Scalable Total Synthesis Of Anthrabenzoxocinones Antibiotics And Derivatives For Commercial Production
The pharmaceutical industry continuously seeks robust supply chains for complex bioactive molecules, particularly those with potent antibiotic properties. Patent CN113045579B represents a significant breakthrough in the field of medicinal chemistry by disclosing the first total synthesis of Anthrabenzoxocinones, a class of aromatic polyketide natural products including ABX-A, ABX-C, ABX-E, and BABX. Historically, obtaining these compounds relied solely on fermentation and isolation from Streptomyces strains, a process plagued by low yields and inconsistent supply. This patent introduces a rationally designed chemical synthesis pathway that not only secures the supply of these critical antibiotics but also opens avenues for creating novel derivatives through late-stage functionalization. For R&D directors and procurement specialists, this technology translates to a reliable pharmaceutical intermediates supplier capability, ensuring that drug development pipelines are not bottlenecked by raw material scarcity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of Anthrabenzoxocinones such as ABX-A was dependent on biological isolation from specific microbial strains like Streptomyces violaceum A24566. This conventional approach suffers from inherent limitations typical of natural product extraction, including extremely low titers in fermentation broths and complex downstream purification processes required to separate the target molecule from structurally similar metabolites. Furthermore, the biological source is subject to variability due to strain degeneration or environmental factors, leading to unpredictable batch-to-batch consistency. For a procurement manager, this volatility creates significant supply chain risks, making it difficult to secure the quantities necessary for preclinical and clinical trials. The inability to chemically modify the natural skeleton easily also restricts structure-activity relationship (SAR) studies, limiting the potential for optimizing the antibiotic efficacy against resistant strains like MRSA.
The Novel Approach
The synthetic methodology outlined in the patent offers a transformative solution by constructing the complex polycyclic architecture from simple, commercially available starting materials. The route employs a convergent strategy where two key fragments are assembled via a series of stereoselective reactions. By utilizing an intermolecular Diels-Alder reaction followed by an intramolecular hetero-Diels-Alder cyclization, the synthesis efficiently builds the core ring system with high precision. This chemical approach eliminates the dependency on biological sources, thereby ensuring cost reduction in pharmaceutical intermediates manufacturing through predictable reaction yields and scalable processes. Moreover, the synthetic intermediates are amenable to modification, allowing chemists to introduce diverse functional groups at specific positions, which is crucial for developing next-generation antibiotics with improved pharmacokinetic profiles.
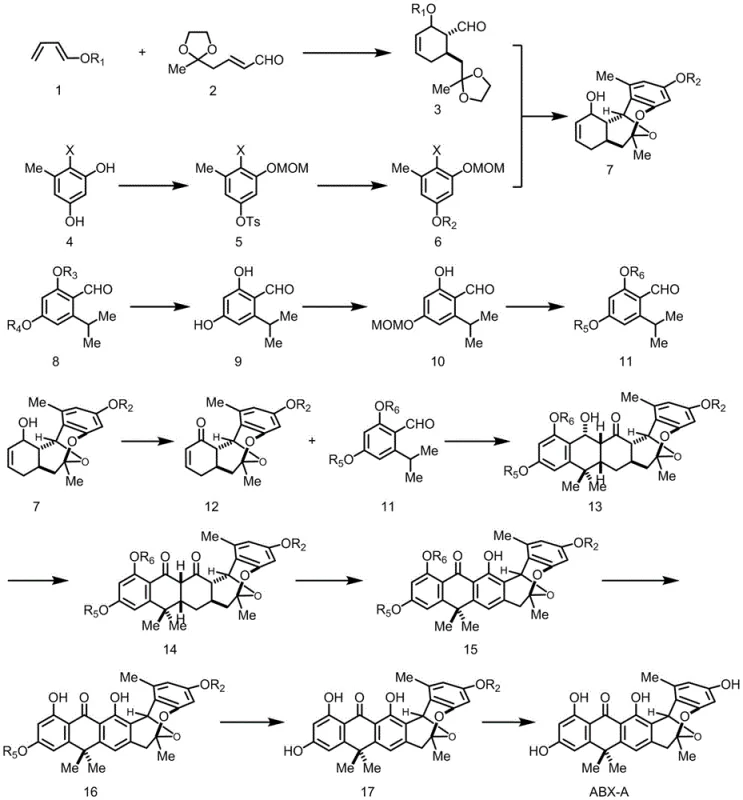
Mechanistic Insights into Titanium-Mediated Photocatalytic Cycloaddition
A cornerstone of this synthetic achievement is the application of a titanium-mediated photocatalytic Diels-Alder reaction to construct the challenging eight-membered ring system characteristic of Anthrabenzoxocinones. In this critical step, the enone intermediate (compound 12) reacts with the aryl aldehyde fragment (compound 11) under irradiation with 300nm ultraviolet light in the presence of tetraisopropyl titanate. The Lewis acidic nature of the titanium species likely coordinates with the carbonyl oxygens, lowering the LUMO energy of the dienophile and facilitating the [4+2] cycloaddition under mild conditions. This mechanistic pathway is superior to thermal methods which might lead to decomposition or lack of stereocontrol. Following the cycloaddition, the resulting adduct undergoes oxidative aromatization using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) at 80°C in toluene. This oxidation step is vital for establishing the fully conjugated aromatic system that defines the biological activity of the final natural product.
Impurity control is meticulously managed throughout the synthesis through the strategic use of orthogonal protecting groups. For instance, the phenolic hydroxyl groups are protected as benzyl ethers or silyl ethers (such as TIPS) at different stages to prevent unwanted side reactions during the harsh oxidation or lithiation steps. The selective deprotection sequences, such as using magnesium iodide for demethylation or tetrabutylammonium fluoride for desilylation, ensure that the final product possesses the exact substitution pattern required for bioactivity. This level of control minimizes the formation of regioisomers and structural analogs that would complicate purification, thereby enhancing the overall purity profile of the high-purity pharmaceutical intermediates produced via this route.
How to Synthesize Anthrabenzoxocinones Efficiently
The synthesis of these complex natural products requires precise control over reaction parameters to ensure high fidelity and yield. The process begins with the thermal Diels-Alder reaction at 100°C to form the initial cyclic adduct, followed by protection group manipulations to prepare the coupling partners. The detailed standardized synthesis steps see the guide below, which outlines the specific stoichiometry and conditions required for each transformation, from the initial fragment coupling to the final deprotection and chlorination steps.
- Perform intermolecular Diels-Alder reaction between compound 1 and 2 at 100°C to form intermediate 3.
- Execute nucleophilic addition and intramolecular hetero-Diels-Alder reaction to construct the polycyclic core structure 7.
- Utilize tetraisopropyl titanate mediated photocatalytic Diels-Alder reaction followed by oxidative aromatization to yield the final anthrabenzoxocinone scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition from extraction to total synthesis offers profound strategic advantages. The primary benefit lies in the decoupling of production from biological constraints, which significantly enhances supply chain reliability. Synthetic routes can be scaled from gram to kilogram levels much more rapidly than optimizing fermentation processes, reducing lead time for high-purity pharmaceutical intermediates. Additionally, the use of commodity chemicals as starting materials mitigates the risk of raw material shortages that often plague natural product sourcing.
- Cost Reduction in Manufacturing: The synthetic route eliminates the expensive and time-consuming processes associated with fermentation, extraction, and extensive purification of natural broths. By replacing these biological steps with efficient chemical transformations, the overall cost of goods sold is drastically reduced. Furthermore, the ability to recycle solvents and reagents in a controlled chemical environment contributes to substantial cost savings compared to the variable costs of biological media and downstream processing.
- Enhanced Supply Chain Reliability: Chemical synthesis provides a consistent and predictable output, unaffected by the biological variabilities of microbial strains. This reliability ensures that production schedules can be met with high certainty, preventing delays in drug development timelines. The modular nature of the synthesis also means that if one supply line for a specific reagent is disrupted, alternative synthetic pathways or protecting group strategies can often be employed without halting the entire production campaign.
- Scalability and Environmental Compliance: The reactions described, such as the Diels-Alder cycloadditions and oxidations, are well-suited for scale-up in standard chemical reactors. The process avoids the generation of large volumes of biological waste, simplifying wastewater treatment and environmental compliance. The use of defined chemical reagents allows for better management of hazardous materials and waste streams, aligning with modern green chemistry principles and regulatory requirements for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the method for industrial application.
Q: What is the key advantage of this synthetic route over natural extraction?
A: This total synthesis route overcomes the low yield and supply instability associated with isolating Anthrabenzoxocinones from Streptomyces strains, enabling reliable large-scale production.
Q: Can this method produce derivatives like ABX-C and BABX?
A: Yes, the route is highly modular. Intermediate 21 (ABX-E) can be selectively chlorinated using n-butyllithium and tert-butyl hypochlorite to generate ABX-C and BABX efficiently.
Q: What are the critical reaction conditions for the core scaffold formation?
A: The formation of the core scaffold relies on a titanium-mediated photocatalytic Diels-Alder reaction using UV light at 300nm, followed by oxidation with DDQ to achieve aromatization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anthrabenzoxocinones Supplier
The successful total synthesis of Anthrabenzoxocinones marks a new era for the availability of these potent antibiotic candidates. NINGBO INNO PHARMCHEM stands at the forefront of this technological advancement, offering comprehensive CDMO services to bring these molecules from the laboratory to the clinic. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with the highest standards of quality and efficiency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of intermediate meets the exacting requirements of global regulatory bodies.
We invite you to collaborate with us to leverage this innovative synthesis route for your drug discovery programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget for the development of next-generation antimicrobial therapies.
