ensuring a more predictable expenditure profile for raw materials and processing time.
- Cost Reduction in Manufacturing: The streamlined synthesis pathway eliminates the need for multiple protection and deprotection cycles that are typical in conventional methods, leading to substantial cost savings in reagents and labor. The use of organosilicon compounds facilitates a cleaner reaction profile, which reduces the burden on waste treatment facilities and lowers the environmental compliance costs associated with hazardous solvent disposal. Furthermore, the higher yields achieved through this method mean that less starting material is required to produce the same amount of active intermediate, maximizing the return on investment for every kilogram of raw material purchased. These cumulative efficiencies create a leaner manufacturing model that is highly resilient to fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The improved solubility and stability of intermediates during synthesis reduce the likelihood of production bottlenecks caused by precipitation or decomposition issues. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug formulation teams receive their materials on schedule. The process utilizes readily available organosilicon reagents and common organic solvents, mitigating the risk of supply disruptions associated with specialty or scarce chemicals. By establishing a more stable production rhythm, manufacturers can better forecast inventory levels and maintain safety stocks, thereby safeguarding the continuity of supply for critical antimicrobial medications in the global market.
- Scalability and Environmental Compliance: The mild reaction conditions inherent to this process make it exceptionally well-suited for the commercial scale-up of complex pharmaceutical intermediates from laboratory to industrial tonnage. The avoidance of extreme temperatures and pressures simplifies the engineering requirements for reactor vessels, allowing for easier technology transfer between sites. Moreover, the reduced generation of hazardous byproducts aligns with increasingly stringent environmental regulations, facilitating smoother permitting processes for new manufacturing lines. This alignment with green chemistry principles not only enhances corporate sustainability profiles but also future-proofs the supply chain against evolving regulatory landscapes regarding chemical manufacturing and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers are derived directly from the technical specifications and beneficial effects documented in the patent literature, ensuring accuracy and relevance for decision-makers. . These insights provide a clear picture of how this innovation translates into tangible operational improvements and competitive advantages in the fine chemical sector.
Q: What are the primary advantages of using organosilicon compounds in this synthesis?
A: The use of organosilicon compounds allows for cyclization under milder reaction conditions compared to prior art, significantly improving product yield and purity while preventing degradation of sensitive functional groups.
Q: Which lactam moieties are compatible with this manufacturing process?
A: This process is highly versatile and supports various lactam moieties including penems, carbapenems, cephems, carbacephems, and monocyclic beta-lactams, offering broad application potential.
Q: How does this method impact supply chain reliability for antimicrobial intermediates?
A: By eliminating harsh reagents and polar solvents that cause insolubility issues, this method ensures consistent batch-to-batch quality and reduces processing time, thereby enhancing overall supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinolonyl Lactam Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate complex patent methodologies like CN1159809A into reality for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of quinolonyl lactam intermediate meets the highest international standards. Our commitment to quality assurance means that you can rely on us as a reliable pharmaceutical intermediates supplier for your most critical antibiotic development projects, minimizing risk and accelerating time to market.
We invite you to contact our technical procurement team to discuss how we can tailor this synthesis route to your specific needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this advanced manufacturing process. We are ready to provide specific COA data and route feasibility assessments to support your internal evaluation. Partner with us to leverage our deep domain knowledge and state-of-the-art facilities for the production of high-purity quinolonyl lactam, securing a competitive edge in the rapidly evolving antimicrobial landscape.
