Scaling High-Purity Olmesartan Medoxomil Intermediates with Novel Solid Phosgene Catalysis
Introduction to Next-Generation ARB Intermediate Manufacturing
The global demand for Angiotensin II Receptor Blockers (ARBs) continues to surge, driven by the increasing prevalence of hypertension and the proven clinical efficacy of drugs like olmesartan medoxomil. At the heart of this therapeutic value chain lies the efficient and safe production of key intermediates, specifically 4,5-dimethyl-1,3-dioxol-2-one (DMDO) and its derivatives. Recent intellectual property developments, notably patent CN113214215A, have introduced a transformative approach to synthesizing these critical building blocks. This technology addresses long-standing industrial bottlenecks by replacing hazardous gaseous reagents with safer solid alternatives and integrating advanced purification techniques. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and scalable production methodologies that align with modern regulatory standards for safety and environmental compliance.
The significance of this patent extends beyond mere chemical novelty; it offers a robust solution to the supply chain vulnerabilities inherent in traditional phosgene-based chemistry. By utilizing solid phosgene, the process mitigates the extreme risks associated with gas handling, storage, and transportation, which have historically limited production capacity and increased operational costs. Furthermore, the integration of molecular distillation technology ensures that the final products meet the stringent purity specifications required for active pharmaceutical ingredient (API) synthesis. This report analyzes the technical merits of this novel route, providing R&D directors and procurement leaders with a clear understanding of how adopting this methodology can enhance product quality while optimizing manufacturing economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of DMDO and its chlorinated or hydroxylated derivatives has been plagued by significant safety and efficiency challenges. The most common traditional route involves the direct reaction of 3-hydroxy-2-butanone with gaseous phosgene. While chemically effective, this method imposes severe logistical burdens due to the extreme toxicity of phosgene gas, requiring specialized containment infrastructure and rigorous safety protocols that drastically increase capital expenditure. Alternative routes utilizing trichloromethyl chloroformate suffer from reagent instability, leading to inconsistent batch quality and potential safety incidents during scale-up. Additionally, chlorination steps often rely on elemental chlorine gas, which introduces corrosive acid gases into the waste stream, necessitating expensive scrubbing systems and creating environmental liabilities that complicate long-term production planning.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113214215A leverages solid phosgene (often referred to as triphosgene equivalents in broader chemistry, though specified here as a safer solid form) to achieve the same cyclization efficiency without the associated gas hazards. This substitution fundamentally alters the risk profile of the manufacturing process, allowing for operation in standard enamel reaction kettles under micro-negative pressure rather than specialized gas-tight reactors. The subsequent chlorination step utilizes chlorotriazine under UV irradiation, a mild condition that avoids the harsh corrosion of chlorine gas while generating s-triazine as a recoverable byproduct. This dual innovation of safer reagents and value-added byproduct recovery creates a process architecture that is inherently more suitable for large-scale commercialization and regulatory approval in strict jurisdictions.
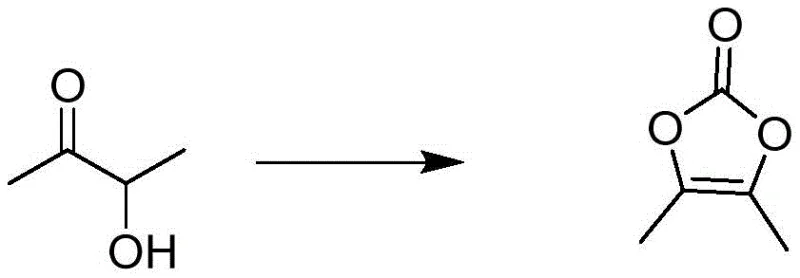
Mechanistic Insights into Solid Phosgene Cyclization and Radical Functionalization
The core chemical transformation in this novel pathway is the cyclization of 3-hydroxy-2-butanone to form the dioxolone ring of DMDO. Mechanistically, this involves the nucleophilic attack of the hydroxyl group on the carbonyl carbon of the solid phosgene, followed by intramolecular cyclization and elimination of hydrogen chloride. The use of a mixed solvent system, such as isopropanol and cyclohexane, plays a critical role in controlling the crystallization kinetics, ensuring that the product precipitates with high purity directly from the reaction mixture. This precise control over solubility parameters minimizes the entrapment of impurities within the crystal lattice, a common issue in rapid precipitation scenarios, thereby reducing the burden on downstream purification units and enhancing the overall mass balance of the synthesis.
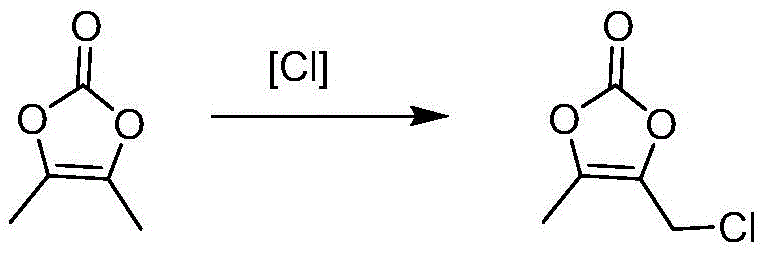
Following the formation of DMDO, the functionalization to DMDO-Cl proceeds via a free-radical mechanism initiated by ultraviolet light. The chlorotriazine reagent undergoes homolytic cleavage under UV irradiation (specifically at 254nm), generating chlorine radicals that selectively abstract a hydrogen atom from the methyl group of the DMDO ring. This selectivity is paramount, as it prevents unwanted chlorination of the ring oxygen or other positions that would lead to difficult-to-separate impurities. The propagation of the radical chain is carefully managed by the concentration of the initiator, azoisobutyronitrile, ensuring a steady reaction rate that prevents thermal runaway. This controlled radical environment is what allows the process to achieve high conversion rates while maintaining the structural integrity of the sensitive cyclic carbonate moiety.
The final transformation to DMDO-OH introduces a hydroxymethyl group through an iron-catalyzed radical process. The patent highlights the use of lithium iron phosphate as a highly effective catalyst, which likely facilitates the generation of hydroxyl radicals from hydrogen peroxide in a controlled manner similar to Fenton chemistry but with greater specificity. The choice of solvent, particularly DMF or 1,4-dioxane, is crucial for stabilizing the transition states and solubilizing the polar intermediates. This step exemplifies the shift towards greener chemistry, replacing toxic selenium dioxide oxidants with benign hydrogen peroxide and earth-abundant iron catalysts. The result is a cleaner reaction profile with fewer heavy metal contaminants, simplifying the purification process and ensuring the final intermediate meets the rigorous heavy metal limits imposed by pharmacopeial standards for API precursors.
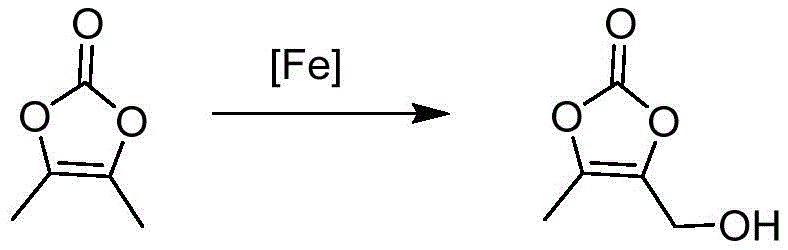
How to Synthesize DMDO Derivatives Efficiently
The synthesis of these high-value intermediates requires precise adherence to the optimized reaction conditions detailed in the patent to ensure maximum yield and purity. The process begins with the careful addition of solid phosgene to the ketone substrate under controlled temperature gradients to manage the exotherm effectively. Following the initial cyclization, the crude DMDO is subjected to a specific recrystallization protocol using mixed solvents to remove unreacted starting materials and oligomeric byproducts. For the subsequent chlorination and hydroxylation steps, the maintenance of specific UV wavelengths and catalyst loadings is critical to driving the reactions to completion without degrading the product. Detailed standardized operating procedures for each unit operation, from reagent dosing to final molecular distillation, are essential for replicating the laboratory success on a commercial manufacturing scale.
- Cyclization of 3-hydroxy-2-butanone with solid phosgene in dichloromethane at controlled temperatures to form DMDO.
- Radical chlorination of DMDO using chlorotriazine under UV irradiation to produce DMDO-Cl with high selectivity.
- Hydroxylation of DMDO using hydrogen peroxide and a lithium iron phosphate catalyst followed by molecular distillation purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid phosgene-based technology offers profound strategic advantages that extend well beyond simple chemical yield improvements. The primary benefit lies in the drastic simplification of raw material logistics; by eliminating the need for pressurized gas cylinders and specialized gas delivery infrastructure, facilities can significantly reduce their fixed capital costs and insurance premiums. The ability to store solid reagents in standard warehouses removes the geographical constraints often imposed by gas supply networks, thereby enhancing supply chain resilience and reducing the risk of production stoppages due to delivery delays. This flexibility allows manufacturers to maintain leaner inventory levels while ensuring continuous operation, a critical factor in meeting the just-in-time delivery demands of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the valorization of byproducts and the reduction of waste treatment expenses. In the chlorination step, the formation of s-triazine as a stoichiometric byproduct transforms a waste stream into a revenue-generating co-product, as s-triazine itself is a valuable intermediate in other medicinal chemistry applications. Furthermore, the replacement of corrosive chlorine gas with solid chlorotriazine reduces the maintenance costs associated with reactor corrosion and the consumption of neutralizing agents for acidic off-gases. The implementation of molecular distillation, while an upfront equipment investment, lowers the long-term variable costs by minimizing product loss due to thermal degradation, ensuring that a higher percentage of theoretical yield is captured as saleable high-purity material.
- Enhanced Supply Chain Reliability: The robustness of the solid phosgene route directly translates to improved reliability in the supply of olmesartan intermediates. Traditional gas-based processes are highly susceptible to disruptions caused by safety inspections, transport regulations, or supplier shortages of hazardous gases. By shifting to solid reagents that are stable and easy to handle, manufacturers can decouple their production schedules from these external volatility factors. Additionally, the mild reaction conditions reduce the likelihood of batch failures due to thermal excursions or equipment malfunctions, leading to more predictable lead times. This stability is invaluable for downstream API manufacturers who require consistent quality and timely delivery to maintain their own production schedules for finished antihypertensive medications.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology positions manufacturers favorably against increasingly stringent global regulations. The elimination of toxic gas emissions and the reduction of heavy metal waste from oxidation steps simplify the permitting process for facility expansions and new plant constructions. The process generates less hazardous waste, lowering the costs associated with disposal and treatment, which is a significant component of the total cost of ownership in fine chemical manufacturing. Moreover, the scalability of the molecular distillation step ensures that quality remains consistent whether producing hundreds of kilograms or multiple metric tons, facilitating seamless technology transfer from pilot plants to full-scale commercial production without the need for extensive re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical disclosures within patent CN113214215A, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies for olmesartan medoxomil precursors.
Q: Why is solid phosgene preferred over gas phosgene for DMDO synthesis?
A: Solid phosgene eliminates the severe safety hazards and logistical complexities associated with storing and transporting toxic gas phosgene, while maintaining high reaction efficiency and yield in the cyclization process.
Q: How does molecular distillation improve the quality of DMDO-Cl?
A: Molecular distillation operates under high vacuum and low residence time, preventing thermal polymerization of the sensitive dioxolone ring and ensuring the removal of high-boiling impurities for superior purity.
Q: What are the commercial benefits of using chlorotriazine as a chlorinating agent?
A: Chlorotriazine offers mild reaction conditions and generates s-triazine as a byproduct, which is a valuable medical intermediate that can be recovered and sold, thereby reducing overall waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DMDO Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven execution capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the solid phosgene technology are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including UV-irradiated reactors and high-vacuum molecular distillation units. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of DMDO and its derivatives meets the exacting standards required for global pharmaceutical registration and commercial success.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these technological advancements for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this patented chemistry can drive efficiency and reliability in your olmesartan medoxomil production.
