Optimizing Eribulin Intermediate Production: A Novel Low-Chromium High-Yield Route
Introduction to Advanced Eribulin Intermediate Manufacturing
The global demand for high-purity oncology therapeutics continues to surge, placing immense pressure on the supply chains of complex marine natural product derivatives like Eribulin Mesylate, commercially known as Halaven. As a potent microtubule dynamics inhibitor approved for metastatic breast cancer, the commercial viability of Eribulin hinges entirely on the efficiency of its synthetic route. Patent CN116159601A introduces a transformative methodology for synthesizing key eribulin intermediates, specifically addressing the historical bottlenecks of low total yield and excessive heavy metal consumption. This technical disclosure outlines a robust pathway that leverages 4-methylcyclohexanone as a superior hydroxyl protecting group and implements a modified Nozaki-Hiyama-Kishi (NHK) reaction.
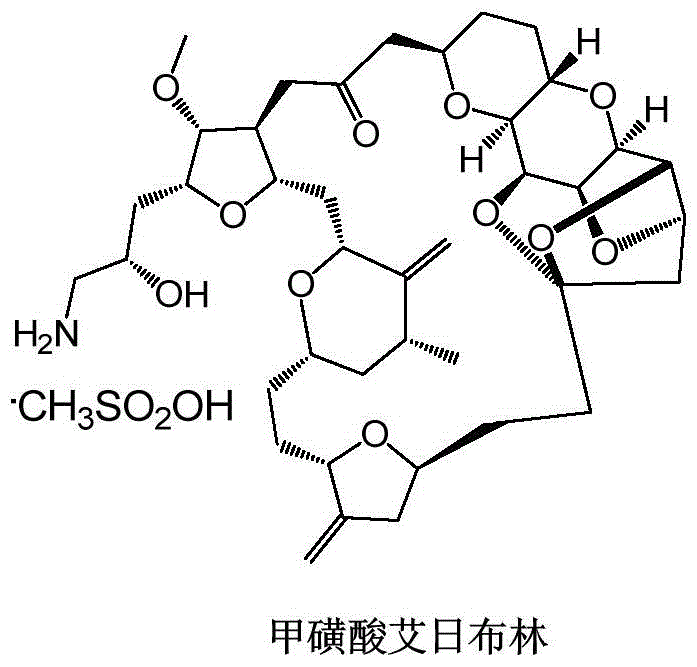
For procurement executives and R&D directors, the implications of this patent are profound. The traditional synthesis routes, often characterized by cumbersome step counts and hazardous reagent profiles, have long hindered the cost-effective scale-up of this life-saving medication. By integrating Lewis acid catalysis and optimizing stereochemical control during dihydroxylation steps, the disclosed method not only enhances the purity profile of the intermediates but also aligns with modern green chemistry mandates. This report analyzes the technical nuances of this innovation, demonstrating how it serves as a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eribulin key intermediates has been plagued by inefficiencies that render large-scale production economically challenging. As detailed in foundational literature such as WO9965894 and reports by Kishi et al., the conventional approach relies on protecting the hydroxyl groups of the starting material, D-gulonolactone, using cyclohexanone. While chemically feasible, this legacy method suffers from a critically low overall yield, reported at merely 19.2% for the synthesis of the crucial Compound B fragment. Furthermore, the reliance on stoichiometric amounts of chromium reagents in the NHK coupling reaction presents a dual burden: exorbitant raw material costs and severe environmental liabilities due to hexavalent chromium waste.
The conventional NHK reaction typically demands chromium reagent dosages ranging from 400 to 1600 mol percent, creating a massive waste stream that requires expensive remediation protocols. For a supply chain manager, this translates to volatile pricing and potential regulatory hurdles regarding heavy metal discharge. Additionally, the poor stereocontrol in earlier steps often leads to difficult-to-separate isomers, necessitating additional purification cycles that further erode yield and extend lead times. These structural weaknesses in the legacy process highlight the urgent need for a redesigned synthetic strategy that prioritizes both atom economy and operational safety.
The Novel Approach
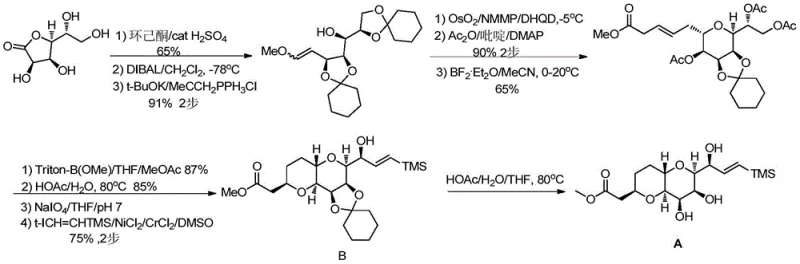
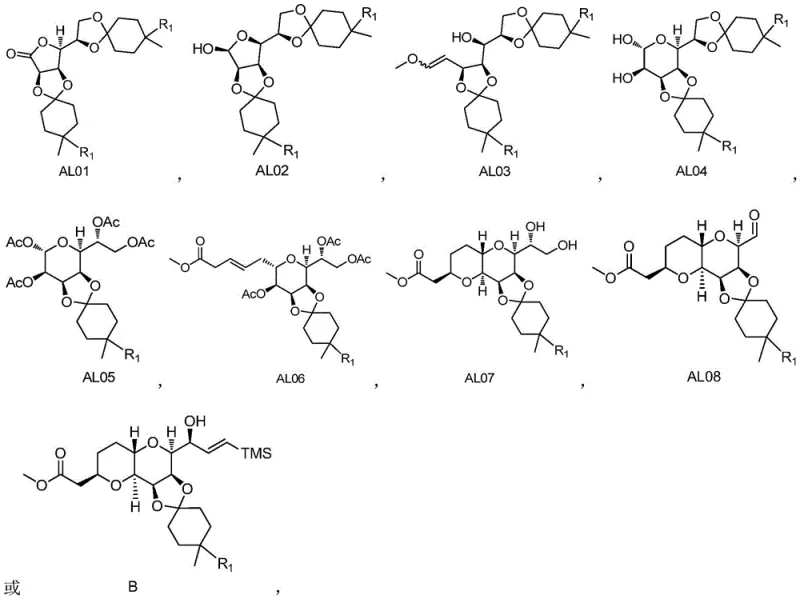
The methodology disclosed in CN116159601A represents a paradigm shift in eribulin intermediate manufacturing by fundamentally re-engineering the protection and coupling strategies. The core innovation lies in the substitution of cyclohexanone with 4-methylcyclohexanone (or its derivatives) for the initial protection of D-gulonolactone. This seemingly minor structural modification exerts a significant influence on the reaction thermodynamics, boosting the yield of the initial condensation product (Compound AL01) from 65% to an impressive 87%. This improvement cascades through the entire synthesis, ultimately elevating the total yield of Compound B to 33.7%, nearly doubling the efficiency of the prior art.
Beyond yield enhancement, the novel approach introduces a sophisticated catalytic system for the critical carbon-carbon bond formation steps. By incorporating nickel salts (NiBr2 or NiCl2) alongside aluminum scraps and trimethylchlorosilane, the process drastically curtails the requirement for chromium reagents. The patent specifies that chromium usage can be reduced to as low as 0.1-0.3 molar equivalents, a staggering reduction compared to the multi-equivalent loads of the past. This modification not only slashes raw material costs but also simplifies the downstream workup, as the removal of trace heavy metals becomes significantly more manageable, ensuring a high-purity eribulin intermediate suitable for stringent pharmaceutical applications.
Mechanistic Insights into Lewis Acid Catalysis and Modified NHK Coupling
The success of this novel synthetic route is underpinned by precise mechanistic controls, particularly in the initial protection and the final coupling stages. The condensation of D-gulonolactone with 4-methylcyclohexanone is catalyzed by anhydrous zinc chloride, a mild yet effective Lewis acid. Operating at temperatures between 120-150°C, the zinc chloride facilitates the formation of the ketal linkage while minimizing side reactions that could degrade the sensitive lactone ring. The presence of the methyl group on the cyclohexanone ring introduces subtle steric bulk that appears to favor the formation of the desired kinetic product, thereby enhancing the diastereomeric ratio in subsequent steps, such as the conversion of AL03 to AL04 where the isomer ratio improves from 4-5:1 to 8-9:1.
In the terminal stage of the synthesis, the modified Nozaki-Hiyama-Kishi (NHK) reaction serves as the linchpin for constructing the complex carbon skeleton. Unlike traditional methods that rely solely on chromium(II) for the reductive coupling of aldehydes with vinyl halides, this patent employs a synergistic Ni-Cr-Al system. The nickel catalyst acts as the primary mediator for the oxidative addition into the carbon-halogen bond, while the aluminum scraps serve as the terminal reductant to regenerate the active low-valent metal species. This cooperative catalysis allows the reaction to proceed with high efficiency even when chromium is present in catalytic or near-stoichiometric quantities (0.1-0.3 equivalents). This mechanistic refinement is crucial for cost reduction in pharmaceutical intermediate manufacturing, as it decouples the reaction efficiency from the massive excess of toxic reagents.
How to Synthesize Eribulin Intermediate B Efficiently
The synthesis of Compound B, a pivotal precursor to Eribulin, is achieved through a concise nine-step sequence that balances operational simplicity with high stereochemical fidelity. The process begins with the Lewis acid-catalyzed protection of D-gulonolactone, followed by a carefully controlled reduction using DIBAL-H to generate the lactol. Subsequent functionalization involves a Wittig olefination to install the methoxymethyl group, followed by a highly stereoselective dihydroxylation using osmium tetroxide and N-methylmorpholine N-oxide. The detailed standardized synthesis steps for this high-yield route are outlined in the guide below.
- Condense D-gulonolactone with 4-methylcyclohexanone using anhydrous zinc chloride to form protected lactone AL01.
- Perform sequential reduction, Wittig olefination, and osmium-catalyzed dihydroxylation to generate tetraol intermediate AL04.
- Execute selective acetylation, Lewis acid-mediated coupling, ring closure, oxidation, and finally a modified Nozaki-Hiyama-Kishi (NHK) reaction to yield Compound B.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the procurement of specialty chemicals and API intermediates, the adoption of the technology described in CN116159601A offers compelling economic and logistical benefits. The primary advantage lies in the substantial cost savings derived from yield optimization and reagent minimization. By nearly doubling the overall yield of the key intermediate, manufacturers can significantly reduce the volume of starting materials required per kilogram of final product. This efficiency gain directly translates to a lower cost of goods sold (COGS), providing a buffer against raw material price volatility and enabling more competitive pricing structures for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of excessive chromium reagents is a major driver for cost optimization. Traditional NHK reactions consume vast quantities of chromium salts, which are not only expensive but also incur high disposal costs due to their classification as hazardous waste. By reducing chromium usage to trace catalytic levels through the Ni-Al co-catalyst system, the process removes the need for expensive heavy metal scavenging resins and complex wastewater treatment protocols. This streamlining of the purification workflow results in significant operational expenditure reductions without compromising the quality of the output.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route enhances supply chain continuity by reducing the risk of batch failures associated with difficult purifications. The improved isomer ratios achieved during the dihydroxylation and protection steps mean that less material is lost to chromatographic separation or recrystallization. For supply chain heads, this predictability is invaluable; it ensures consistent batch-to-batch quality and reduces the lead time for high-purity pharmaceutical intermediates. Furthermore, the use of more stable and readily available reagents like 4-methylcyclohexanone mitigates the risk of supply disruptions common with specialized or hazardous reagents.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on heavy metal emissions, the ability to manufacture complex intermediates with minimal environmental footprint is a strategic asset. The drastic reduction in chromium waste aligns the manufacturing process with green chemistry principles, facilitating easier regulatory approval and permitting for production facilities. This environmental compliance future-proofs the supply chain against evolving legislation, ensuring long-term viability. Additionally, the simplified workup procedures inherent in this high-yield route make the process inherently more scalable, allowing for seamless transition from pilot plant to multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel eribulin intermediate synthesis. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical advantages of this methodology.
Q: How does the new protection strategy improve yield compared to prior art?
A: By replacing cyclohexanone with 4-methylcyclohexanone for hydroxyl protection, the steric and electronic environment is optimized, increasing the yield of the initial condensation step from 65% to 87% and improving downstream isomer ratios.
Q: What is the environmental benefit of the modified NHK reaction?
A: The novel process drastically reduces chromium reagent usage from 400-1600 mol% in conventional methods to merely 0.1-0.3 molar equivalents by introducing a nickel-aluminum co-catalytic system, significantly lowering heavy metal waste.
Q: What is the overall yield improvement for Compound B?
A: The cumulative effect of optimized protection groups and improved stereoselectivity raises the total yield for preparing key intermediate Compound B from D-gulonolactone from 19.2% in prior art to 33.7%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate Supplier
The technological advancements detailed in patent CN116159601A underscore the complexity and sophistication required to produce high-value oncology intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such innovative routes to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of eribulin intermediate we produce adheres to the highest global pharmaceutical standards. We understand that consistency and quality are non-negotiable in the life sciences sector.
We invite procurement leaders and R&D teams to collaborate with us to leverage this advanced synthetic technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project benefits from the latest developments in efficient, eco-friendly pharmaceutical manufacturing.
