Advanced Synthetic Strategy for Relugolix: Enhancing Purity and Scalability for Global API Supply Chains
The pharmaceutical landscape for gonadotropin-releasing hormone (GnRH) antagonists has been significantly advanced by the development of Relugolix, a potent small molecule indicated for uterine fibroids and prostate cancer. Patent CN114031626A introduces a transformative synthetic methodology that addresses critical bottlenecks in the manufacturing of this high-value active pharmaceutical ingredient (API). By optimizing the final functionalization steps, specifically the conversion of the urea intermediate to the final drug substance, this technology delivers a robust pathway characterized by exceptional purity and operational simplicity. For global supply chain stakeholders, this represents a pivotal shift towards more reliable and efficient production capabilities, mitigating the risks associated with complex multi-step syntheses.
Historically, the synthesis of Relugolix has been plagued by significant technical hurdles that impede commercial viability. Prior art, such as the route disclosed in CN104703992B, suffered from severe processing issues where the cyclization reaction generated foamy solids, making mechanical stirring extremely difficult and necessitating excessive solvent volumes. Furthermore, the use of carbonyldiimidazole in the urea formation step led to the generation of approximately 2.0% of a persistent urea condensation by-product, which proved increasingly difficult to remove upon scale-up. Another approach, detailed in CN202010227369.1, attempted to mitigate impurity profiles but resulted in a suboptimal overall yield of only 54%, rendering it economically unfeasible for large-scale procurement.
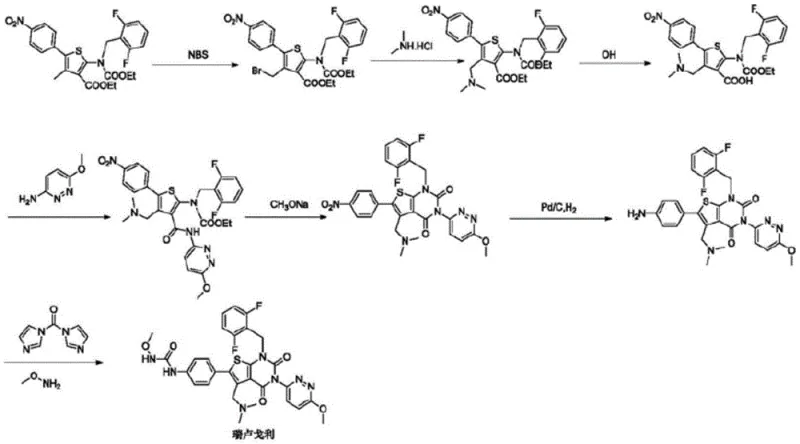
The novel approach presented in the subject patent fundamentally re-engineers the terminal steps of the synthesis to overcome these legacy deficiencies. Instead of relying on problematic condensing agents or harsh halogenation conditions, the new method employs a specific halogenation strategy using 1-chloroethyl chloroformate to activate the intermediate, followed by a clean amination step. This strategic modification not only eliminates the formation of foamy solids but also drastically reduces the generation of hard-to-remove impurities. The result is a streamlined process that operates under mild conditions, typically at room temperature, thereby reducing energy consumption and equipment stress while maximizing the recovery of high-purity product suitable for clinical and commercial applications.
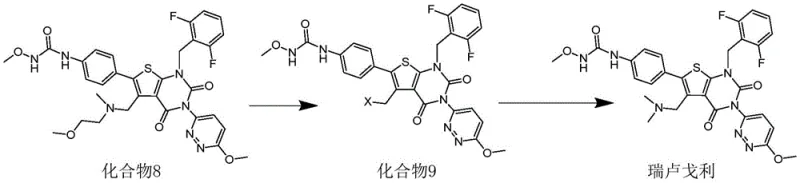
The mechanistic superiority of this synthesis lies in the precise selection of the halogenating reagent and the subsequent nucleophilic substitution. In the critical conversion of Compound 8 to Compound 9, the use of 1-chloroethyl chloroformate facilitates a highly efficient activation of the methyl group without inducing side reactions common with aggressive reagents like thionyl chloride or phosphorus oxychloride. Experimental data confirms that this specific reagent achieves a yield of 95.1% and a purity of 98.5% for the halo-intermediate, whereas alternative reagents yielded significantly lower purities and required more rigorous purification protocols. This selectivity is paramount for maintaining the integrity of the sensitive thieno[2,3-d]pyrimidine core structure throughout the synthesis.
Furthermore, the final amination step demonstrates remarkable efficiency due to the optimized leaving group installed in the previous stage. By reacting the chloromethyl intermediate (Compound 9) with dimethylamine in the presence of an acid-binding agent like N,N-diisopropylethylamine, the reaction proceeds to completion with a yield of 97.7% and a final purity of 99%. This high level of conversion minimizes the burden on downstream purification processes, such as column chromatography or recrystallization, which are often the most costly and time-consuming aspects of API manufacturing. The ability to achieve such high purity directly from the reaction mixture underscores the chemical elegance and practical utility of this patented route for industrial implementation.
How to Synthesize Relugolix Efficiently
The implementation of this synthetic route requires strict adherence to the optimized reaction parameters defined in the patent examples to ensure reproducibility and safety. The process begins with the preparation of the key urea intermediate, Compound 8, which is subsequently subjected to the novel halogenation protocol. Operators must maintain precise temperature controls, particularly during the addition of the halogenating agent, to prevent exothermic runaways and ensure the formation of the desired chloromethyl species. The subsequent amination step is equally critical, requiring anhydrous conditions and appropriate stoichiometric ratios of dimethylamine to drive the reaction to completion without generating secondary amines or over-alkylated by-products.
- Perform a halogenation reaction on Compound 8 using 1-chloroethyl chloroformate in dichloromethane at room temperature to obtain Compound 9.
- React the resulting Compound 9 with dimethylamine in the presence of N,N-diisopropylethylamine to cyclize and form the final Relugolix product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers tangible benefits that extend beyond mere chemical yield. The elimination of problematic physical phenomena, such as foaming and difficult stirring, directly translates to enhanced batch consistency and reduced downtime in manufacturing facilities. Traditional methods often required specialized agitation equipment or dilute conditions to manage rheological issues, which increased solvent costs and reduced reactor throughput. By resolving these engineering challenges at the molecular level, this new process allows for higher concentration reactions and faster cycle times, effectively increasing the production capacity of existing infrastructure without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial improvement in overall yield and the reduction of raw material waste. By achieving a total yield of 67.4% compared to the 54% of previous methods, the consumption of expensive starting materials and intermediates is significantly lowered per kilogram of final API produced. Additionally, the avoidance of transition metal catalysts in the final steps and the use of readily available reagents like 1-chloroethyl chloroformate contribute to a leaner cost structure. The high purity of the crude product further reduces the reliance on costly purification resins and solvents, leading to comprehensive cost optimization across the entire manufacturing value chain.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which are mild and tolerant of minor variations in operating parameters. The use of stable, commercially available reagents ensures that the supply chain is not vulnerable to the shortages often associated with exotic or highly specialized catalysts. Furthermore, the simplified work-up procedures, which avoid complex extractions or difficult filtrations caused by foaming, reduce the risk of batch failures and delays. This reliability is crucial for meeting the stringent delivery schedules required by global pharmaceutical partners and regulatory bodies.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, addressing the specific pain points that arise when moving from laboratory to pilot and production scales. The absence of foaming issues allows for the safe use of standard stainless steel reactors at larger volumes, facilitating a smoother technology transfer. From an environmental perspective, the higher atom economy and reduced solvent usage align with green chemistry principles, minimizing the generation of hazardous waste streams. This compliance with environmental standards simplifies waste disposal logistics and reduces the regulatory burden on manufacturing sites, ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Relugolix synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of this approach. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new Relugolix synthesis method?
A: The method described in patent CN114031626A offers a total yield of 67.4% and final product purity of 99%, significantly outperforming previous methods which suffered from yields as low as 54% and difficult-to-remove impurities.
Q: How does this process address scalability issues found in prior art?
A: Unlike previous routes that generated foamy solids causing stirring difficulties, this novel approach utilizes mild reaction conditions and specific halogenating reagents that prevent foaming, ensuring smooth operation in large-scale commercial reactors.
Q: What specific reagent drives the high efficiency in the final steps?
A: The use of 1-chloroethyl chloroformate as the halogenating agent in the conversion of Compound 8 to Compound 9 is critical, providing a 95.1% yield and 98.5% purity, superior to thionyl chloride or phosphorus oxychloride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Relugolix Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global API market. Our team of expert chemists has thoroughly analyzed the methodology described in CN114031626A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Relugolix and its intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a seamless transition to a more efficient and reliable source for your Relugolix requirements.
