Advanced Synthesis of 9,10-Thiophene/Furyl Anthracene AIE Compounds for Commercial OLED Applications
Advanced Synthesis of 9,10-Thiophene/Furyl Anthracene AIE Compounds for Commercial OLED Applications
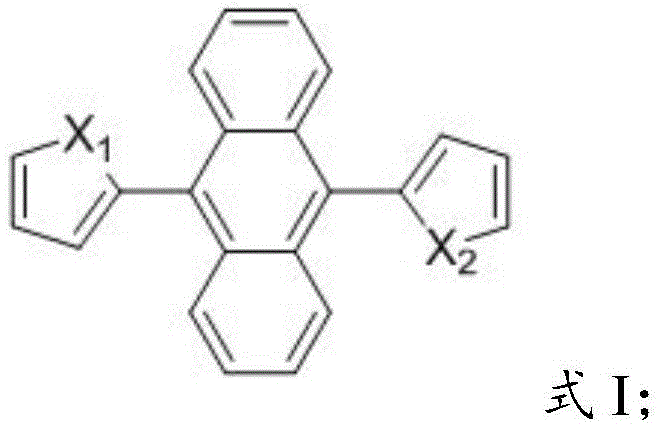
The rapid evolution of organic photoelectric functional materials has necessitated the development of novel compounds that overcome the inherent limitations of traditional fluorophores, specifically the phenomenon of Aggregation-Caused Quenching (ACQ). Patent CN110295040B introduces a groundbreaking class of 9,10-thiophene/furyl anthracene aggregation-induced emission (AIE) compounds that exhibit sensitive emission properties and a remarkably simple structural architecture. These materials represent a significant leap forward for the electronic chemical manufacturing sector, offering a robust solution for organic electroluminescence and solid-state laser applications. The core innovation lies in the strategic substitution of the anthracene core with thiophene or furan rings, which restricts intramolecular rotation upon aggregation, thereby inducing sharp enhancements in fluorescence emission intensity. This patent not only details the molecular design but also provides a scalable preparation method that is highly suitable for industrial production, addressing the critical need for reliable OLED material suppliers who can deliver high-performance luminescent compounds.
Historically, the commercialization of organic light-emitting diodes (OLEDs) and related photoluminescent devices has been hindered by the ACQ effect, where molecules that fluoresce brightly in dilute solutions lose their efficiency in the solid state due to tight stacking and excimer formation. Conventional methods often rely on complex molecular engineering to prevent this stacking, frequently resulting in synthetic routes that are cumbersome, low-yielding, and economically unviable for large-scale commercial scale-up of complex polymer additives or small molecule emitters. Traditional anthracene derivatives, while possessing good fluorescence quantum yields in solution, often fail to maintain this performance in the aggregated state required for device fabrication. Furthermore, existing synthesis pathways for modified anthracenes frequently involve harsh reaction conditions, multiple protection-deprotection steps, and the use of expensive transition metal catalysts that are difficult to remove, leading to impurity profiles that are unacceptable for high-purity electronic chemical applications. These limitations create a bottleneck in the supply chain, increasing lead times and manufacturing costs significantly.
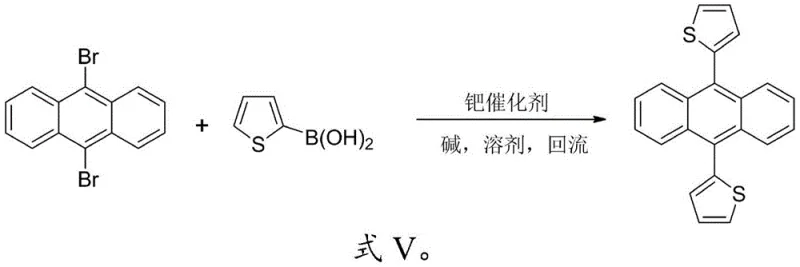
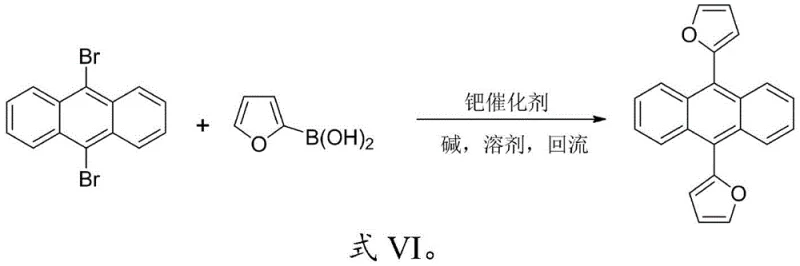
In stark contrast, the novel approach detailed in the patent utilizes a direct and efficient Suzuki coupling reaction to construct the target 9,10-thiophene/furyl anthracene framework. This method leverages the high reactivity and selectivity of palladium-catalyzed cross-coupling to attach heterocyclic rings directly to the 9,10-positions of the anthracene core. For the synthesis of symmetrical derivatives like 9,10-dithienylanthracene (DTA), the process involves mixing 9,10-dibromoanthracene with 2-boronic acid-thiophene in a molar ratio of 1:2 to 1:2.5. Similarly, for 9,10-difuranylanthracene (DFA), 2-boronic acid-furan is employed under identical conditions. This streamlined strategy eliminates the need for intermediate isolation in the symmetrical cases and allows for a one-pot transformation that drastically simplifies the workflow. The reaction conditions are mild, typically requiring heating under reflux for 11 to 13 hours in common solvents such as THF or DMF, which are easily recoverable and recycle-friendly. This simplicity translates directly into operational efficiency, making it an ideal candidate for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Palladium-Catalyzed Suzuki Coupling
The success of this synthesis relies on the precise orchestration of the catalytic cycle involving the palladium catalyst, which facilitates the cross-coupling between the aryl halide (9,10-dibromoanthracene) and the organoboron species (heterocyclic boronic acids). The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-bromine bond of the anthracene derivative, forming an organopalladium(II) intermediate. This step is critical and is influenced by the electronic nature of the anthracene core; the presence of electron-withdrawing or donating groups on the heterocycles can modulate the rate of this insertion. Following oxidative addition, the transmetallation step occurs, where the boron-bound heterocycle transfers to the palladium center, a process accelerated by the presence of the alkaline compound such as sodium carbonate or potassium phosphate. The final reductive elimination step releases the coupled product and regenerates the active palladium(0) catalyst, allowing the cycle to continue. The choice of ligands on the palladium, such as triphenylphosphine in Pd(PPh3)4, plays a pivotal role in stabilizing the active species and preventing the formation of palladium black, which would deactivate the catalyst and introduce metallic impurities.
Impurity control is paramount in the production of high-purity OLED materials, and this synthetic route offers inherent advantages in this regard. The use of stoichiometric control, particularly in the asymmetric synthesis of 9-(thiophene-2-yl)-10-(furan-2-yl)anthracene (TFA), allows for the selective mono-substitution of the dibromoanthracene precursor. By adjusting the molar ratio of 9,10-dibromoanthracene to 2-boranyl-thiophene to 1:1 to 1:1.2, the reaction can be halted at the mono-substituted stage, yielding 9-(thiophene-2-yl)-10-bromoanthracene with high selectivity. This intermediate is then subjected to a second Suzuki coupling with 2-boranyl-furan. This stepwise approach minimizes the formation of homocoupled byproducts (such as DTA or DFA) that would be difficult to separate if a one-pot mixed reaction were attempted. Furthermore, the post-treatment process involving extraction with dichloromethane and purification via column chromatography using petroleum ether ensures the removal of residual palladium, boronic acid byproducts, and unreacted starting materials, resulting in a final product with the stringent purity specifications required for optoelectronic applications.
How to Synthesize 9,10-Thiophene/Furyl Anthracene Efficiently
The synthesis of these advanced AIE compounds is designed to be accessible yet rigorous, ensuring reproducibility across different scales of production. The process begins with the careful selection of high-quality starting materials, specifically 9,10-dibromoanthracene and the respective boronic acid derivatives, which are readily available from global chemical suppliers. The reaction is conducted under a protective nitrogen atmosphere to prevent the oxidation of the palladium catalyst and the sensitive boronic acid reagents. Solvents like THF are dried and degassed prior to use to maintain anhydrous conditions, which are crucial for the efficiency of the transmetallation step. The reaction mixture is heated to reflux, maintaining a temperature consistent with the boiling point of the solvent system, typically for a duration of 11 to 13 hours to ensure complete conversion of the starting bromide. Upon completion, the reaction mixture is cooled to room temperature, and the solvent is removed via rotary evaporation. The residue is then subjected to liquid-liquid extraction, where the organic phase containing the product is separated from the aqueous layer containing inorganic salts. Final purification is achieved through column chromatography, utilizing specific eluent systems such as petroleum ether or mixtures of petroleum ether and dichloromethane to isolate the target compound as a solid with distinct coloration, ranging from green to pale yellow depending on the specific heterocyclic substitution.
- Mix 9,10-dibromoanthracene with 2-boronic acid-thiophene or 2-boronic acid-furan, a palladium catalyst, base, and solvent under protective atmosphere.
- Heat the reaction mixture to reflux for 11-13 hours to facilitate the Suzuki coupling reaction.
- Perform post-treatment including cooling, extraction, drying, and column chromatography to isolate the pure AIE compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to substantial cost savings. By utilizing a single-step Suzuki coupling for symmetrical derivatives and a straightforward two-step sequence for asymmetric ones, the need for complex multi-stage synthesis is eliminated. This reduction in synthetic complexity means fewer unit operations, lower energy consumption, and reduced labor costs, all of which contribute to a more competitive pricing structure for the final electronic chemical products. Furthermore, the reliance on commodity chemicals like 9,10-dibromoanthracene and simple heterocyclic boronic acids ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or proprietary intermediates that often plague the fine chemical industry.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of cost-effective catalysts and bases that are widely available in the bulk chemical market. The ability to use standard solvents like THF and DMF, which can be recovered and recycled through distillation, further lowers the operational expenditure. Additionally, the high selectivity of the Suzuki coupling minimizes the generation of difficult-to-separate byproducts, reducing the load on purification units and increasing the overall yield of the desired AIE compound. This efficiency translates into a lower cost of goods sold (COGS), allowing manufacturers to offer high-performance materials at a price point that supports widespread adoption in consumer electronics and display technologies.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis are not subject to the same geopolitical or logistical constraints as rare earth elements or highly specialized precursors. 9,10-dibromoanthracene and thiophene/furan boronic acids are produced by multiple vendors globally, ensuring a diversified supply base that protects against shortages. The robustness of the reaction conditions, which tolerate mild variations in temperature and pressure without significant loss of yield, adds another layer of reliability to the production schedule. This stability allows for consistent batch-to-batch quality, which is critical for long-term contracts with major display manufacturers who require uninterrupted supply continuity for their production lines.
- Scalability and Environmental Compliance: From a scalability perspective, the process is inherently adaptable to larger reactor volumes without the need for specialized high-pressure equipment or cryogenic conditions. The reaction proceeds efficiently under atmospheric pressure and moderate heating, making it suitable for standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. Moreover, the environmental footprint of the process is manageable; the aqueous waste streams primarily contain inorganic salts which can be treated using standard wastewater protocols, and the organic solvents can be reclaimed. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the burden of hazardous waste disposal, supporting the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and application potential of these novel AIE compounds. The answers are derived directly from the experimental data and characterization results presented in the patent documentation, providing a clear understanding of the material's capabilities. These insights are intended to assist technical teams in evaluating the suitability of these compounds for their specific research and development projects, particularly in the fields of bio-imaging and optoelectronics.
Q: What is the primary advantage of these anthracene derivatives over traditional organic light-emitting molecules?
A: Unlike traditional molecules that suffer from Aggregation-Caused Quenching (ACQ) where fluorescence decreases in solid state, these compounds exhibit Aggregation-Induced Emission (AIE), meaning their fluorescence intensity sharply enhances when aggregated or in solid state, making them ideal for solid-state luminescent materials.
Q: How does the synthesis method ensure cost-effectiveness for industrial production?
A: The preparation method utilizes a straightforward Suzuki coupling reaction with commercially available raw materials like 9,10-dibromoanthracene and heterocyclic boronic acids. The process involves simple steps, mild conditions, and standard workup procedures, eliminating the need for complex multi-step syntheses or expensive specialized reagents.
Q: Can these compounds be used for biological applications?
A: Yes, the compounds have demonstrated the ability to effectively enter HeLa cells and function as cell fluorescent markers. Their significant AIE characteristics and biocompatibility make them suitable for cell fluorescence imaging and biochemical detection applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-Thiophene/Furyl Anthracene Supplier
As the demand for advanced luminescent materials continues to surge, partnering with a knowledgeable and capable CDMO is essential for bringing these innovations to market. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors and rigorous QC labs capable of meeting the stringent purity specifications demanded by the OLED and bio-imaging sectors. We understand that the transition from laboratory benchtop to pilot plant requires meticulous attention to detail, particularly in managing exotherms and optimizing workup procedures for large batches. Our team of expert chemists is dedicated to refining the Suzuki coupling process described in CN110295040B to ensure maximum efficiency and minimal impurity formation, guaranteeing a product that performs consistently in your final applications.
We invite you to collaborate with us to explore the full potential of these aggregation-induced emission compounds. Whether you are developing next-generation displays or advanced cellular probes, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how our manufacturing capabilities can support your project timelines. By choosing NINGBO INNO PHARMCHEM, you are securing a partner committed to quality, reliability, and the continuous advancement of fine chemical manufacturing.
