Scalable Total Synthesis of Anthrabenzoxocinones and Fasamycin B Intermediates
The pharmaceutical landscape is constantly evolving with the demand for complex natural product derivatives that exhibit potent biological activity against resistant bacterial strains. Patent CN113045393B, published on July 8, 2022, introduces a groundbreaking synthetic methodology for aromatic polyketone compounds, specifically targeting the Anthrabenzoxocinones family including ABX-A, ABX-C, ABX-E, and BABX, as well as the Fasamycin and Naphthacemycin series. This intellectual property represents a significant leap forward in organic synthesis, offering a robust pathway to access these highly functionalized scaffolds which have demonstrated excellent activity against Methicillin-resistant Staphylococcus aureus (MRSA). For R&D directors and procurement specialists, understanding the nuances of this patent is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for drug discovery programs.
The development of efficient synthetic routes for complex polycyclic unsaturated ketal compounds has historically been fraught with challenges. Prior art, such as the total synthesis of Fasamycin A reported by the Kak-Shan Shia group, relied heavily on convergent strategies involving Hauser-Klaus cyclization. While scientifically elegant, these conventional methods often suffer from cumbersome fragment synthesis, limiting the ability to prepare substantial quantities of natural products or their derivatives. Furthermore, the reliance on toxic and hazardous reagents, notably potassium cyanide, poses significant safety and environmental compliance risks that are untenable for modern commercial scale-up of complex pharmaceutical intermediates. These limitations hinder the rapid exploration of structure-activity relationships (SAR) essential for advancing lead compounds.
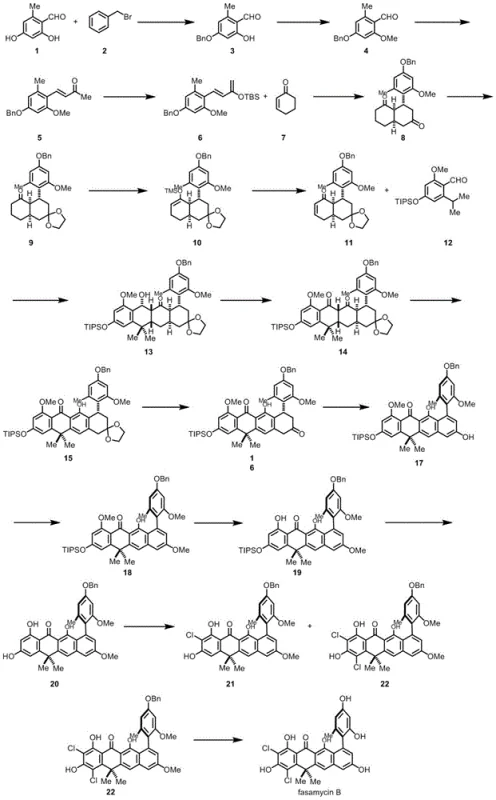
In stark contrast, the novel approach disclosed in CN113045393B utilizes a streamlined sequence centered around Diels-Alder cascades. The strategy initiates with an intermolecular Diels-Alder reaction between formula 1a and formula 2a to construct the initial cyclic framework. This is followed by a sophisticated intramolecular hetero Diels-Alder reaction that rapidly builds molecular complexity. By replacing hazardous cyanide chemistry with safer, catalytic processes, this new route not only enhances operator safety but also simplifies waste management protocols. The design allows for the modular assembly of the tetracyclic core, making it significantly easier to introduce structural modifications at various stages, thereby accelerating the timeline for medicinal chemistry optimization.
Mechanistic Insights into Titanium-Mediated Photocatalytic Cycloaddition
The cornerstone of this synthetic innovation is the titanium tetraisopropoxide-mediated photocatalytic Diels-Alder reaction. In this critical step, the enone intermediate (formula 12a) reacts with an aryl aldehyde derivative (formula 11a) under 300 nm ultraviolet irradiation. The Lewis acidic nature of the titanium species likely coordinates with the carbonyl oxygens, lowering the LUMO energy of the dienophile and facilitating the [4+2] cycloaddition under mild conditions. This photochemical activation allows for the construction of sterically congested quaternary centers with high stereocontrol, which is often difficult to achieve using thermal conditions alone. The subsequent oxidative aromatization using DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone) efficiently converts the cycloadduct into the fully aromatic anthrabenzoxocinone core found in formula 15a.
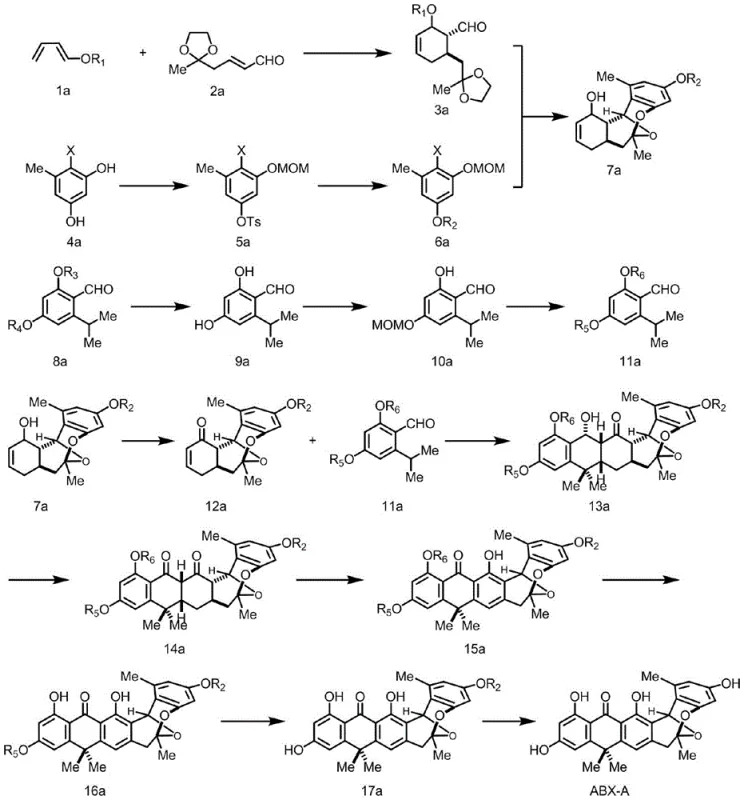
Impurity control is meticulously managed through the selection of protecting groups and oxidation states. The use of silyl ethers (TIPS, TBS) and benzyl groups provides orthogonal protection strategies that allow for selective deprotection later in the synthesis. For instance, the selective chlorination on the benzene ring to produce ABX-C and BABX from ABX-E is achieved using tert-butyl hypochlorite and n-butyllithium at low temperatures (0°C). This precision ensures that halogenation occurs only at the desired positions without affecting the sensitive epoxide or ketone functionalities elsewhere in the molecule. Such control over the impurity profile is vital for meeting the stringent purity specifications required for clinical grade API intermediates.
How to Synthesize ABX-A Efficiently
The synthesis of ABX-A involves a logical progression from simple starting materials to the complex natural product. The process begins with the preparation of key fragments via Diels-Alder chemistry, followed by their coupling and subsequent oxidative aromatization. The final steps involve global deprotection to reveal the active phenolic hydroxyl groups. This standardized approach minimizes the number of isolation steps and maximizes overall throughput. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below which outlines the critical process controls.
- Perform intermolecular Diels-Alder reaction between formula 1a and 2a at 100°C to obtain formula 3a.
- Execute nucleophilic addition and intramolecular hetero Diels-Alder reaction between formula 3a and 6a to generate formula 7a.
- Conduct titanium tetraisopropoxide-mediated photocatalytic Diels-Alder reaction followed by oxidative aromatization to yield formula 15a.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the methodology described in CN113045393B offers substantial strategic benefits. The reliance on cheap and easily obtained raw materials, such as simple aldehydes and dienes, reduces the vulnerability of the supply chain to fluctuations in exotic reagent pricing. By eliminating the need for transition metal catalysts that require expensive removal processes, the manufacturing cost structure is significantly optimized. This translates to a more competitive cost position for the final intermediates, allowing procurement managers to negotiate better terms while maintaining high quality standards. The avoidance of toxic cyanide reagents also reduces the regulatory burden and insurance costs associated with hazardous material handling.
Enhanced supply chain reliability is another critical advantage derived from this synthetic route. The robustness of the Diels-Alder reactions ensures consistent yields across different batches, reducing the risk of production delays. The scalability of the photochemical step, traditionally a bottleneck, is addressed by the specific conditions outlined in the patent, which utilize standard UV sources compatible with flow chemistry or large batch reactors. This adaptability means that suppliers can rapidly ramp up production from pilot scale to commercial tonnage without extensive re-engineering of the process. Consequently, lead times for high-purity pharmaceutical intermediates can be drastically reduced, ensuring continuity of supply for downstream drug development projects.
Furthermore, the environmental compliance profile of this method aligns with modern green chemistry principles. The reduction in hazardous waste generation and the use of safer solvents contribute to a lower environmental footprint. This is increasingly important for multinational corporations aiming to meet sustainability goals. The ease of purification, facilitated by the distinct polarity differences between intermediates, allows for efficient chromatography or crystallization steps. This efficiency not only saves time but also reduces solvent consumption, further driving down the total cost of ownership. Ultimately, this synthesis route provides a sustainable, scalable, and economically viable pathway for producing complex aromatic polyketones.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Anthrabenzoxocinones and related compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders assess the feasibility of integrating these intermediates into their existing pipelines. For more specific data regarding batch records or certificate of analysis (COA) parameters, direct consultation with the technical team is recommended.
Q: What are the key advantages of this synthesis method over prior art?
A: The method avoids toxic reagents like potassium cyanide used in previous Fasamycin A syntheses. It utilizes cheap, readily available raw materials and features a concise route amenable to large-scale preparation.
Q: Can this route produce derivatives beyond the natural products?
A: Yes, the key reaction intermediates are easily modified. The strategy allows for the synthesis of various aromatic polyketone natural products and their analogues, facilitating medicinal chemistry research.
Q: What is the role of titanium tetraisopropoxide in the reaction?
A: Titanium tetraisopropoxide acts as a mediator in the photocatalytic Diels-Alder reaction between formula 12a and formula 11a under 300 nm UV irradiation, enabling the construction of the complex polycyclic core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ABX-A Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex organic synthesis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We are fully equipped to implement the advanced photocatalytic and Diels-Alder methodologies described in CN113045393B, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch. Our commitment to excellence makes us the preferred choice for sourcing high-value pharmaceutical intermediates.
We invite you to contact our technical procurement team to discuss your specific requirements. We can provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized processes can reduce your overall expenditure. Don't hesitate to request specific COA data and route feasibility assessments to validate our capabilities against your internal standards. Let us collaborate to accelerate your drug discovery timeline with reliable, high-quality intermediates produced through state-of-the-art synthetic chemistry.
