Advanced Synthesis of 9,10-Thiophene/Furyl Anthracene AIE Compounds for OLED Applications
The landscape of organic optoelectronic materials is undergoing a transformative shift with the emergence of Aggregation-Induced Emission (AIE) luminogens, which overcome the notorious Aggregation-Caused Quenching (ACQ) effect that has long plagued traditional fluorophores. Patent CN110295040B introduces a groundbreaking class of 9,10-thiophene/furyl anthracene compounds that exhibit sensitive AIE properties, making them ideal candidates for next-generation organic light-emitting diodes (OLEDs) and advanced bio-imaging applications. Unlike conventional dyes that lose efficiency in solid states, these novel anthracene derivatives demonstrate sharply enhanced fluorescence upon aggregation, driven by the restriction of intramolecular rotation. This patent not only discloses the molecular architecture but also provides a robust, scalable preparation method that leverages transition metal catalysis to achieve high purity and yield. For industry leaders seeking a reliable OLED material supplier, this technology represents a critical advancement in developing stable, high-efficiency blue light emitters and cellular markers.
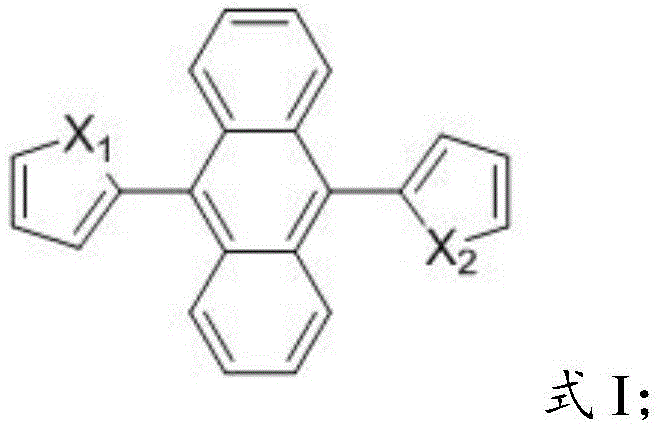
The structural versatility of these compounds allows for fine-tuning of electronic properties by varying the heteroatoms (sulfur or oxygen) in the peripheral rings, enabling precise control over emission wavelengths and quantum yields. The simplicity of the molecular design, combined with the profound photophysical benefits, positions these materials at the forefront of electronic chemical manufacturing innovation. By addressing the fundamental limitations of solid-state luminescence, this invention opens new avenues for high-performance display technologies and sensitive diagnostic tools, ensuring that supply chains can meet the growing demand for superior optical materials without compromising on stability or cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance anthracene-based luminogens has been fraught with challenges, primarily stemming from complex multi-step synthetic routes that require harsh reaction conditions and expensive reagents. Traditional methods often involve tedious protection and deprotection strategies, leading to low overall yields and significant generation of hazardous waste, which complicates commercial scale-up of complex organic intermediates. Furthermore, many existing AIE materials suffer from batch-to-batch variability due to the sensitivity of the reaction parameters, resulting in inconsistent purity profiles that are unacceptable for high-end electronic applications. The reliance on difficult-to-source precursors and the need for rigorous purification processes further inflate production costs, creating bottlenecks for procurement teams aiming to optimize their supply chain expenditures. Additionally, the structural rigidity required to prevent ACQ in older designs often limits the chemical tunability, restricting the ability to modify the molecules for specific wavelength requirements or solubility characteristics needed for diverse device architectures.
The Novel Approach
In stark contrast, the methodology outlined in patent CN110295040B employs a streamlined Suzuki-Miyaura cross-coupling strategy that drastically simplifies the synthetic pathway while maintaining exceptional control over product quality. This novel approach utilizes readily available starting materials such as 9,10-dibromoanthracene and heterocyclic boronic acids, which are accessible from global chemical suppliers, thereby enhancing supply chain resilience. The reaction proceeds under mild heating reflux conditions in common organic solvents, eliminating the need for extreme temperatures or pressures that typically demand specialized reactor infrastructure. By adjusting the stoichiometric ratios of the reactants, manufacturers can selectively produce either symmetric or asymmetric derivatives, offering a flexible platform for generating a library of functional materials from a single core process. This adaptability significantly reduces the time-to-market for new material variants, allowing R&D departments to iterate designs rapidly without being hindered by synthetic complexity. The result is a cost-effective, environmentally friendlier process that aligns perfectly with modern green chemistry principles while delivering materials with superior aggregation-induced emission characteristics.
Mechanistic Insights into Suzuki-Miyaura Coupling for AIE Compounds
The core of this innovative synthesis lies in the palladium-catalyzed Suzuki-Miyaura coupling reaction, a powerful tool for constructing carbon-carbon bonds between aryl halides and organoboron species. In the context of producing 9,10-dithienylanthracene (DTA) or 9,10-difuranylanthracene (DFA), the mechanism involves the oxidative addition of the palladium catalyst to the carbon-bromine bonds of the 9,10-dibromoanthracene core, forming a reactive organopalladium intermediate. This is followed by transmetallation with the thiophene or furan boronic acid, facilitated by the presence of a base such as sodium carbonate or potassium phosphate, which activates the boron species for nucleophilic attack. The final reductive elimination step releases the coupled product and regenerates the active palladium catalyst, completing the catalytic cycle. The choice of ligand on the palladium center, such as triphenylphosphine or dppf, plays a crucial role in stabilizing the catalyst and preventing the formation of palladium black, which ensures consistent reaction rates and high turnover numbers throughout the process.
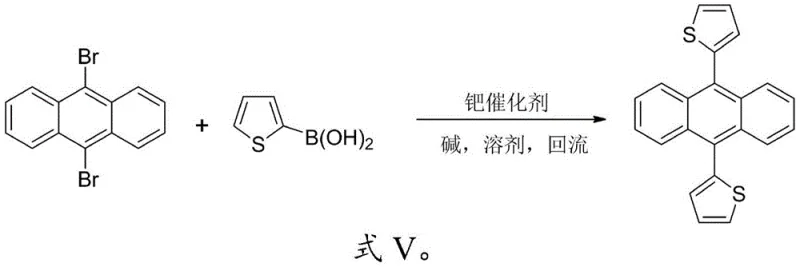
For the asymmetric variant, 9-(thiophene-2-yl)-10-(furan-2-yl)anthracene (TFA), the mechanism requires precise kinetic control to achieve mono-substitution before introducing the second heterocycle. By limiting the molar ratio of the first boronic acid to approximately 1:1 relative to the dibromoanthracene, the reaction favors the formation of the mono-substituted intermediate, 9-(thiophene-2-yl)-10-bromoanthracene. This intermediate is then isolated and subjected to a second coupling cycle with the alternative boronic acid. This stepwise approach prevents the statistical mixture of symmetric and asymmetric products that would occur in a one-pot reaction, ensuring high regioselectivity and purity. The robustness of the Suzuki coupling under these conditions allows for the tolerance of various functional groups, making it a versatile method for future derivatization. Understanding these mechanistic nuances is vital for reducing lead time for high-purity fluorescence markers, as it enables process chemists to optimize reaction times and minimize impurity formation effectively.

How to Synthesize 9,10-Thiophene/Furyl Anthracene Efficiently
The preparation of these high-value AIE compounds is designed to be operationally simple, requiring standard laboratory equipment and commonly used reagents, which facilitates easy transfer from pilot scale to full industrial production. The process begins with the careful selection of high-quality 9,10-dibromoanthracene and the corresponding boronic acid derivatives, ensuring that moisture content is minimized to prevent catalyst deactivation. The reaction is typically conducted under an inert nitrogen atmosphere to protect the sensitive palladium catalyst from oxidation, with the mixture heated to reflux for a duration of 11 to 13 hours to ensure complete conversion. Following the reaction, a straightforward workup procedure involving solvent removal, liquid-liquid extraction, and drying yields a crude product that is easily purified via column chromatography. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during manufacturing operations.
- Mix 9,10-dibromoanthracene with 2-boronic acid-thiophene or furan, a palladium catalyst, base, and solvent under protective atmosphere.
- Heat the reaction mixture to reflux for 11-13 hours to facilitate the Suzuki coupling reaction.
- Perform post-treatment including extraction, drying, and column chromatography to isolate the pure AIE compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial strategic benefits for organizations looking to optimize their material sourcing and production costs. The reliance on commodity chemicals like 9,10-dibromoanthracene and simple heterocyclic boronic acids means that raw material procurement is not subject to the volatility associated with exotic or proprietary reagents. This stability in the supply base translates directly into more predictable pricing models and reduced risk of production stoppages due to material shortages. Furthermore, the high atom economy of the Suzuki coupling reaction minimizes waste generation, lowering the costs associated with waste disposal and environmental compliance. For procurement managers, this means achieving cost reduction in electronic chemical manufacturing not just through cheaper inputs, but through a more efficient overall process flow that maximizes output per unit of input.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and harsh reaction conditions significantly lowers the operational expenditure required for production. By avoiding the need for cryogenic temperatures or high-pressure reactors, facilities can utilize existing infrastructure, thereby deferring capital investment. The use of recyclable solvents and heterogeneous catalysts where applicable further drives down the variable costs per kilogram of product. Additionally, the high yields reported in the patent examples indicate that less raw material is wasted, directly improving the gross margin on the final AIE compound. This economic efficiency makes the technology highly attractive for large-scale manufacturing where even marginal savings per unit can result in substantial annual financial gains.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible production scheduling, as the symmetric and asymmetric variants share common intermediates and processing steps. This flexibility enables manufacturers to respond quickly to fluctuating market demands without needing to retool entire production lines. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring a consistent and reliable flow of goods to downstream customers. For supply chain heads, this reliability is paramount in maintaining just-in-time inventory levels and meeting the strict delivery timelines demanded by the electronics and pharmaceutical sectors. The ability to source key precursors from multiple global vendors further mitigates the risk of supply chain disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, utilizing standard reflux and filtration techniques that are easily adapted to larger reactor volumes. The mild conditions reduce energy consumption, contributing to a lower carbon footprint for the manufacturing process. Moreover, the absence of toxic heavy metals in the final product, thanks to efficient purification, simplifies regulatory compliance for end-use applications in consumer electronics and biomedical devices. This alignment with environmental, social, and governance (ESG) goals enhances the brand value of the final product and facilitates easier market entry in regions with stringent chemical regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel AIE compounds, based on the detailed disclosures within the patent literature. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their product portfolios. The answers reflect the practical realities of scaling this chemistry and the specific performance benefits observed in experimental data.
Q: What are the primary advantages of this AIE compound synthesis method?
A: The method utilizes simple Suzuki coupling reactions with commercially available raw materials, offering mild conditions, low cost, and suitability for industrial scale-up compared to complex multi-step traditional routes.
Q: Can these compounds be used for biological applications?
A: Yes, the synthesized 9,10-thiophene/furyl anthracene compounds exhibit excellent cell permeability and fluorescence, making them effective markers for HeLa cell imaging and biochemical detection.
Q: How does the stoichiometry affect the product symmetry?
A: Using a 1:2 molar ratio of dibromoanthracene to boronic acid yields symmetric compounds (DTA or DFA), while a controlled 1:1 ratio followed by a second coupling step produces asymmetric derivatives (TFA).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Thiophene/Furyl Anthracene Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this patented technology to deliver high-performance AIE compounds that meet the rigorous standards of the global optoelectronics and life sciences industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready product is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 9,10-thiophene/furyl anthracene meets your exact requirements. Our commitment to quality and consistency makes us the preferred choice for companies seeking a dependable source of advanced functional materials.
We invite you to collaborate with us to explore the full potential of these aggregation-induced emission materials in your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how our optimized processes can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with our superior manufacturing capabilities and deep chemical expertise.
