Advanced Chemical Synthesis of Cholic Acid Intermediate A2 for Pharmaceutical Manufacturing
The pharmaceutical industry is currently witnessing a significant paradigm shift in the production of bile acid derivatives, driven by the urgent need for safer, virus-free supply chains. Patent CN115611961A, published in early 2023, introduces a robust chemical synthesis method for producing Cholic Acid Intermediate A2 and ultimately Cholic Acid itself. Unlike traditional methods that rely on the extraction of cholic acid from the internal organs of cattle or sheep—a process inherently fraught with the risk of viral contamination—this novel approach utilizes a common plant-source compound as the starting material. The technical breakthrough lies in the efficient conversion of 9α-hydroxy BA (Compound SM) through a series of controlled organic transformations, including oxidation, elimination, and stereoselective reduction. This patent not only addresses critical safety concerns regarding biological contaminants but also establishes a reproducible framework for the commercial scale-up of complex pharmaceutical intermediates. By transitioning from biological extraction to total chemical synthesis, manufacturers can achieve unprecedented consistency in impurity profiles and batch-to-batch reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the global supply of cholic acid has been dependent on the slaughterhouse industry, where bile is harvested from animal viscera. This dependency creates severe vulnerabilities in the reliable pharmaceutical intermediates supplier network. Firstly, the biological origin introduces an unavoidable risk of pathogen transmission, including viruses that may survive standard purification processes, posing a significant threat to patient safety in downstream drug formulations. Secondly, the availability of raw materials is subject to agricultural fluctuations, seasonal variations, and regulatory changes regarding animal by-products, leading to volatile pricing and inconsistent lead times. Furthermore, the purification of natural extracts often requires extensive chromatography to remove structurally similar bile acids like chenodeoxycholic acid or deoxycholic acid, resulting in lower overall yields and higher waste generation. These factors combined make the traditional extraction model unsustainable for modern, high-compliance pharmaceutical manufacturing environments that demand rigorous quality control and traceability.
The Novel Approach
The methodology disclosed in CN115611961A offers a transformative solution by establishing a fully synthetic route starting from plant-derived precursors. The core innovation involves the preparation of Cholic Acid Intermediate A2 via a mild elimination reaction, which serves as a pivotal branching point for synthesizing various bile acid derivatives. This chemical approach decouples production from the livestock industry, thereby stabilizing the supply chain and eliminating biological safety risks. The process utilizes readily available reagents such as concentrated sulfuric acid, glacial acetic acid, and specific oxidants under controlled temperatures ranging from 20°C to 50°C. By defining precise reaction parameters and workup procedures, including specific solvent exchanges and crystallization steps, the patent ensures that the intermediate A2 is produced with high purity. This shift enables cost reduction in pharmaceutical intermediates manufacturing by streamlining the purification process and allowing for continuous production schedules that are immune to agricultural supply shocks.
Mechanistic Insights into Acid-Catalyzed Elimination and Stereoselective Hydrogenation
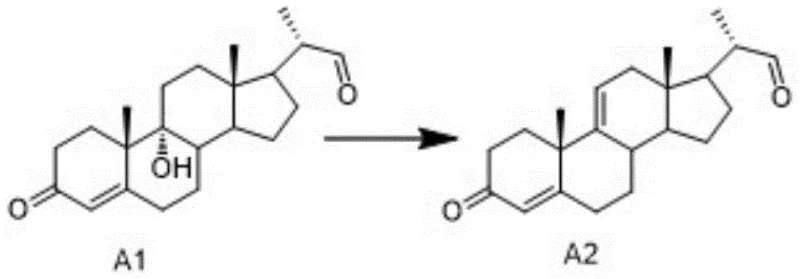
A critical component of this synthesis is the conversion of Compound A1 to Cholic Acid Intermediate A2, which proceeds via an acid-catalyzed elimination reaction. As illustrated in the reaction scheme below, the process involves treating the 9α-hydroxy precursor with concentrated sulfuric acid in the presence of glacial acetic acid or acetic anhydride. The mechanism likely proceeds through the protonation of the hydroxyl group followed by the loss of a water molecule to form a carbocation intermediate, which then undergoes deprotonation to establish the double bond characteristic of Intermediate A2. The patent specifies maintaining the reaction temperature between 20-50°C, preferably 35-40°C, to balance reaction kinetics with the stability of the steroid backbone. Excessive heat could lead to unwanted rearrangement or degradation of the sensitive polycyclic structure. The use of water or dichloromethane as a solvent system facilitates the dissolution of reagents while allowing for effective heat dissipation. Post-reaction treatment involves careful quenching in an ice bath and neutralization, ensuring that the acidic catalyst is completely removed before proceeding to subsequent steps, which is vital for preventing corrosion in large-scale reactors and ensuring product stability.
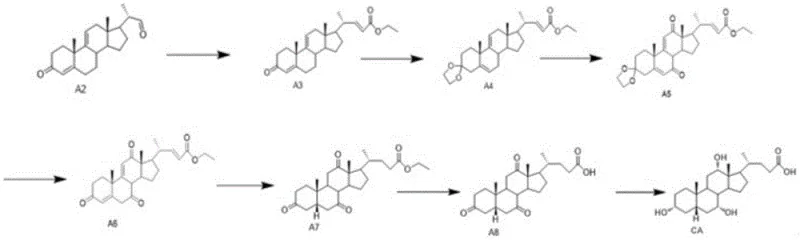
Furthermore, the patent details a highly sophisticated stereoselective hydrogenation step (conversion of A6 to A7) that is crucial for establishing the correct configuration at the C-5 position of the steroid nucleus. This step utilizes palladium on carbon (Pd/C) as a catalyst under hydrogen pressure of 0.4-0.45 MPa. What distinguishes this method is the specific solvent system comprising absolute ethanol and pyridine, potentially supplemented with additives like 1-methylimidazole or DMAP (4-Dimethylaminopyridine). The inclusion of these nitrogenous bases modulates the catalyst surface activity, favoring the formation of the desired 5α-H isomer over the 5β-isomer. Experimental data within the patent indicates that optimizing the ratio of ethanol to pyridine and adding DMAP can push the isomeric purity to an impressive 98.3:1.7 ratio. This level of stereocontrol is essential because the biological activity of bile acids is strictly dependent on their three-dimensional structure. Achieving such high diastereoselectivity without requiring difficult downstream separation techniques represents a significant advancement in process chemistry, directly impacting the efficiency and high-purity pharmaceutical intermediates output of the manufacturing line.
How to Synthesize Cholic Acid Intermediate A2 Efficiently
The synthesis of Cholic Acid Intermediate A2 is a multi-stage process that requires precise control over reaction conditions to ensure high yield and purity. The procedure begins with the oxidation of the starting material SM to intermediate A1, followed by the key elimination step to generate A2. Subsequent steps involve chain extension via Wittig reaction, protection group manipulation, and stereoselective reductions. Each stage has been optimized in the patent to minimize byproduct formation and simplify isolation procedures. For process engineers and R&D teams looking to implement this technology, understanding the specific stoichiometry, temperature profiles, and workup protocols is essential for successful technology transfer. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Oxidize compound SM (9α-hydroxy BA) using TEMPO/sodium hypochlorite or Jones reagent to obtain intermediate A1.
- Perform an elimination reaction on compound A1 using concentrated sulfuric acid and glacial acetic acid at 20-50°C to generate Cholic Acid Intermediate A2.
- Subject Intermediate A2 to Wittig reaction, ketal protection, oxidation, hydrolysis, hydrogenation, and selective reduction to yield final Cholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthetic route described in CN115611961A offers substantial strategic benefits beyond mere technical feasibility. The transition from animal extraction to chemical synthesis fundamentally alters the risk profile of the supply chain. By sourcing raw materials from plant-based or synthetic origins rather than animal organs, companies can mitigate the risks associated with zoonotic diseases and regulatory bans on animal by-products. This shift ensures a more predictable and stable supply of critical intermediates, reducing the likelihood of production stoppages due to raw material shortages. Additionally, the chemical process allows for better inventory management and forecasting, as production rates can be scaled according to demand rather than being limited by the availability of biological feedstock.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and complex purification steps required to separate cholic acid from other naturally occurring bile acids. The use of mild reaction conditions, such as the elimination reaction at 20-50°C, reduces energy consumption compared to high-temperature processes. Furthermore, the high stereoselectivity achieved in the hydrogenation step minimizes the loss of material due to isomer formation, thereby improving overall mass balance and reducing the cost of goods sold. The avoidance of heavy biological waste disposal also contributes to lower operational expenditures related to environmental compliance.
- Enhanced Supply Chain Reliability: Relying on chemical synthesis decouples the production of cholic acid intermediates from the volatility of the agricultural sector. This independence ensures that reducing lead time for high-purity pharmaceutical intermediates becomes achievable, as production schedules are no longer subject to seasonal slaughtering cycles or livestock disease outbreaks. The ability to produce intermediates like A2 on demand allows for a more responsive supply chain that can adapt quickly to fluctuations in market demand for downstream bile acid therapeutics.
- Scalability and Environmental Compliance: The processes described utilize standard organic solvents and reagents that are compatible with existing chemical infrastructure, facilitating easy scale-up from kilogram to tonne quantities. The reaction conditions are designed to be robust, with wide operating windows that accommodate large-scale reactor dynamics. Moreover, the synthetic route generates less biological waste compared to extraction methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The ability to recycle solvents and manage chemical waste streams effectively enhances the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cholic Acid Intermediate A2. These answers are derived directly from the experimental data and specifications provided in patent CN115611961A, offering clarity on reaction mechanisms, purity standards, and process scalability. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this synthetic route into their existing manufacturing portfolios.
Q: What is the primary advantage of synthesizing Cholic Acid chemically versus extraction?
A: Chemical synthesis eliminates the risk of virus infection associated with extracting cholic acid from animal viscera (bile), ensuring a safer and more consistent supply chain for pharmaceutical applications.
Q: How is stereochemical purity controlled during the hydrogenation step?
A: The patent specifies using a solvent system of absolute ethanol, pyridine, and additives like DMAP or 1-methylimidazole during Pd/C catalyzed hydrogenation. This specific mixture optimizes the 5α-H isomer purity up to 98.3:1.7 ratios.
Q: What are the reaction conditions for converting Intermediate A1 to A2?
A: The elimination reaction is conducted under mild conditions, typically between 20-50°C, using water or dichloromethane as a solvent with concentrated sulfuric acid and glacial acetic acid or acetic anhydride.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cholic Acid Intermediate A2 Supplier
The technological advancements detailed in CN115611961A represent a significant opportunity for pharmaceutical manufacturers to secure a safer and more efficient supply of bile acid derivatives. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of steroid chemistry, including the precise temperature control and specialized hydrogenation setups needed for this synthesis. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch of Cholic Acid Intermediate A2 meets the highest international standards for pharmaceutical use.
We invite potential partners to engage with our technical procurement team to discuss how this synthetic route can be tailored to your specific production needs. By collaborating with us, you can access a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this virus-free synthetic method. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of our intermediates with your downstream processes. Together, we can build a resilient supply chain that delivers high-quality cholic acid derivatives while minimizing risk and maximizing efficiency.
