Advanced Relugolix Manufacturing: Scalable High-Purity Synthesis for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing routes for critical therapeutics like Relugolix, a potent gonadotropin-releasing hormone (GnRH) receptor antagonist used in treating advanced prostate cancer and uterine fibroids. Patent CN114907370A introduces a transformative preparation method for high-purity thienopyrimidine compounds, specifically addressing the longstanding challenges of impurity control and reagent toxicity in existing synthesis pathways. This innovation enables the production of the target compound, often referred to as Formula N-a, with exceptional chemical purity exceeding 99.5% without relying on toxic condensing agents or harsh alkaline conditions. The strategic shift towards using acyl chlorides and inorganic bases represents a significant leap forward in process chemistry, offering a viable solution for commercial scale-up that aligns with modern environmental and quality standards. 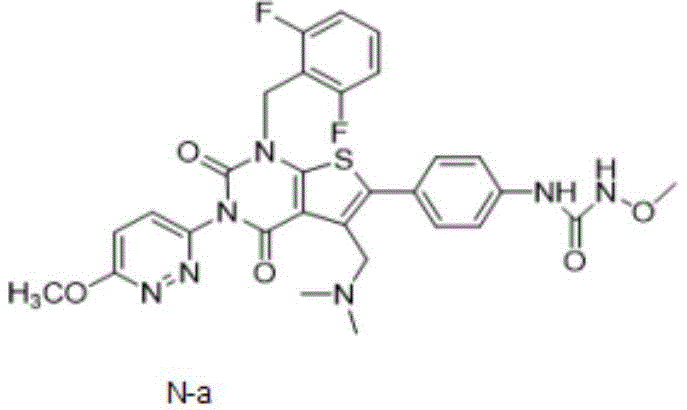
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thienopyrimidine derivatives has relied heavily on condensing agents such as diethyl cyanophosphate (DEPC) or propylphosphonic anhydride (T3P), which present severe drawbacks for large-scale manufacturing. These reagents are not only prohibitively expensive but also highly toxic, generating substantial phosphorus-containing wastewater that complicates environmental compliance and waste treatment protocols. Furthermore, conventional ring-closing steps often utilize strong alkali metal alkoxides, such as sodium ethoxide, which induce undesirable side reactions leading to dark-colored reaction mixtures and complex impurity profiles. The use of such harsh conditions frequently results in the formation of degradation byproducts like Impurity I and Impurity L, necessitating costly and inefficient chromatographic purification steps that hinder industrial throughput. 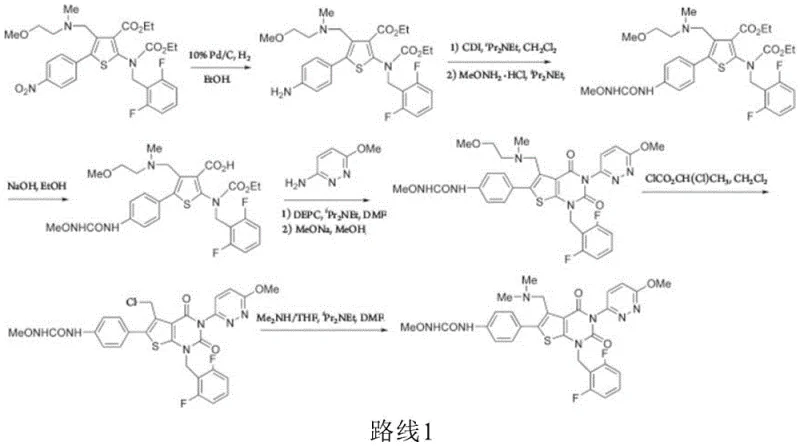
The Novel Approach
The methodology disclosed in CN114907370A circumvents these issues by employing acyl chlorides, such as thionyl chloride or benzenesulfonyl chloride, in the presence of organic bases for the initial condensation reaction. This substitution drastically reduces raw material costs and eliminates the generation of phosphorus waste, thereby enhancing the overall environmental profile of the synthesis. Subsequent ring closure is achieved using mild inorganic bases like potassium carbonate or cesium carbonate in inert solvents, which effectively suppresses the formation of alkali-sensitive degradation impurities. This refined approach allows for the isolation of high-purity intermediates and final products through simple filtration and recrystallization, completely removing the need for column chromatography and significantly streamlining the production workflow for commercial applications. 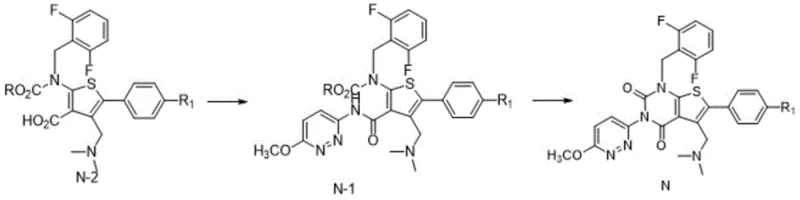
Mechanistic Insights into Acyl Chloride-Mediated Condensation
The core of this technological advancement lies in the activation of the carboxylic acid group in Formula N-2 using acyl chlorides, which facilitates a highly efficient nucleophilic attack by 3-amino-6-methoxypyridazine. Unlike phosphorus-based coupling reagents that leave behind difficult-to-remove byproducts, the acyl chloride mechanism generates soluble salts that are easily removed during the aqueous work-up phase. The reaction is conducted at controlled low temperatures, typically between -10°C and 20°C, which kinetically favors the formation of the desired amide bond while minimizing thermal degradation of sensitive functional groups. This precise control over reaction thermodynamics ensures that the intermediate Formula N-1 is obtained with high conversion rates and minimal side products, setting a clean foundation for the subsequent cyclization step.
Impurity control is further optimized during the ring-closing phase by avoiding strong nucleophilic bases that typically attack the urea side chain or the thienopyrimidine core. By utilizing inorganic carbonates or phosphates, the process mitigates the risk of forming specific degradation impurities such as Impurity G, Impurity K, and Impurity I, which are known to arise from intermolecular substitution or oxidative degradation under harsher conditions. The patent data indicates that single impurities can be consistently controlled below 0.15%, and often below 0.10%, through this mild basic environment. This level of purity is critical for meeting stringent regulatory requirements for pharmaceutical active ingredients, ensuring that the final drug substance is safe for patient administration without requiring extensive downstream purification.
How to Synthesize Relugolix Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and purity. The process begins with the activation of the starting material in an inert solvent, followed by the addition of the amine component under cooled conditions to manage exothermicity. Detailed standardized synthesis steps see the guide below.
- Condense formula N-2 with 3-amino-6-methoxypyridazine using acyl chloride and organic base in solvent A at low temperature.
- Isolate the intermediate formula N-1 without chromatographic separation to maintain efficiency.
- Perform ring closure on formula N-1 using inorganic base in solvent B, followed by recrystallization to achieve over 99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible benefits in terms of cost stability and operational reliability. By eliminating the dependency on volatile and expensive coupling reagents like T3P, manufacturers can secure a more predictable cost structure that is less susceptible to market fluctuations in specialty chemical pricing. The removal of chromatographic purification steps not only reduces solvent consumption and waste disposal costs but also significantly shortens the overall production cycle time, allowing for faster turnaround on large-volume orders. This efficiency translates directly into improved supply chain resilience, ensuring consistent availability of high-quality intermediates for downstream drug formulation.
- Cost Reduction in Manufacturing: The substitution of expensive phosphorus-based condensing agents with commodity acyl chlorides results in substantial raw material cost savings, as these reagents are widely available and significantly cheaper on a molar basis. Additionally, the avoidance of column chromatography reduces the consumption of high-purity solvents and silica gel, further lowering the operational expenditure per kilogram of product. The simplified work-up procedure also minimizes labor hours and equipment usage, contributing to a more lean and cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: The use of stable, commercially available inorganic bases and common organic solvents ensures that raw material sourcing is robust and less prone to disruption compared to specialized coupling agents. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or sensitivity to moisture, leading to higher first-pass yields and more reliable delivery schedules. This stability is crucial for maintaining continuous production lines and meeting the rigorous just-in-time delivery requirements of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as it avoids hazardous reagents and complex purification steps that often bottleneck production at larger volumes. The reduction in toxic waste generation, particularly phosphorus-containing effluents, simplifies environmental permitting and waste treatment, aligning with increasingly strict global sustainability regulations. This eco-friendly profile enhances the long-term viability of the manufacturing site and reduces the regulatory burden associated with hazardous chemical handling and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thienopyrimidine synthesis method. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply chains.
Q: How does this process improve impurity control compared to prior art?
A: By replacing strong alkali sodium alkoxide with mild inorganic bases like potassium carbonate, the formation of degradation impurities such as Impurity I and Impurity L is significantly suppressed, ensuring single impurities remain below 0.15%.
Q: Is chromatographic purification required for this synthesis?
A: No, the novel method is designed for industrial scalability and eliminates the need for column chromatography. High purity is achieved through controlled reaction conditions and simple recrystallization.
Q: What are the cost advantages of using acyl chloride over T3P?
A: Acyl chlorides like thionyl chloride are substantially cheaper and more atom-economical than propylphosphonic anhydride (T3P), reducing raw material costs and eliminating phosphorus-containing wastewater treatment burdens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Relugolix Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate this patented laboratory methodology into a robust, commercial-scale manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and low impurity profiles demonstrated in the patent are maintained at industrial volumes. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for pharmaceutical intermediates and active ingredients.
We invite you to discuss how this optimized synthesis route can enhance your supply chain efficiency and reduce overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis, and let us provide you with specific COA data and route feasibility assessments tailored to your project needs. Our commitment to quality and innovation makes us the ideal partner for your thienopyrimidine sourcing requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
