Scalable Synthesis of Granisetron Intermediate via Novel Red-Al Reduction
Scalable Synthesis of Granisetron Intermediate via Novel Red-Al Reduction
The pharmaceutical industry constantly seeks robust and scalable pathways for critical antiemetic agents, and the preparation method disclosed in patent CN110804051B represents a significant advancement in the synthesis of granisetron intermediates. This intellectual property details a sophisticated chemical route for producing 3-alpha-homotropylamine, a pivotal building block for the potent 5-HT3 receptor antagonist granisetron, which is widely utilized to prevent nausea and vomiting induced by chemotherapy and radiotherapy. The disclosed methodology addresses historical challenges associated with stereoselectivity and hazardous reaction conditions, offering a streamlined approach that transitions from acetone dicarboxylic acid through a series of optimized transformations. By leveraging specific catalytic systems such as red aluminum combined with Lewis acids, or alternatively employing Raney nickel followed by a novel purification protocol, the inventors have established a process that balances high yield with exceptional purity profiles. This technical breakthrough provides a compelling foundation for manufacturers aiming to secure a reliable supply of high-quality pharmaceutical intermediates while mitigating the risks associated with traditional high-pressure hydrogenation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-alpha-homotropylamine has been plagued by significant technical hurdles that impede efficient commercial manufacturing and inflate production costs. Prior art methods, such as those referenced in U.S. Pat. No. 4,470,585A, often relied on the utilization of expensive platinum catalysts and highly reactive reducing agents like lithium aluminum hydride, which pose severe safety risks due to their pyrophoric nature and sensitivity to moisture. Furthermore, conventional catalytic hydrogenation routes frequently suffered from poor stereoselectivity, resulting in crude product mixtures containing substantial amounts of the unwanted 3-beta isomer alongside the target 3-alpha compound. This lack of selectivity necessitated complex and costly downstream purification steps to meet the stringent purity requirements demanded by regulatory bodies for active pharmaceutical ingredients. Additionally, the requirement for high-pressure hydrogenation equipment in older processes imposed heavy capital expenditure burdens on manufacturing facilities and introduced potential safety hazards related to handling hydrogen gas at elevated pressures, thereby limiting the scalability and economic viability of these legacy synthetic routes.
The Novel Approach
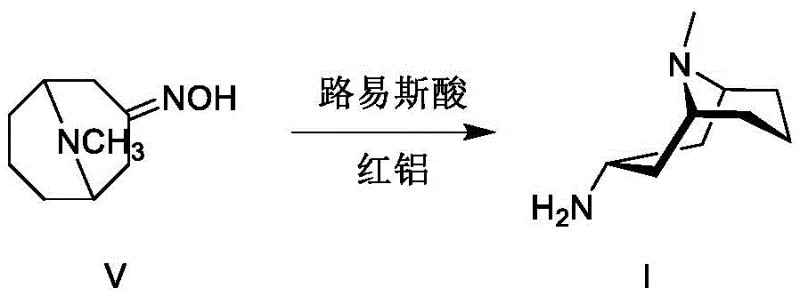
In stark contrast to these cumbersome legacy techniques, the novel approach outlined in the patent introduces a versatile dual-pathway strategy that prioritizes both safety and stereochemical control. The primary innovation involves the use of red aluminum (sodium bis(2-methoxyethoxy)aluminum hydride) in conjunction with a Lewis acid catalyst to effectuate the reduction of 3-homotropinone oxime under remarkably mild conditions, typically ranging from minus 10 degrees Celsius to 20 degrees Celsius. This method inherently favors the formation of the desired 3-alpha isomer, achieving crude purities of approximately 90 percent without the need for extreme pressures or precious metal catalysts. Alternatively, the patent describes a secondary pathway utilizing Raney nickel for hydrogenation, coupled with a groundbreaking purification sequence involving Boc-protection and copper sulfate treatment. This secondary route effectively salvages the stereoselectivity by chemically differentiating the isomers, allowing for the isolation of the target amine with purity levels exceeding 99 percent. Both pathways eliminate the dependency on hazardous high-pressure reactors and expensive platinum group metals, marking a substantial leap forward in process chemistry efficiency.
Mechanistic Insights into Red-Al Catalyzed Stereoselective Reduction
The core mechanistic advantage of this patented process lies in the synergistic interaction between red aluminum and the selected Lewis acid during the reduction of the oxime functionality. When red aluminum is activated by a Lewis acid such as tetraisopropyl titanate or titanium tetrachloride in a tetrahydrofuran solvent system, it generates a highly reactive yet controllable hydride species that attacks the oxime nitrogen-oxygen bond. The presence of the Lewis acid likely coordinates with the oxygen atom of the oxime, increasing the electrophilicity of the nitrogen center and directing the hydride delivery from a specific spatial trajectory that favors the formation of the 3-alpha stereoisomer. This coordination complex stabilizes the transition state in a manner that minimizes the formation of the thermodynamic 3-beta byproduct, which is a common issue in non-catalyzed reductions. The reaction proceeds smoothly at near-ambient temperatures, avoiding the thermal degradation of sensitive intermediates and ensuring that the stereochemical integrity of the tropane ring system is maintained throughout the transformation. This precise control over the reduction mechanism is what allows the process to deliver a crude product with a favorable isomeric ratio of roughly 9:1 in favor of the alpha isomer, significantly reducing the burden on subsequent purification stages.
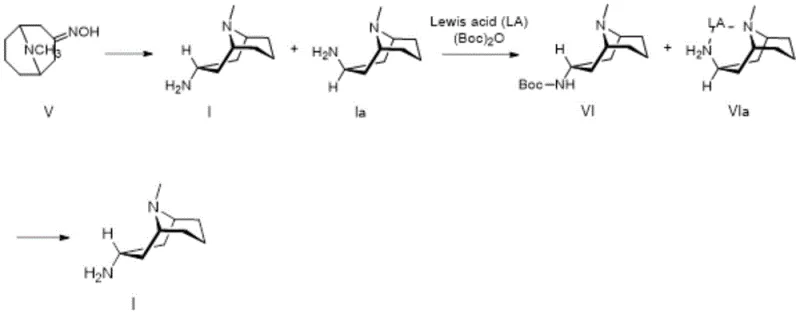
Furthermore, the patent elucidates a sophisticated impurity control mechanism specifically designed for scenarios where the isomeric ratio is less favorable, such as when using the Raney nickel hydrogenation route. In this pathway, the crude mixture containing both 3-alpha and 3-beta isomers is subjected to a selective protection strategy using di-tert-butyl dicarbonate (Boc2O) in the presence of copper sulfate pentahydrate and sodium bicarbonate. The copper ions appear to play a critical role in facilitating the differential reactivity or solubility of the isomers during the Boc-protection step, effectively masking the amine functionality of the desired 3-alpha isomer as a carbamate while leaving the impurities more susceptible to removal. Following the protection, the intermediate Boc-protected amine is isolated and subsequently deprotected using methanolic hydrogen chloride to regenerate the free amine hydrochloride salt. This chemical differentiation strategy acts as a powerful filter, scrubbing away the persistent 3-beta impurity that co-elutes during standard distillation or crystallization, ultimately yielding a final product with purity specifications greater than 99 percent, which is essential for downstream coupling reactions to form granisetron.
How to Synthesize 3-alpha-homotropylamine Efficiently
The synthesis of this critical granisetron intermediate begins with the preparation of the ketoxime precursor, which is subsequently reduced using the optimized catalytic systems described in the patent documentation. Operators must strictly adhere to the temperature controls and reagent ratios specified, particularly when handling red aluminum and Lewis acids, to ensure maximum stereoselectivity and safety during the exothermic reduction phase. For facilities equipped for hydrogenation, the Raney nickel route offers a viable alternative provided that the rigorous purification protocol involving copper sulfate and Boc-anhydride is implemented to guarantee final purity. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures required to replicate these high-yield results, are outlined in the comprehensive guide below for technical reference.
- Perform Mannich reaction on acetone dicarboxylic acid to obtain pseudopelletierine, followed by oximation to form 3-homotropinone oxime.
- Reduce the oxime using Red-Al and a Lewis acid at controlled low temperatures to favor the 3-alpha isomer, or use Raney Nickel hydrogenation.
- Purify the crude amine mixture via Boc-protection using di-tert-butyl dicarbonate and copper sulfate separation to achieve over 99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this patented synthesis route offers profound benefits that extend well beyond simple chemical yield improvements, fundamentally altering the cost structure and risk profile of the supply chain. By eliminating the requirement for expensive platinum catalysts and specialized high-pressure hydrogenation vessels, manufacturers can realize significant capital expenditure savings and reduce the operational complexity associated with maintaining such hazardous equipment. The shift towards milder reaction conditions also translates into lower energy consumption for heating and cooling, as the process operates effectively near room temperature rather than requiring extreme thermal inputs. Furthermore, the enhanced safety profile of using red aluminum or Raney nickel under ambient pressure reduces insurance premiums and regulatory compliance costs, creating a more resilient and cost-effective manufacturing environment. These factors collectively contribute to a more stable pricing model for the intermediate, shielding buyers from the volatility often associated with precious metal markets and complex high-pressure processing fees.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts like platinum and the avoidance of high-risk reagents such as lithium aluminum hydride drastically simplifies the raw material procurement strategy and lowers direct material costs. The process utilizes commercially abundant reagents like red aluminum and Raney nickel, which are not only more affordable but also easier to source in bulk quantities without supply bottlenecks. Additionally, the high stereoselectivity achieved in the primary reduction step minimizes the loss of valuable starting materials to unwanted isomers, thereby improving the overall mass balance and atom economy of the synthesis. This efficiency gain means that less waste is generated per kilogram of product, reducing the costs associated with waste disposal and environmental remediation while maximizing the output from each batch run.
- Enhanced Supply Chain Reliability: The reliance on standard laboratory and plant equipment rather than specialized high-pressure reactors enhances the flexibility of the supply chain, allowing for production to be scaled up or shifted between different manufacturing sites with greater ease. The robustness of the chemical process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by failed runs or out-of-specification results that require reprocessing. Moreover, the use of stable and storable reagents mitigates the risk of supply disruptions that can occur with sensitive or highly regulated chemicals, ensuring a continuous flow of intermediates to downstream API manufacturers. This reliability is crucial for maintaining uninterrupted production schedules for finished granisetron formulations, safeguarding against market shortages.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of heavy metal contaminants in the final product streamline the environmental compliance process, making it easier to obtain necessary permits for large-scale production. The purification method effectively removes impurities without generating excessive hazardous waste streams, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing operation. The process is inherently scalable, as demonstrated by the patent examples which utilize kilogram-scale quantities, proving that the chemistry holds up when transitioning from pilot plants to commercial tonnage production. This scalability ensures that suppliers can meet growing global demand for granisetron without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the specific embodiments and comparative data presented in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the documented performance metrics and procedural details to ensure accurate expectation setting for potential partners and licensees.
Q: What are the advantages of the Red-Al reduction method over traditional hydrogenation?
A: The Red-Al reduction method described in the patent offers milder reaction conditions and avoids the use of expensive platinum catalysts or high-pressure hydrogenation equipment, significantly enhancing operational safety and reducing capital expenditure requirements.
Q: How is the stereochemical purity of the 3-alpha isomer ensured?
A: The process utilizes a specific purification strategy involving Boc-protection and copper sulfate treatment which effectively separates the desired 3-alpha isomer from the 3-beta impurity, ensuring final product purity exceeds 99%.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its mild conditions, high yield, and the use of commercially available reagents like Red-Al and Raney Nickel which are easier to handle at scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Granisetron Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the successful formulation of life-saving antiemetic medications, and we possess the technical expertise to bring this advanced synthesis route to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 3-alpha-homotropylamine meets or exceeds the 99 percent purity benchmark required for pharmaceutical applications. Our commitment to quality assurance means that we can consistently deliver material that minimizes downstream processing issues for our clients, acting as a true extension of your own R&D and manufacturing capabilities.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative technology for their granisetron supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that details how switching to this specific manufacturing route can optimize your overall budget. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
